How ferroptosis entered our CLL research program: from redox biology to therapeutic vulnerability
Published in Cancer, Cell & Molecular Biology, and General & Internal Medicine
The idea for this project began in 2020 with a simple question: could ferroptosis matter in chronic lymphocytic leukemia (CLL)? At first glance, this may have looked like an unusual direction for a CLL laboratory. But in reality, it grew very naturally out of a much longer scientific path in our group. For years, we had been working on CLL biology, immunometabolism, stromal interactions, and oxidative stress. Even before our CLL-focused program gained momentum around 2014, Prof. Dimitrios Mougiakakos had already been deeply interested in redox biology during his postdoctoral work at the Karolinska Institute in Stockholm in 2009, where he studied oxidative stress in T cells. I was fortunate to be his master's student there at the time. Looking back, that early exposure to redox biology strongly shaped how both of us later thought about malignant hematology.
From 2013 onward, our group increasingly focused on the metabolic vulnerabilities of CLL. Earlier studies had shown that CLL cells are characterized by oxidative stress and altered mitochondrial metabolism, that energy metabolism differs between biological subgroups, and that stromal interactions profoundly shape leukemia cell behavior. This work - some of it performed together with colleagues who are also co-authors on the present paper, including Wolfgang Huber, Thorsten Zenz, and Junyan Lu – contributed to the conceptual basis for the current study. By 2020, it had become clear to us that CLL cells in the peripheral blood display active oxidative phosphorylation and substantial oxidative stress, whereas stromal niches can buffer these stress states and support survival. Around the same time, ibrutinib had already changed CLL therapy, not only through BTK inhibition but also by mobilizing CLL cells out of protective tissue niches. These pieces fit together into a hypothesis: perhaps ferroptosis is a real vulnerability in CLL, and perhaps the stromal compartment protects against it.
I was encouraged by my mentor and current chair, Prof. Mougiakakos, to turn it into a grant application. Because this was a high-risk idea, we applied for an European Hematology Association (EHA) Kick-Off Grant, which is specifically designed to support bold and risky projects, and the first experiments were launched in late 2020 to generate preliminary data. I still remember how happy I was when the grant was approved in 2021. At the annual congress of the European Hematology Association (EHA) in Vienna that year, another important piece of the story fell into place. During the award ceremony, I met Paul Hengeveld from Ton Langerak's group at Erasmus UMC Rotterdam. In conversation, he mentioned that he had observed differences in the transferrin receptor system between IGHV-mutated and IGHV-unmutated CLL cases which might be of relevance to my project. That immediately caught my attention. IGHV status is one of the most important biological and prognostic classifiers in CLL, and transferrin receptor biology is intimately linked to ferroptosis. So, almost on the spot, this observation became part of our project.
Science, however, rarely progresses in a straight line. At the beginning of 2022, our lab moved to a new location, and I simultaneously became director of the new laboratory. We had to rebuild workflows that had worked well before, re-establish assays, and train new colleagues. This delayed the project considerably, and there were moments when it felt as though the elegant idea on paper would take much longer than expected to become real data. But by mid-2022, the project was finally running at full speed. I was very fortunate to work with exceptionally dedicated technicians who performed the experiments with great care and reliability, while I had constant scientific support from my mentor, colleague, and friend Prof. Mougiakakos.
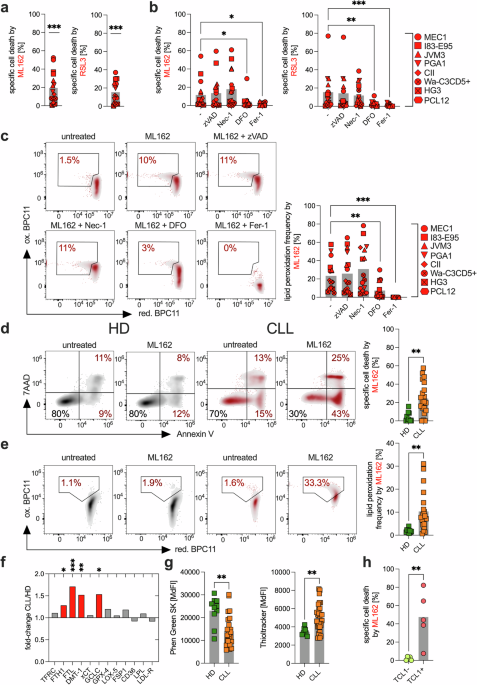
Once the study was fully underway, the biology became increasingly exciting. We found that CLL cells indeed carry several features that predispose them to ferroptosis, including elevated basal lipid peroxidation and increased levels of labile iron. At the same time, we saw that the stromal compartment can protect CLL cells by reinforcing antioxidant defenses, boosting glutathione-related pathways, and improving access to cystine and cysteine. This was satisfying because it matched our original conceptual framework: ferroptosis sensitivity in CLL is not fixed, but shaped by the microenvironment.
We also found that IGHV status mattered in a very functional way. CLL with mutated IGHV genes displayed higher transferrin receptor expression and greater ferroptosis sensitivity than unmutated cases. By contrast, the more therapy-refractory IGHV-unmutated subgroup showed relative resistance. Importantly, this resistance was not absolute. We could sensitize these cells either through ibrutinib-mediated changes in transferrin receptor biology or through metabolic targeting of ACSL1-associated fatty acid pathways. That was one of the most rewarding aspects of the work: the biology was not only interesting, it also pointed toward therapeutic ideas for exactly the patients who need better options.
A particularly productive phase of the project began in 2023, when Lea Reemts joined our lab as a master's student. I assigned the stromal compartment analyses to her project - and this turned out to be a real stroke of luck. She learned very quickly and, within only six months, generated a remarkable amount of data under my supervision. By then, the project had grown from an initially risky idea into a broad and mechanistically layered story that combined ex vivo work, stromal coculture systems, multi-omics input, and in vivo experiments. The progress and promise of the work also helped us secure follow-up funding from the Wilhelm Sander-Foundation, which started in 2024 and provided important support for further developing the project.
The publication process then became its own lesson in persistence. Our first submission in early 2025 was rejected, although the reviewers' comments were scientifically addressable. We used that feedback to strengthen the manuscript, submitted to another journal, and were rejected again by the editor. A third attempt led to another editorial rejection. At that point, we did not abandon the study - we expanded it. Lea had by then continued in our lab as a PhD student after successfully defending her master's thesis, and together with the whole team we added even more data to further solidify the story.
When we finally submitted the manuscript to STTT in July 2025, it entered peer review - which already felt like progress. Then came one of the most stressful moments of the entire project: the reviewer comments arrived just two days before I was due to leave for vacation, and the revision window was very tight. Together with my mentor, I designed the revision strategy and the necessary experiments, and Lea and our technicians executed them with incredible commitment. Some reviewer requests were extremely demanding, including genetic perturbation experiments that were not yet fully established in our lab. Still, the team managed to address almost all major points within the deadline. After the revised submission, one reviewer remained unconvinced and requested even more evidence, especially regarding genetic modification. Eventually, we were able to address these concerns as well, and in December 2025 the manuscript was accepted. The relief and joy in the group were enormous.
This project is also a story of collaboration. Throughout the study, we benefited from long-standing and newly formed partnerships - with Prof. Langerak and his group in Rotterdam, the Human Genetics Department at our medical faculty, the Fraunhofer team in Regensburg, Prof. Huber and Dr. Lu in Heidelberg, Prof. Zenz in Zurich, and Prof. Dietrich in Duesseldorf. Some of these collaborations had been built over many years through earlier CLL and immunometabolism projects, others developed specifically through this work. Together, they made it possible to connect cell death biology, microenvironmental signaling, clinical subgrouping, and translational ideas in a way that no single lab could easily achieve alone.
What motivated us throughout was the hope that better understanding of ferroptosis biology could eventually help patients with CLL, especially those with high-risk disease. Our study does not offer an immediate cure, but it identifies therapeutically actionable nodes and shows that ferroptosis sensitivity in CLL is structured by IGHV biology and stromal protection. For a disease in which allogeneic stem cell transplantation is still the only curative option - and one that many elderly patients cannot tolerate - every credible new therapeutic angle matters. That is why this project meant so much to us, scientifically and personally.
Follow the Topic
-
Signal Transduction and Targeted Therapy
This is an international, peer-reviewed, open-access journal publishing articles related to signal transduction in physiological and pathological processes, alongside signal transduction-targeted therapeutics in the form of biological agents and small molecular drugs used to treat human diseases.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in