Modeling study of the effect of placebo and medical therapy on storage and voiding symptoms, nocturia, and quality of life in men with prostate enlargement at risk for progression
Published in Biomedical Research
Ageing men frequently report lower urinary tract symptoms (LUTS) as a result of benign growth of the prostate gland, known as benign prostatic enlargement (BPE). While LUTS/BPE is rarely life-threatening, associated symptoms such as nocturia have been linked to an increased risk of death [Bliwise et al 2019; Moon et al 2022], and are a major reason for physician consultations [Oelke et al 2014]. The impact of the condition on quality of life is also substantial. LUTS/BPE is usually a progressive condition, and left untreated can lead to worsening symptoms and more serious complications such as acute urinary retention (AUR) and BPE-related surgery.
Treatment approaches to LUTS/BPE should ideally target the underlying disease mechanism and improve different symptom types, as well as reducing the risk of AUR/surgery. Hundreds (if not thousands) of clinical trials have been conducted in men with LUTS/BPE; however since results from trials are typically presented as average values for a carefully defined population, it is often difficult to understand how available evidence best informs personalized treatment decisions. Application of predictive analytics to large clinical datasets can support physicians in understanding the key characteristics and relationships behind responses to individual treatments. This knowledge can help to tailor the treatment approach in patients with LUTS/BPE at different levels of progression risk. Visualizing individual risks and how they may change in the future depending on treatment choice (active or placebo), may support healthcare professionals in making more personalized, data-driven decisions.
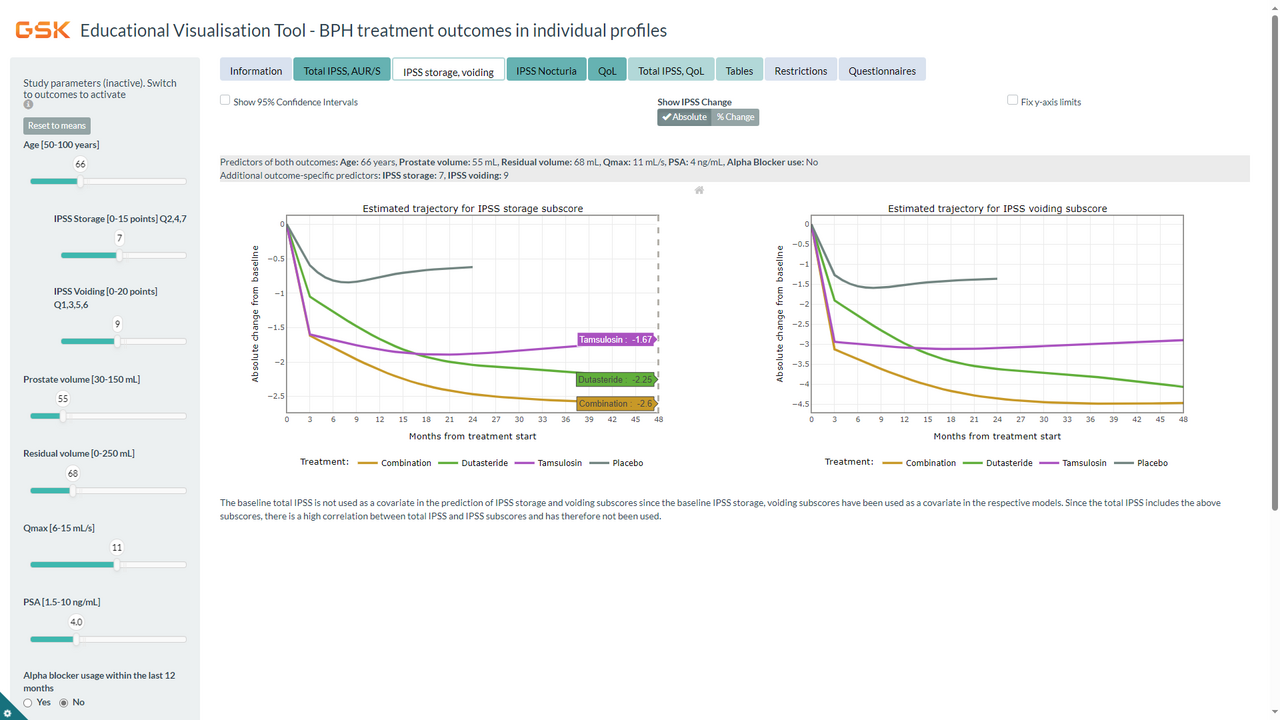
We have previously developed a predictive analytics solution, using large datasets of patients receiving placebo, dutasteride, tamsulosin, or dutasteride/tamsulosin combination therapy, to project the change from baseline in LUTS (via the International Prostate Symptom Score [IPSS]) and risk of AUR or surgery under these different treatment regimens, according to baseline characteristics that commonly define patients at risk of disease progression (e.g., age ≥50 years, moderate-to-severe LUTS, PV ≥30 mL, and PSA ≥1.5 ng/mL) [Gravas et al 2023]. An educational, interactive web-based tool was developed to facilitate visualization and understanding of predicted outcomes for any individual profile meeting the CombAT study entry criteria [Roehborn et al 2010], integrating any possible combination of variables (predictors). The tool is available at www.bphtool.com.
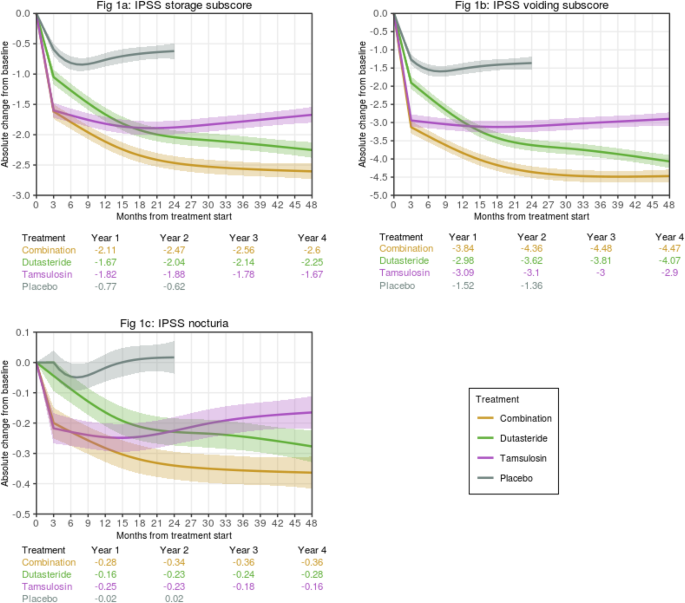
In a follow-up study, we expanded this predictive analytics solution to better understand how placebo, dutasteride, tamsulosin, or combination therapy impact different symptom types (IPSS subscores for storage, voiding, and nocturia [IPSS Q7]) over time, as well as quality of life (IPSS Q8) and BPH Impact Index (BII), in different profiles of men with BPE at risk of progression as defined by their baseline characteristics. The web-based tool was updated to facilitate visualization and understanding of study results. Our findings show that the predicted benefit over time of treatment with dutasteride/tamsulosin combination therapy is greater than with either monotherapy for most patient risk profiles. Our results reinforce the benefit of a treatment approach that targets the underlying disease mechanism in the short and long term in men with LUTS/BPE at risk of disease progression. They also provide insight on the impact of the studied treatments on different types of symptoms and QoL measures for individual patient profiles, which is typically not available from clinical trials. In addition, the results allow greater understanding of the contribution of individual baseline characteristics that commonly define risk of LUTS/BPE disease progression.
This study entitled “Modelling study of the effect of placebo and medical therapy on storage and voiding symptoms, nocturia, and quality of life in men with prostate enlargement at risk for progression”, has been published in Prostate Cancer and Prostatic Diseases (https://doi.org/10.1038/s41391-023-00731-w).
References
Bliwise DL, Howard LE, Moreira DM, Andriole GL, Hopp ML, Freedland SJ. Nocturia and associated mortality: observational data from the REDUCE trial. Prostate Cancer Prostatic Dis. 2019;22:77–83.
Gravas S, Palacios-Moreno JM, Thompson D, Concas F, Kamola PJ, Roehrborn CG, et al. Understanding treatment response in individual profiles of men with prostatic enlargement at risk of progression. Eur Urol Focus. 2023;9:178–87.
Moon S, Kim YJ, Chung HS, Yu JM, Park II, Park SG, et al. The relationship between nocturia and mortality: Data from the National Health and Nutrition Examination Survey. Int Neurourol J. 2022;26:144–52.
Oelke M, Wiese B, Berges R. Nocturia and its impact on health-related quality of life and health care seeking behaviour in German community-dwelling men aged 50 years or older. World J Urol. 2014;32:1155–62.
Roehrborn CG, Siami P, Barkin J, Damiao R, Major-Walker K, Nandy I, et al. The effects of combination therapy with dutasteride and tamsulosin on clinical outcomes in men with symptomatic benign prostatic hyperplasia: 4-year results from the CombAT study. Eur Urol. 2010;57:123–31.
Follow the Topic
-
Prostate Cancer and Prostatic Diseases
This journal covers all aspects of prostatic diseases, in particular prostate cancer, the subject of intensive basic and clinical research world-wide.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in