Molybdenum derived from nanomaterials incorporates into molybdenum enzymes and affects their activities in vivo
Published in Electrical & Electronic Engineering
We are in a heyday of rapid development of nanobiotechnology. The field has covered many aspects, including the nanomedicine, nanotoxicology, biosensor, tissue engineering, drug delivery, medical imaging, etc. Nowadays, nanobiotechnology has become the forefront and hot research area in the world, with a wide range of applications and promising industrialization prospects. In this area, in vivo biological behavior of nanomaterials is a key indicator of biosafety evaluation, determining the transformation of nanomedicine to clinical applications and guiding the design of bionanomaterials.
Nanomaterials will encounter complex biological barriers and physiological structures once entering the biosystem. Interaction of nanomaterials with various organs, cells, biological milieu and other complex nano-bio interfaces is the decisive step that determines the delivery to the target site. For a long time, Prof. Chunying Chen’s lab at National Center for Nanoscience and Technology in China has devoted to developing methods analyzing the nano-bio interactions1-6. The research group has collaborated with National Synchrotron Radiation Facility for many years and developed advanced techniques before I became a PhD student in the lab. With the synchrotron radiation-based techniques, our group quantitatively analyzed the interaction of nanoparticles with macromolecules (proteins, phospholipids) at the nanoparticle-biological interface7,8 and qualitatively characterized the chemical behavior of nanomaterials in organisms9-11.
Except for the interfacial interactions of nanoparticles with biological barriers, we became curious about the in vivo journey of nanoparticles. In vivo transport process from the administration to elimination is still a “black box”. As demonstrated by many studies, mononuclear phagocyte system (MPS) is the main site for nanomaterials accumulation. How do nanoparticles ‘travel’ from the blood to MPS? What role does the protein corona play in the journey? What fate do they go through after the interception in MPS? I set out with the questions and realized that more systematic strategy is needed to clarify the whole journey inside the body. The lack of multiscale, highly sensitive and in-situ methods analyzing the metabolism of nanomaterials at the tissue, single-cell or even single-molecule level is the key issue.
The whole journey was revealed step by step via studying the nano-blood, nano-protein and nano-target organs interactions. We selected MoS2 nanomaterials as the material model because of two reasons: 1) MoS2 nanomaterial is a star material among the transition metal dichalcogenides nanomaterials, as promising nano-platforms for the drug delivery, cancer theranostics, bioimaging, and biosensing.; 2) The essential trace element molybdenum, a distinguishing feature of MoS2 nanomaterials, may bring surprising findings, we suspected so.
The experiment process didn’t go smoothly, as most researchers encountered. It took long time to deal with many difficulties at the pre-experimental stage. The research strategy to understand the in vivo journal and the role of protein corona was finally proposed after exploring for a long time. We integrated multi-disciplinary state-of-the-art techniques, i.e., intravital confocal fluorescence microscopy, in-situ imaging at tissue and cell levels, metabolic analysis methods, proteomics, and molecular dynamics simulations, to study the in vivo transport, chemical transformation and metabolism of MoS2 nanomaterials.
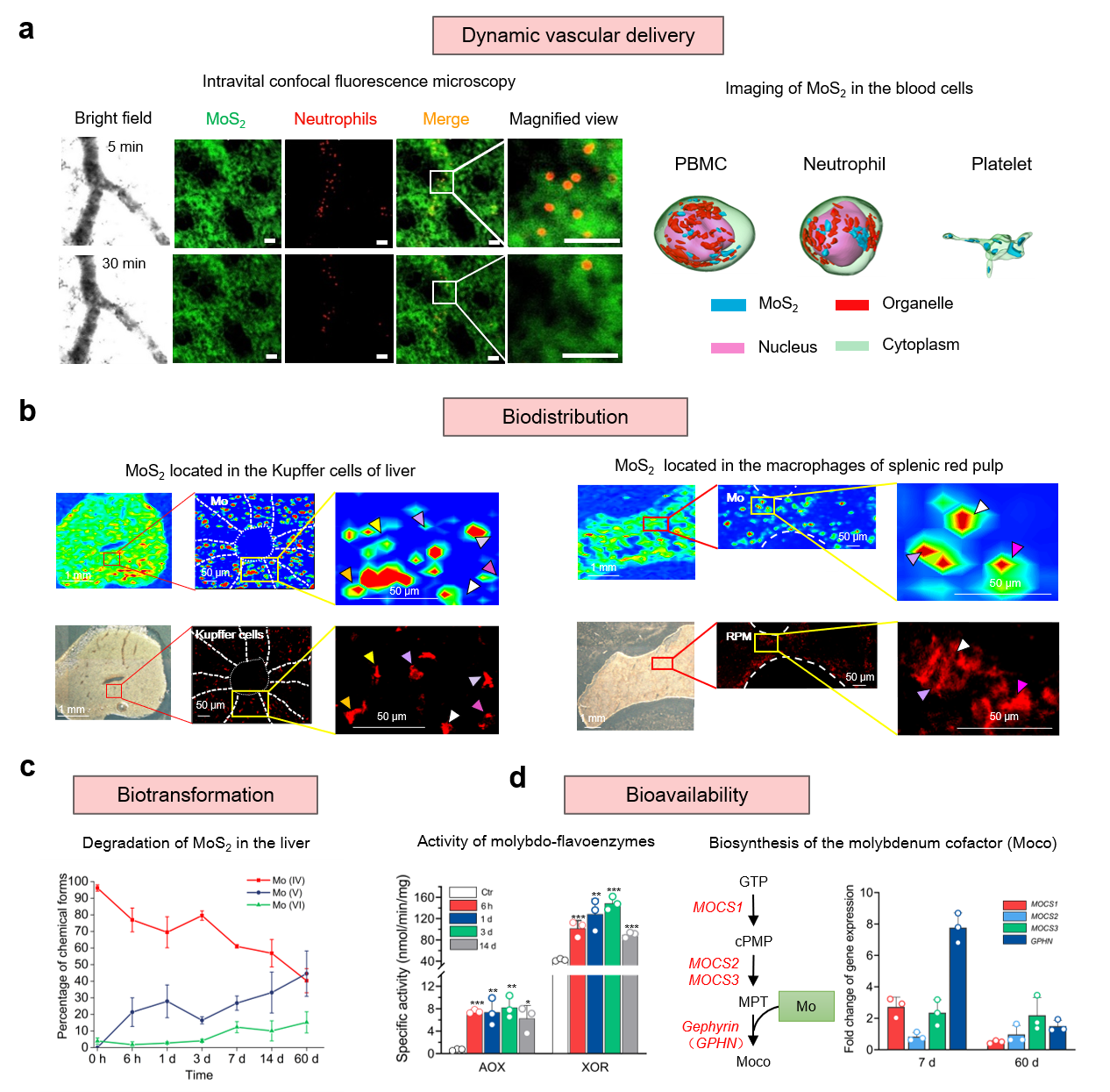
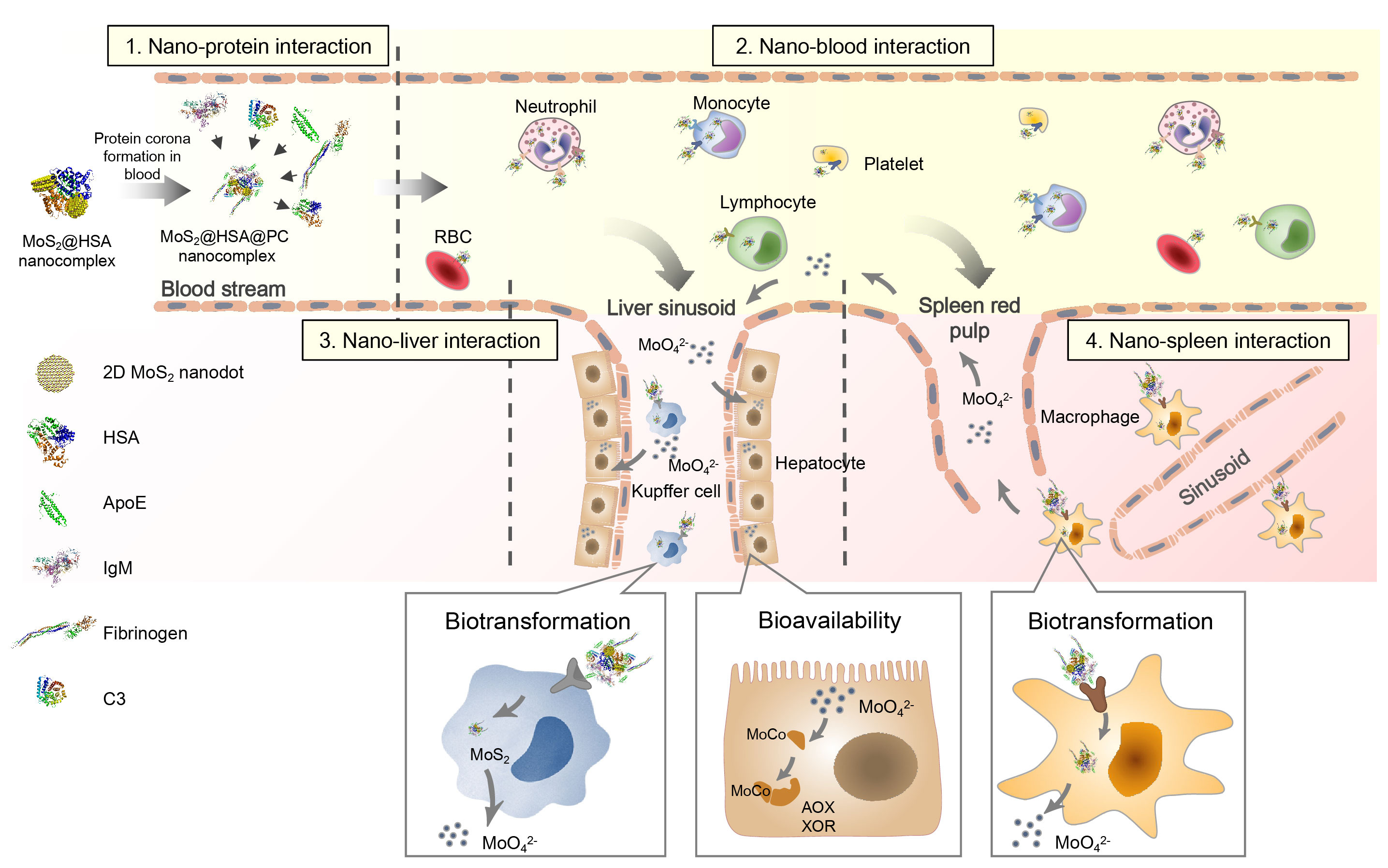
Figure. The in vivo journey of MoS2 nanomaterials, including the vascular delivery, biodistribution, biotransformation and bioavailability
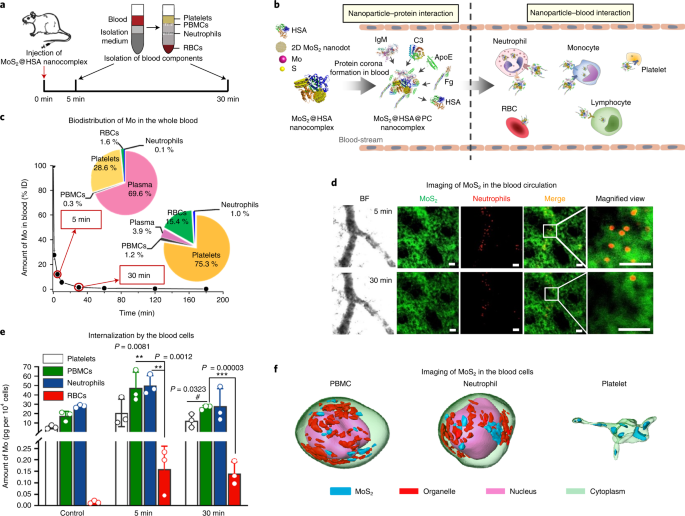
The in vivo journey of MoS2 started from the vascular delivery after intravenous administration. The real-time images and the quantification data told us most of the MoS2 presented in the plasma and the rest were internalized by blood cells. Synchrotron radiation-based soft X-ray nanocomputed tomography (Nano-CT) suggested the high internalization of MoS2 in peripheral blood mononuclear cell (PBMC), neutrophil and platelet with high spatial resolution of 30 nm at single-cell level. The analysis of protein corona and the interaction mechanism were determined with proteomics and molecular dynamics simulations.
Biodistribution and translocation happened after the transport in blood. Next stops of MoS2, as demonstrated by synchrotron radiation X-ray fluorescence imaging of Mo and fluorescence images of macrophages, were hepatic Kupffer cells and splenic red pulp macrophages (RPMs). By developing the transgenic mice models deficient of apolipoprotein E (ApoE), fibrinogen (Fg), complement 3 (C3), and immunoglobulin M (IgM) proteins, we revealed that ApoE was mainly responsible for the sequestration of MoS2 in Kupffer cells and RPMs.
Then MoS2 underwent degradation after the interception in liver and spleen. MoS2 nanomaterials have been demonstrated to be sensitive to the environmental factors, such as O2, H2O2. The in vivo biotransformation was manifested for the first time in our work. The oxidation of Mo (IV) to Mo (VI) in liver and spleen was determined with synchrotron radiation X-ray absorption near-edge spectroscopy. We also found that the oxidation originated from liver microsome and intracellular reactive oxygen species.
As suspected in the beginning, the essential trace element molybdenum in MoS2 nanomaterials brought unique biological effects. We verified the bioavailability of molybdenum derived from MoS2 by the increased activities of hepatic molybdenum enzymes. As clarified, the mechanism was that oxidized molybdenum incorporated into molybdenum enzymes as molybdenum cofactors and increased the enzyme activities. Up to this step, we have revealed the in vivo journey of MoS2 nanomaterials, including vascular delivery, biodistribution, transformation and bioavailability.
We developed systematic methods to clarify the whole fate of nanomaterials from administration to metabolism. We demonstrated the different metabolic fate in which the nanomaterials could be converted into active biological molecules eventually used by the organism. Our finding offers new thinking about the biokinetics of nanomaterials with similar properties to MoS2 bearing essential trace elements. Nanomaterial-based therapies or diagnostics need to pay more attention to the bioavailability in vivo and the underlying impacts on the biological functions.
Of course, there is still much to be done in the future. Our research journey is still on the way.
Click here to read more about our work in Nature Nanotechnology.
References:
1 Chen, C., Li, Y.-F., Qu, Y., Chai, Z. & Zhao, Y. Advanced nuclear analytical and related techniques for the growing challenges in nanotoxicology. Chem. Soc. Rev. 42, 8266-8303 (2013).
2 Wang, L., Yan, L., Liu, J., Chen, C. & Zhao, Y. Quantification of nanomaterial/nanomedicine trafficking in vivo. Anal. Chem. 90, 589-614 (2018).
3 Wang, L. et al. Using hollow carbon nanospheres as a light-induced free radical generator to overcome chemotherapy resistance. J. Am. Chem. Soc. 137, 1947-1955 (2015).
4 Li, J. et al. New power of self-assembling carbonic anhydrase inhibitor: Short peptide–constructed nanofibers inspire hypoxic cancer therapy. Sci. Adv. 5, eaax0937 (2019).
5 Zhou, H. et al. Hypoxia-triggered self-assembly of ultrasmall iron oxide nanoparticles to amplify the imaging signal of a tumor. J. Am. Chem. Soc. 143, 1846-1853 (2021).
6 Lu, X. et al. Long-term pulmonary exposure to multi-walled carbon nanotubes promotes breast cancer metastatic cascades. Nat. Nanotechnol. 14, 719-727 (2019).
7 Wang, L. et al. Revealing the binding structure of the protein corona on gold nanorods using synchrotron radiation-based techniques: Understanding the reduced damage in cell membranes. J. Am. Chem. Soc. 135, 17359-17368 (2013).
8 Wang, L. et al. Stability of ligands on nanoparticles regulating the integrity of biological membranes at the nano–lipid interface. ACS Nano 13, 8680-8693 (2019).
9 Liu, J. et al. Graphdiyne-templated palladium-nanoparticle assembly as a robust oxygen generator to attenuate tumor hypoxia. Nano Today 34, 100907 (2020).
10 Liu, J. et al. Rapid degradation and high renal clearance of Cu3BiS3 nanodots for efficient cancer diagnosis and photothermal therapy in vivo. ACS Nano 10, 4587-4598 (2016).
11 Wang, L. et al. Use of synchrotron radiation-analytical techniques to reveal chemical origin of silver-nanoparticle cytotoxicity. ACS Nano 9, 6532-6547 (2015).
Follow the Topic
-
Nature Nanotechnology
An interdisciplinary journal that publishes papers of the highest quality and significance in all areas of nanoscience and nanotechnology.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in