Medulloblastoma and high-grade glioma organoids for drug screening, lineage tracing, co-culture and in vivo assay
Published in Protocols & Methods
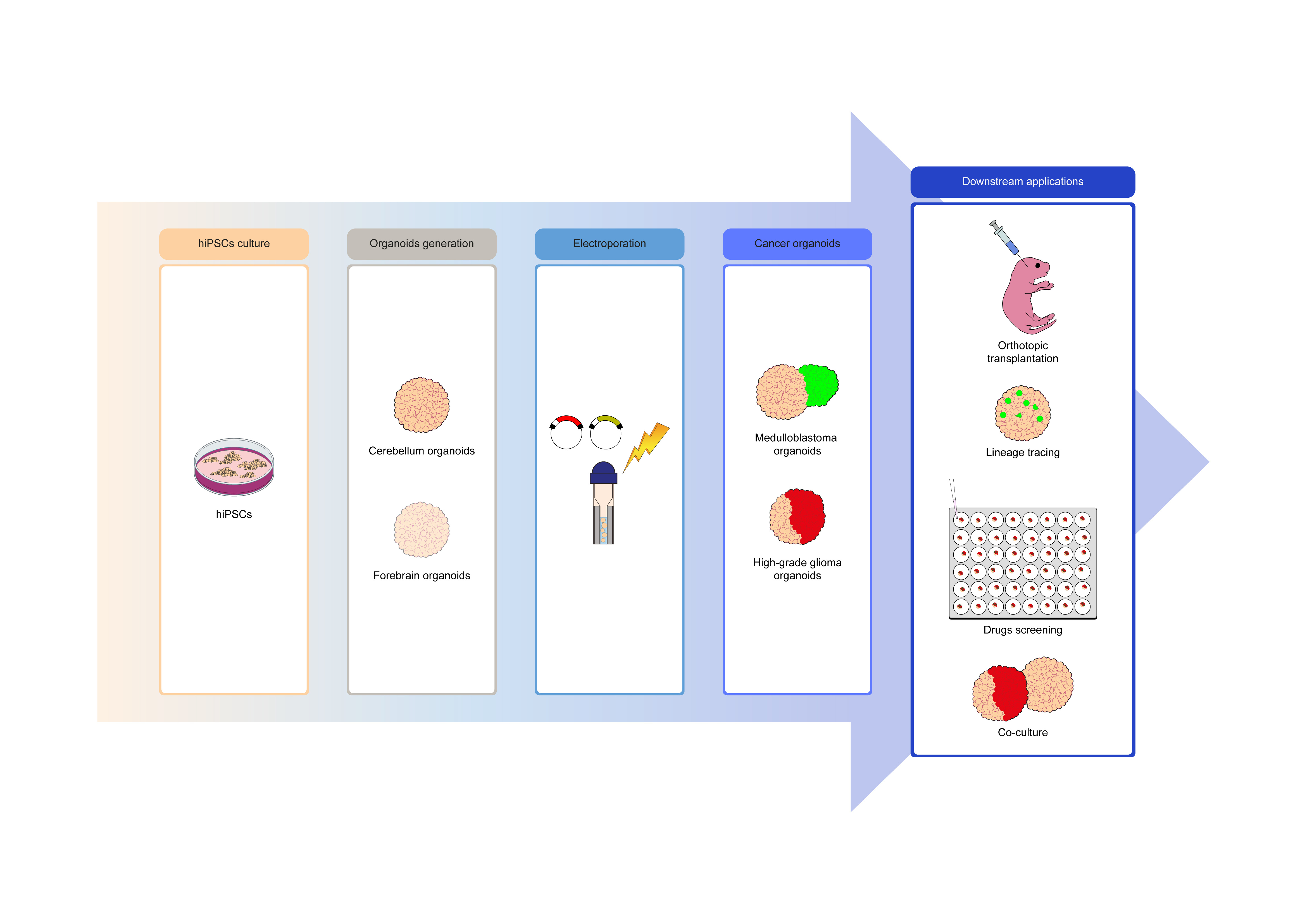
Medulloblastoma (MB) and high-grade glioma (HGG) are the most aggressive and frequent solid tumors affecting children during pediatric age. In the last years, several models have been established to study these types of cancers. Among them, we find organoids, artificial mini-organs that can be grown in vitro and replicate the physiological, molecular, and pathological characteristics of the human organ they mimic. Recently, they have been shown to be a valid model to study several aspects of brain cancer biology and genetics as well as to test therapies. Brain cancer organoids can be generated using genetically modified cerebral organoids differentiated from human induced pluripotent stem cells (hiPSCs). However, protocols to generate brain cancer organoids are rare, as well as protocols describing their possible downstream applications.
In a series of recent studies, we have shown that both cerebellum and forebrain organoids, derived from hiPSCs, can be quickly electroporated to introduce genetic constructs, not only to induce cancer transformation1–3 but also to test different characteristics of the cancer they are modeling, from the susceptibility to different drug treatments1,3 to the interactions with an organism, if engrafted in immunocompromised mice1,3 or with the healthy counterpart when co-culture of tumoral and healthy organoids is performed3. Furthermore, we used brain cancer organoids to perform lineage tracing2,4.
Using genetically modified organoids to model brain cancer is quite a recent approach. Healthy cerebellum and forebrain organoids are generated as previously described by Muguruma et al.5 and Velasco et al.6, respectively. For the genetic modification of organoids, we developed a new buffer that enables the electroporation of cerebellum and forebrain organoids. The PiggyBac (PB) transposon system is used to achieve a continuous and stable overexpression of the genes of interest over time, mimicking the amplification/overexpression of such oncogenes in patients. Indeed, the advantage of the PB transposon system is the multicopy integration within the cell's genome allowing an overexpression of the genes of interest. Furthermore, the PB transposon system coupled to the electroporation is safer than the viral systems for integrating the genes of interest within the genome of target cells. To facilitate the recognition of electroporated cells, fluorescent reporter genes (e.g., Venus or mCherry) are co-electroporated. This allows an easy visualization over time of the expansion and growth of the cells in which oncogenes are overexpressed.
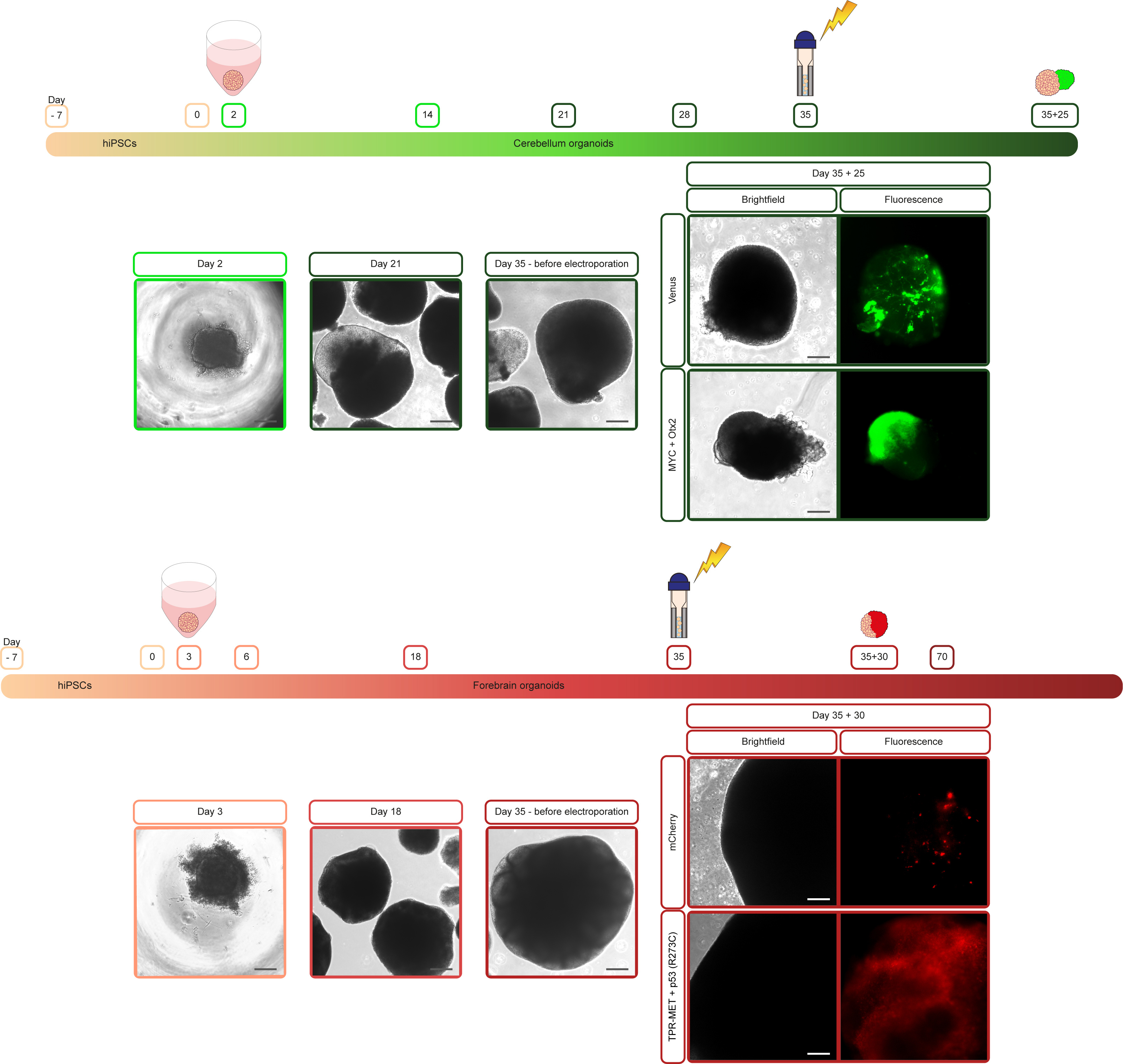
Workflow for generation of cerebellum and forebrain organoids and their electroporation to obtain MB and HGG cancer organoids.
The protocol we developed can be adapted to address different developmental or biological questions. Genetically modified organoids can also be used by developmental biologists aiming to perform lineage tracing experiments and live imaging using normal cerebellum and forebrain organoids even without inducing a cancer-like phenotype, e.g., by applying the Cre-LoxP system2,4. Testing different combinations of genes as cancer inducers in organoids also allows assaying whether such cancer organoids show tumorigenicity in vivo. We used orthotopic transplantation of cancer organoids into cerebellum1 or cerebral cortex3 to test growth, invasion, and spreading of cancer cells in a more physiological environment. We showed that this analysis can be done also in a context where specific populations of cells can be ablated, assaying in vivo their specific contribution. This can be done using the expression of a DTA-p27 construct to remove quiescent cells, thus reducing infiltration of organoid-derived cancer cells in vivo3.
Compared to other methods, this protocol shows how different types of central nervous system (CNS) organoids, i.e., cerebellum1,2 and forebrain3 organoids, can be more quickly and easily electroporated. Orthotopic engraftment of organoids pieces in immunodeficient mice allows for their three-dimensional tumoral structure maintenance, enabling to study how they interact and behave in the ‘native’ tissue with its different cell types. We show that co-culture of a whole brain cancer organoid and a normal one can be set up and used for live imaging of infiltrating cells3. Using whole organoids might represent a new technology to minimize the perturbation of the environment of cancer organoids that could lead to a cellular selection. Lastly, our protocol can be used to set up a platform for drug screening by employing a fluorescent reporter to monitor the effect of the compounds overtime on the same organoids1.
References
- Ballabio, C. et al. Modeling medulloblastoma in vivo and with human cerebellar organoids. Nat. Commun. (2020) doi:10.1038/s41467-019-13989-3.
- Ballabio, C. et al. Notch1 switches progenitor competence in inducing medulloblastoma. Sci. Adv. 7, (2021).
- Antonica, F. et al. A slow-cycling/quiescent cells subpopulation is involved in glioma invasiveness. Nat. Commun. 2022 131 13, 1–15 (2022).
- Zhang, T. et al. Generation of excitatory and inhibitory neurons from common progenitors via Notch signaling in the cerebellum. Cell Rep. 35, 109208 (2021).
- Muguruma, K., Nishiyama, A., Kawakami, H., Hashimoto, K. & Sasai, Y. Self-organization of polarized cerebellar tissue in 3D culture of human pluripotent stem cells. Cell Rep. 10, 537–550 (2015).
- Velasco, S. et al. Individual brain organoids reproducibly form cell diversity of the human cerebral cortex. Nature (2019) doi:10.1038/s41586-019-1289-x.
Follow the Topic
-
Nature Protocols
This journal publishes secondary research articles and covers new techniques and technologies, as well as established methods, used in all fields of the biological, chemical and clinical sciences.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in