Mild aqueous activation unleashes the potential of light alkanes
Published in Chemistry

In Prof. Qi Lu's research group at Tsinghua University, our focus lies in discovering renewable methods for utilizing CO2 and light alkanes. While working on a project on the electrochemical activation of propane1, we serendipitously found this novel aqueous system that can activate light alkanes under room temperature and ambient pressure using only commercially available Cu powder as the catalyst and O2 as the oxidant.
Why activating light alkanes
The rapid growth of shale gas exploitation has resulted in a substantial rise in the production of light alkanes, such as methane, ethane, and propane. These versatile hydrocarbons play a vital role as valuable feedstocks, meeting the increasing energy and chemical demands in diverse industries. On-site utilization of these resources offers advantages by reducing transportation requirements, improving operational efficiency, and minimizing costs and environmental consequences. Embracing sustainable conversion technologies enables us to maximize resource utilization, lower emissions, and foster an efficient and environmentally conscious energy and chemical sector.
Challenges of traditional approaches
While light alkanes present a valuable resource, finding economically viable and environmentally friendly methods for their on-site utilization and storage remains a challenge. Traditional approaches for converting light alkanes into high-value liquid fuels and commodity chemicals often require harsh conditions involving high temperatures and pressures. Conventional methods for converting light alkanes include dehydrogenation, cracking, or partial oxidation, necessitating elevated temperatures and pressures2-7. While these reactions can produce desirable products, they require significant energy inputs and specialized infrastructure. Furthermore, these processes are typically carried out in large-scale centralized plants, limiting the utilization of light alkanes from remote oil or gas fields. In this context, our research explores a promising alternative: an aqueous system for the mild activation of light alkanes, enabling their conversion into valuable oxygenates and olefins.
What's new in our study
In our study, we present a novel aqueous system that enables the activation of light alkanes under ambient conditions. Leveraging copper (Cu) as the catalyst and gaseous oxygen (O2) as the oxidant, we achieved remarkable conversions and selectivities for different light alkanes.
-
Key findings
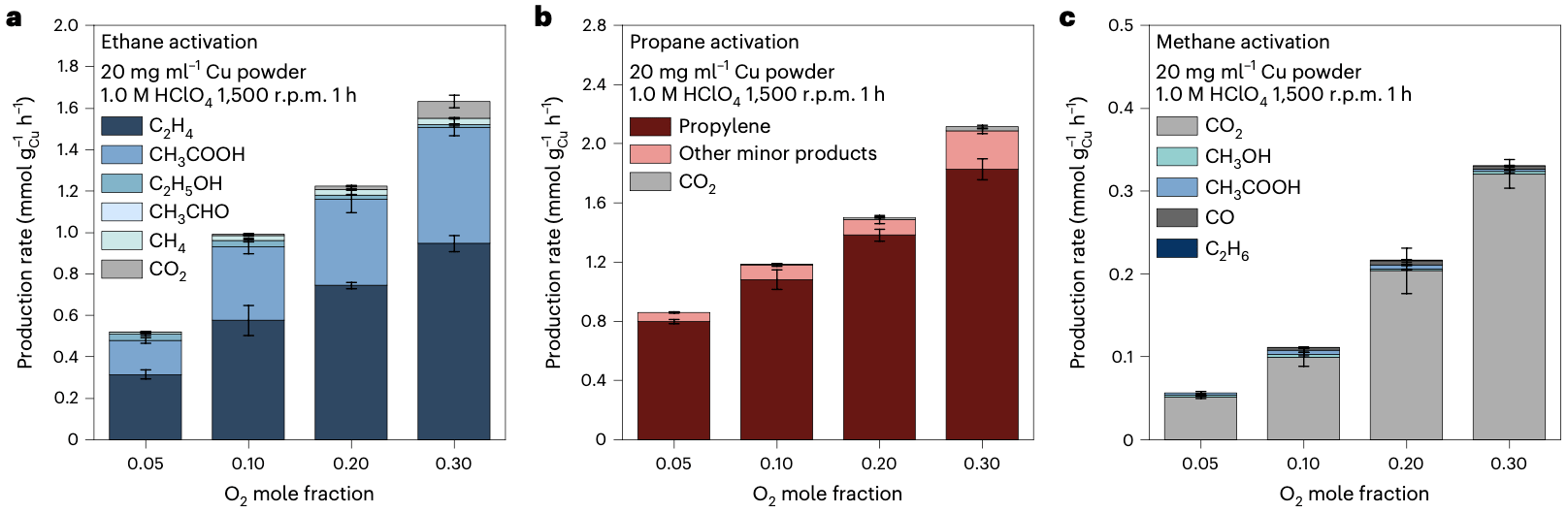
Ethane, under our mild aqueous conditions, was successfully converted into ethylene and acetic acid with an impressive conversion rate of 2.27 mmol·gCu−1·h−1 and a combined selectivity of up to 97%. Similarly, propane demonstrated high conversion rates of up to 1.83 mmol·gCu−1·h−1, yielding propylene with a selectivity of up to 94%. In the case of methane, the main products of activation were carbon dioxide, methanol, and acetic acid.
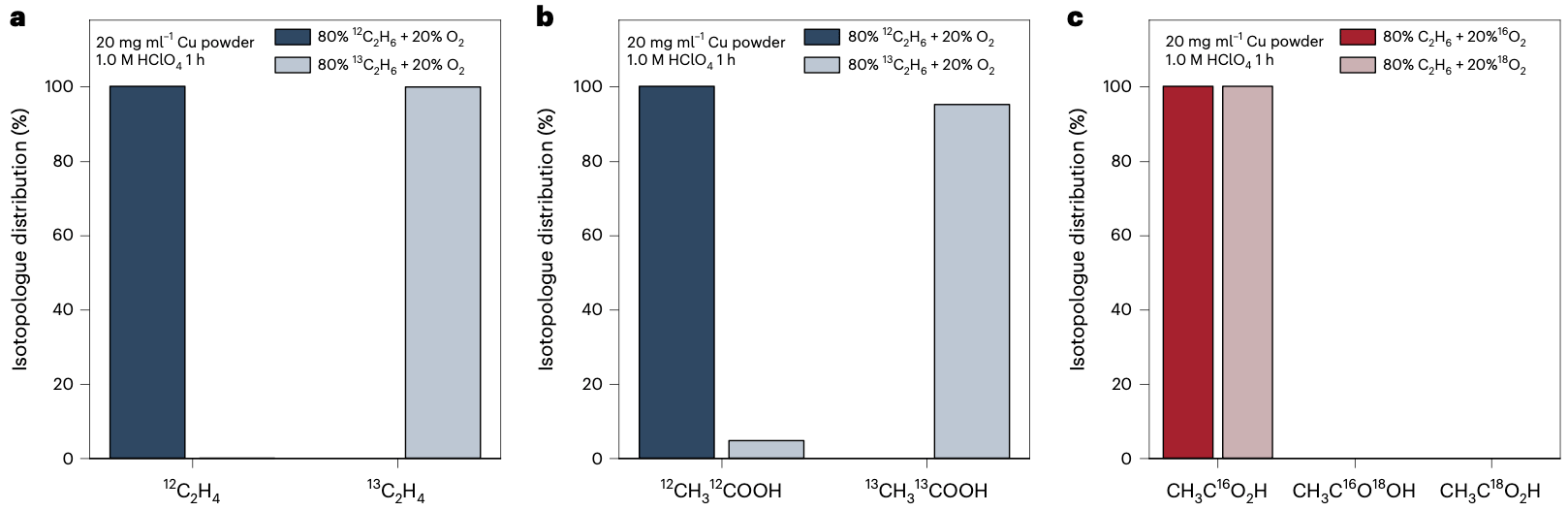
To elucidate the reaction intermediates and investigate the underlying mechanism, we conducted isotopic labeling experiments. Using 13C2H6 as the gas feed, we confirmed that ethane was indeed the carbon source for the products we observed. Interestingly, we found that no acetic acid is 18O-labeled when 18O2 was used, indicating that water, rather than gaseous O2 is the oxygen source for the oxygenates produced from ethane activation.
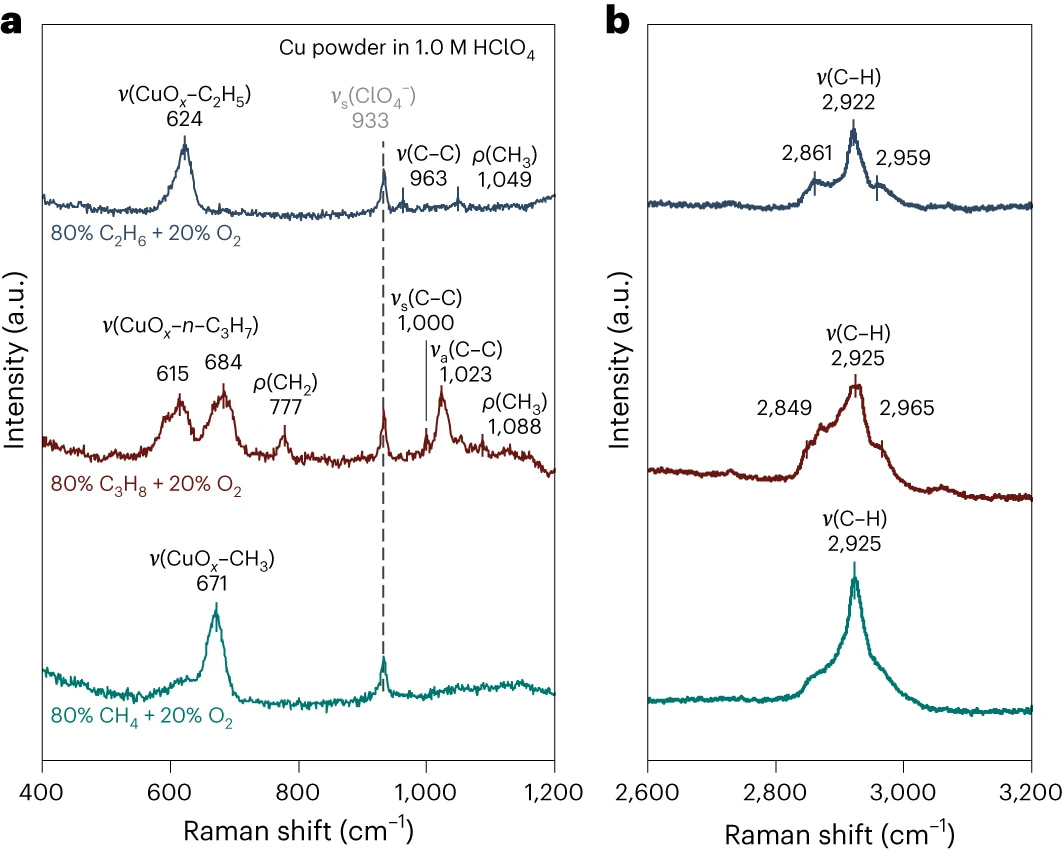
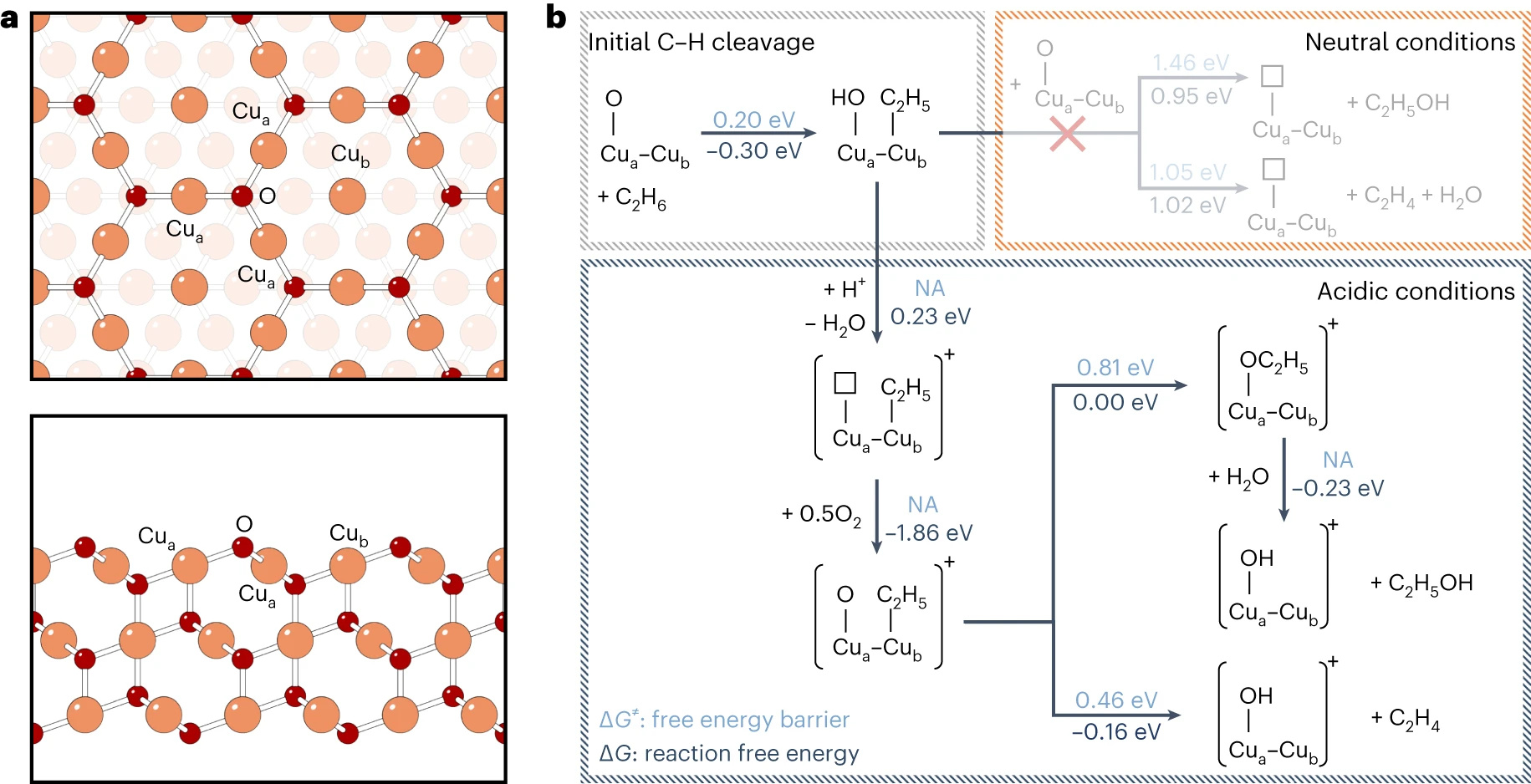
To gain deeper insights into the process of light alkane activation, we employed in situ surface-enhanced Raman spectroscopy. Through a series of control spectroscopic experiments, we successfully identified alkyl groups bound to the Cu oxide surface as the key reaction intermediates. These findings were further substantiated by density functional theory calculations.
-
Significance and implications
The development of this mild aqueous system for light alkane activation holds great promise for the on-site utilization and storage of these valuable resources. By operating under room temperature and ambient pressure conditions, we have overcome the limitations associated with traditional approaches. Our results demonstrate the feasibility of selectively converting light alkanes into valuable oxygenates and olefins, paving the way for more sustainable and cost-effective processes. This approach has the potential to unlock new opportunities for the utilization of light alkanes, particularly in remote oil or gas fields where large-scale centralized plants are impractical.
Conclusion and outlook
Our research showcases a groundbreaking aqueous system for the mild activation of light alkanes, offering a viable pathway for their conversion into oxygenates and olefins. By utilizing Cu as the catalyst and gaseous O2 as the oxidant, we have achieved impressive conversion rates and selectivities under ambient conditions. The development of this process marks a significant step towards the on-site utilization of light alkanes, presenting economic and environmental advantages over traditional methods. Further exploration of this aqueous system and its potential applications may lead to transformative advancements in alkane activation and utilization.
References
1. Zhang, H., Li, C., Lu, Q., Cheng, M. J. & Goddard, W. A., III. Selective Activation of Propane Using Intermediates Generated during Water Oxidation J. Am. Chem. Soc. 143, 3967-3974 (2021).
2. Galvita, V., Siddiqi, G., Sun, P. & Bell, A. T. Ethane dehydrogenation on Pt/Mg(Al)O and PtSn/Mg(Al)O catalysts. J. Catal. 271, 209-219 (2010).
3. Sun, P., Siddiqi, G., Chi, M. & Bell, A. T. Synthesis and characterization of a new catalyst Pt/Mg(Ga)(Al)O for alkane dehydrogenation. J. Catal. 274, 192-199 (2010).
4. Phadke, N. M., Mansoor, E., Bondil, M., Head-Gordon, M. & Bell, A. T. Mechanism and Kinetics of Propane Dehydrogenation and Cracking over Ga/H-MFI Prepared via Vapor-Phase Exchange of H-MFI with GaCl3. J. Am. Chem. Soc. 141, 1614-1627 (2019).
5. Sobolev, V. I., Dubkov, K. A., Panna, O. V. & Panov, G. I. Selective oxidation of methane to methanol on a FeZSM-5 surface. Catal. Today 24, 251-252 (1995).
6. Starokon, E. V. et al. Oxidation of methane to methanol on the surface of FeZSM-5 zeolite. J. Catal. 300, 47-54 (2013).
7. Sushkevich, V. L., Palagin, D., Ranocchiari, M. & van Bokhoven, J. A. Selective Anaerobic Oxidation of Methane Enables Direct Synthesis of Methanol. Science 356, 523-527 (2017).
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in