Learning from a 19-year Experiment
Published in Ecology & Evolution

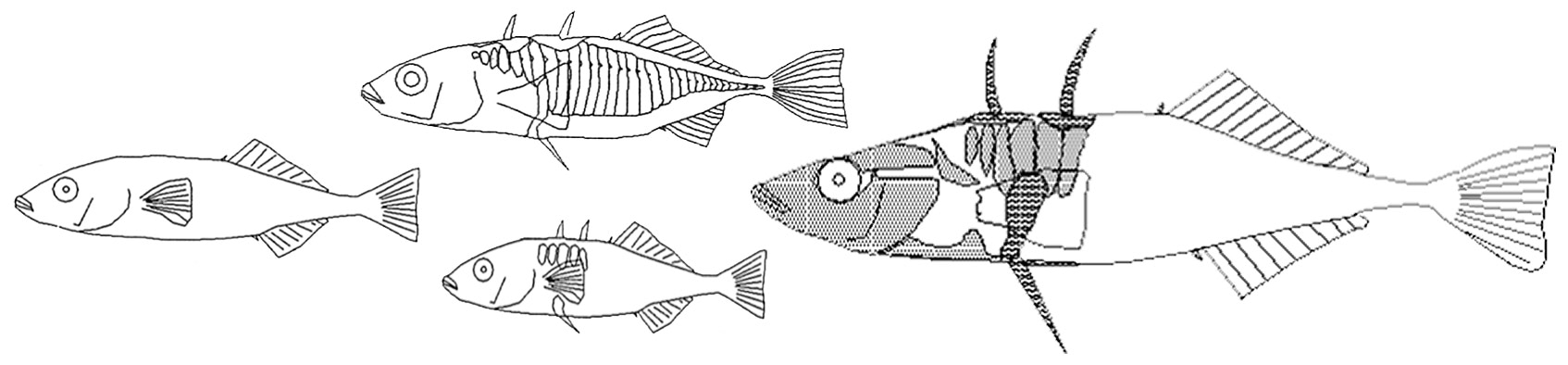
Phenotypic diversity of threespine stickleback in the adaptive radiation on Haida Gwaii. Drawings: Thomas E. Reimchen.
The background for this study began to emerge in the 1960s when Ric Moodie and Tom Reimchen, the former doing his PhD and the latter being a summer assistant at the University of Alberta, Canada, began collections of threespine stickleback from pristine lakes on the Haida Gwaii archipelago (formerly known as Queen Charlotte Islands). During these initial surveys of about 20 localities, they discovered a remarkable diversity of body forms even among adjacent lakes within a small watershed and which was greater than the diversity observed in stickleback populations among watersheds from throughout Europe. After completing his PhD in England on ecological genetics of intertidal snails, Tom Reimchen returned to the archipelago in 1974 and undertook over the next 15 years a survey of some 700 lakes and ponds in this remote region. About 140 of these had stickleback, most of which were distinctive from each other. These were found in 56 different watersheds separated by marine waters and were eventually shown to represent multiple replicated colonizations of freshwater by the widely distributed marine populations of stickleback1.

Tom Reimchen collecting stickleback in the Haida Gwaii archipelago in the 1970s-1980s.
Of great interest was the extensive concordance between the distinctive characteristics of stickleback in each population and the selective landscape unique to each locality. The high replication in this system allowed Tom Reimchen to identify major predictors for this concordance: aquatic spectra (deeply stained blackwater to high clarity), habitat (stream, pond or lake) and predation landscape (fish-, bird- or macroinvertebrate predators)2. This finding emphasized the efficacy and strength of natural selection and while consistent with other studies of adaptive radiation such as Galapagos finches3 and Caribbean Anolis lizards4, the stickleback differentiation involved multiple character complexes including variation in body size and shape, fin size and shape, nuptial coloration, defense and trophic morphology as well as life history2.

Haida Gwaii harbors a diversity of freshwater habitats, from large, deep lakes (left), to small shallow ponds (top middle), to streams (top right). In 1993, Tom Reimchen introduced 100 giant stickleback into a small, shallow clearwater pond (bottom right, Roadside Pond with co-author David Marques for size comparison). Photos: Thomas E. Reimchen (left), Gabriela Blatter (bottom right), David A. Marques.
This diversity of Haida Gwaii stickleback must have evolved over the last 12,000 years following glacial retreat and replicated colonization of vacant habitats by the marine stickleback population. But how fast has adaptation really been? Did the diversity of phenotypes evolve in the first few generations, for example by plasticity, in the first tens of generations from the assembly and assortment of standing genetic variation or in hundreds or thousands of generations of adaptation by de novo mutations? To test the speed of adaptation, Tom Reimchen set up a real-time selection experiment along the three major axis of selection - aquatic spectra, habitat and predation landscape. He chose a highly derived phenotype as source population: stickleback from Mayer Lake, a large and deep blackwater lake with plenty of fish and bird predators, where giant stickleback have evolved that specialize on zooplankton feeding and have become strongly melanic. Identifying an appropriate pond for a transplant was challenging as it had to be ecologically opposite to the selective landscape in the large lake where the source populations was obtained and had to meet basic criteria for any acceptable transplant experiment (eg: fully isolated without inlet or outlet streams and no resident fish, etc). Eventually, Tom identified Roadside Pond as an ideal location for the transplant - a small, shallow clearwater pond dominated by macroinvertebrate predators. Permits for this experiment were granted and Tom released 100 giant stickleback from Mayer Lake into Roadside Pond in 1993.

Mayer Lake (left) is a large, deep blackwater lake with a rich vertebrate predator community and harbors the source population of giant, melanistic stickleback used in the experiment. Roadside Pond (right) is a small, shallow clearwater pond with invertebrate predators. Photos: David A. Marques.
Since 1993, Tom Reimchen has followed the phenotypic evolution of the Roadside Pond population and 19 years after the introduction, in 2012, 11 individuals were used for whole-genome sequencing in collaboration with David Kingsley and Felicity Jones at Stanford University and Federica di Palma at the Broad Institute (now at Earlham Institute). In the same year, Stephen Leaver and Tom Reimchen showed that the phenotype of Roadside Pond stickleback evolved rapidly and in the expected direction: stickleback had shorter spines, less and smaller plates as expected by the lack of vertebrate predators, they showed shorter gill rakers and longer jaws as expected from a shift from zooplankton to invertebrate diet, and they reduced black coloration as expected from a shift from black- to clearwater5. Phenotypic change was ~30% of the differences observed between natural lake and pond populations.
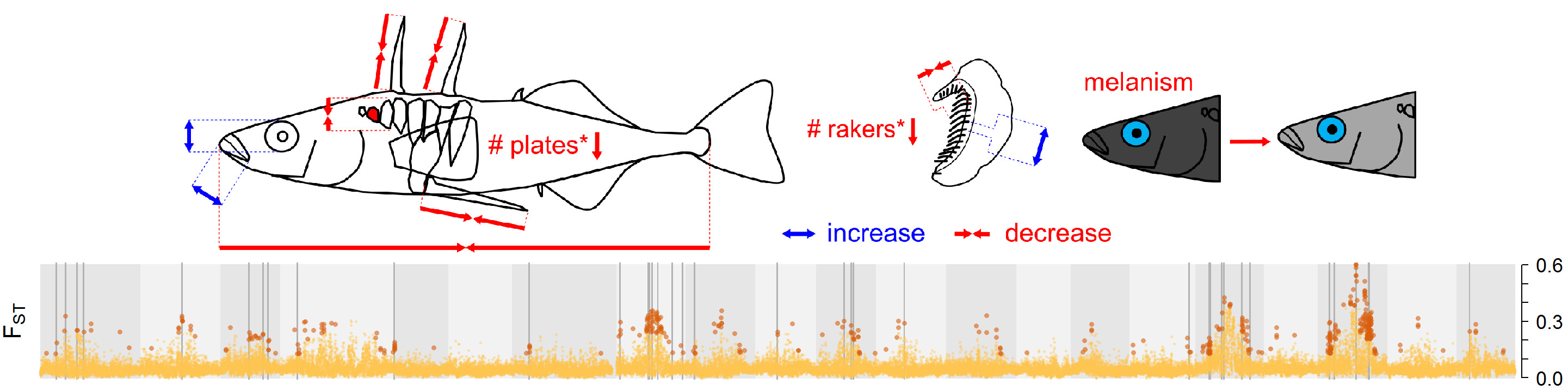
Phenotypic change and genomic differentiation (FST) between the source and transplant populations after 19 years of evolution in isolation. Grey bars indicate likely genomic targets of divergent selection across the 21 stickleback chromosomes (grey shading). Figures: Marques et al. (2018)6.
In our paper now published in Nature Ecology & Evolution6, David Marques analyzed the genomic evolution in the experiment. We found that adaptation is not only fast and repeatable at the phenotypic level, but also at the genomic level. 19 years of evolution have taught us that even a derived adaptive radiation member such as the giant blackwater stickleback from Mayer Lake can maintain the genetic variation necessary to rapidly adapt to a new environment on the opposite side of the three axes of selection. The full story can be read here6.
Text: Tom Reimchen & David Marques
1 Deagle et al. (2013): Phylogeography and adaptation genetics of stickleback from the Haida Gwaii archipelago revealed using genome-wide single nucleotide polymorphism genotyping. Molecular Ecology 22:1917-1932.
2 Reimchen et al. (2012): Natural selection and the adaptive radiation of Haida Gwaii stickleback. Evolutionary Ecology Research 15:241-269.
3 Grant (1981): Speciation and the adaptive radiation of Darwin's finches. American Scientist 69:653-663.
4 Losos et al. (1998): Contingency and determinism in replicated adaptive radiations of island lizards. Science 279:2115-2118.
5 Leaver et al. (2012): Abrupt changes in defence and trophic morphology of the giant threespine stickleback (Gasterosteus sp.) following colonization of a vacant habitat. Biological Journal of the Linnean Society 107:494-509.
6 Marques et al. (2018): Experimental evidence for rapid genomic adaptation to a new niche in an adaptive radiation. Nature Ecology & Evolution, doi:10.1038/s41559-018-0581-8.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in