mRNA-Encoded "Mini-Antibodies": A Next-Generation Shield Against Multidrug-Resistant Bacteria
Published in Biomedical Research

1. Background
Multidrug-resistant (MDR) bacteria pose a growing global threat and are a critical public health concern. If current trends continue, mortality from drug-resistant infections is projected to surpass cancer-related deaths by 2050 [The Review on Antimicrobial Resistance
Chaired by Jim O’Neill, J. , 2014]. Notably, the World Health Organization (WHO) has categorized Pseudomonas aeruginosa as a priority pathogen requiring the highest level of vigilance [Sati, H. et al., 2025]. Primarily affecting immunocompromised patients, it often leads to fatal pneumonia or urinary tract infections. Given the growing resistance to conventional antibiotics, there is an urgent need for novel therapeutic strategies.
In response, our research team developed Antibody Therapy, a therapeutic designed to block the bacterial "syringe" mechanism that injects toxins into host cells. However, the traditional production of this agent is both time-consuming and costly. To overcome these barriers, we utilized antibody messenger RNA technology—the same platform used for COVID-19 vaccines. By delivering the therapeutic "blueprint" directly into the body, we enable the patient's own cells to produce the treatment.
This approach significantly reduces production time and costs. In our study, administering this mRNA-based therapy to mice infected with highly drug-resistant bacteria resulted in a dramatic improvement in survival. These findings demonstrate not only the potential for rapid, cost-effective production but also the promise of this platform to address the urgent need for new treatments in clinical settings where multidrug-resistant infections are prevalent and current options are limited.
2. Research Content and Key Findings
2.1 Overview of the Antibody mRNA Therapy
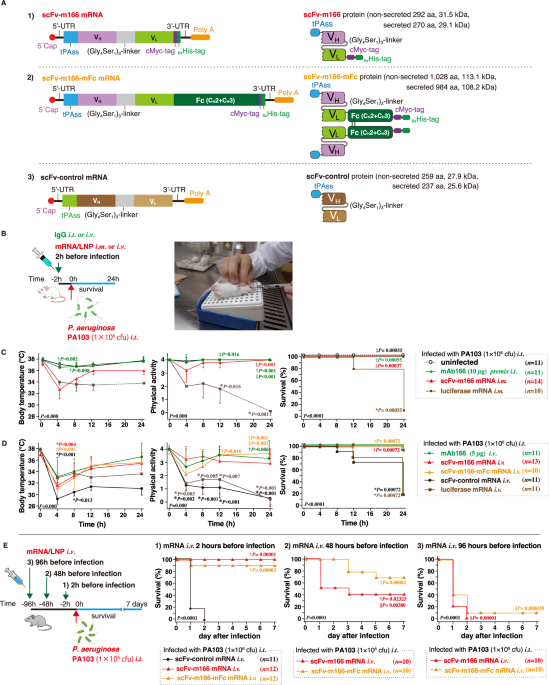
Figure 1 illustrates our therapeutic strategy. Conventional antibodies consist of a target-binding region and a structural scaffold (the Fc region). In this study, we engineered a compact single-chain antibody (scFv) comprising only the target-binding domain. This "miniaturization" was designed to enhance penetration into the distal lung regions most affected by infection.
While direct administration of these small antibody fragments results in rapid systemic clearance—often within minutes—delivering the mRNA blueprint enables the body to maintain endogenous production for several days, ensuring a sustained therapeutic effect. In practice, the mRNA is encapsulated in lipid nanoparticles (LNPs) for delivery. Upon administration, these particles localize primarily to the liver, which serves as a biomanufacturing factory. The secreted antibodies then enter the bloodstream and migrate to the lungs, where they neutralize P. aeruginosa and protect the respiratory epithelium from bacterial attack.
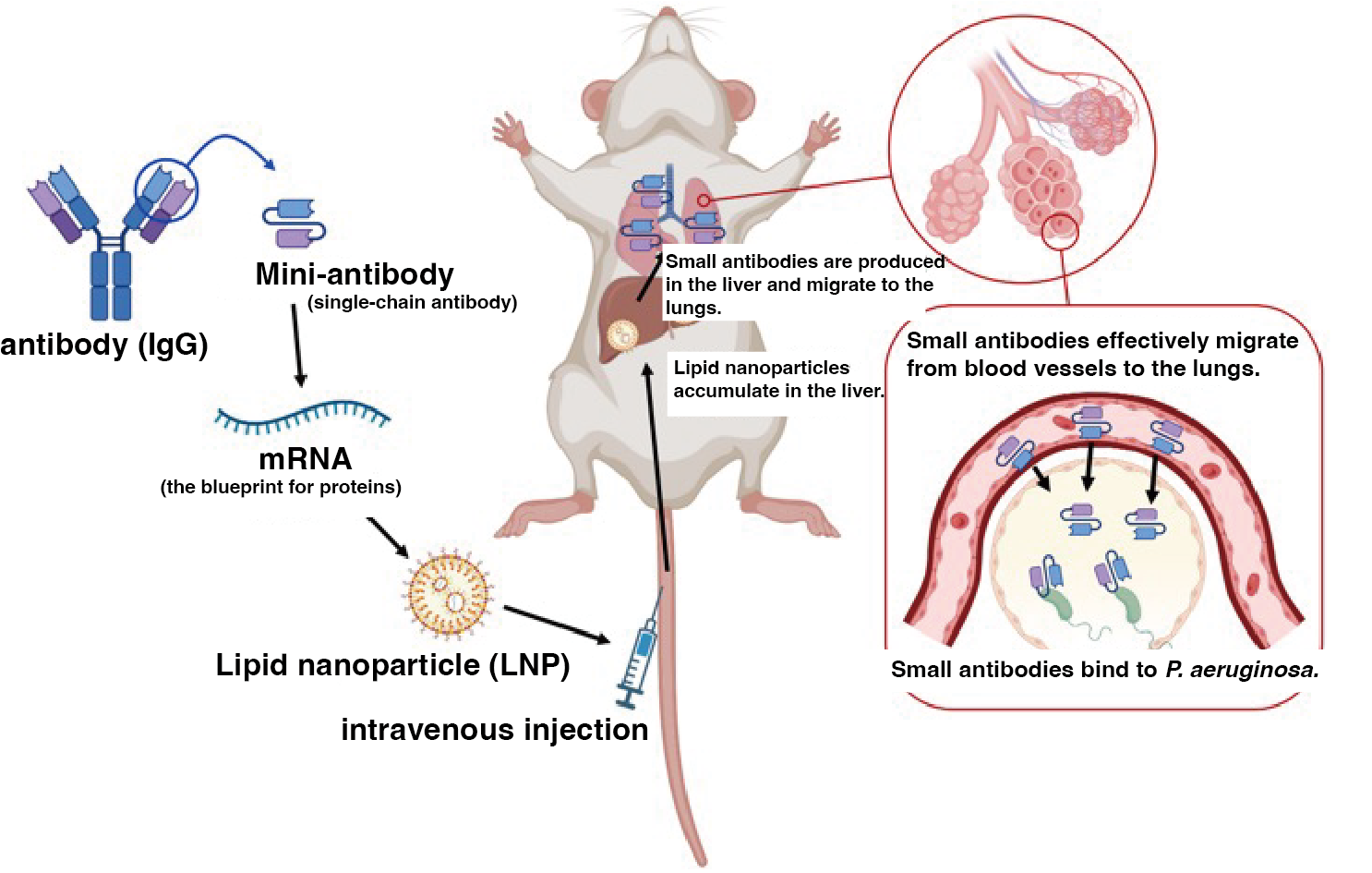
2.2 Elucidating How the Antibody Protects Cells from P. aeruginosa Toxins
2.2.1 The Type III Secretion System of pathogenic Gram-negative bacteria
2.2.2 P. aeruginosa: The Molecular Syringe and New Treatment
2.2.3 PcrV of the P. aeruginosa T3SS
As illustrated in Figure 2A, P. aeruginosa utilizes a needle-like T3SS to inject cytotoxic effector proteins directly into lung cells. Our therapeutic antibody targets PcrV, a scaffold protein located at the distal tip of this needle apparatus. By binding to PcrV, the antibody "caps" the secretion pore, thereby neutralizing toxin delivery, reducing tissue damage, and limiting bacterial proliferation.
Structurally, PcrV assembles into a homopentamer at the tip of the needle complex, forming a central channel that regulates the translocation of toxins into host cells (Figure 2A).
It was previously hypothesized that a single antibody molecule binds to each PcrV pentamer (Figure 2B), thereby occluding the central pore and inhibiting injection.
However, our molecular simulations and experimental data reveal a different mechanism: two antibody molecules can simultaneously bind to a single PcrV pentamer (Figure 2C). This dual binding likely provides more robust inhibition. High-resolution structural predictions using AlphaFold3 (Figure 2D) further suggest that the antibody interface adopts a "lock-and-key" fit, effectively covering the central pore and providing greater steric hindrance to toxin translocation.
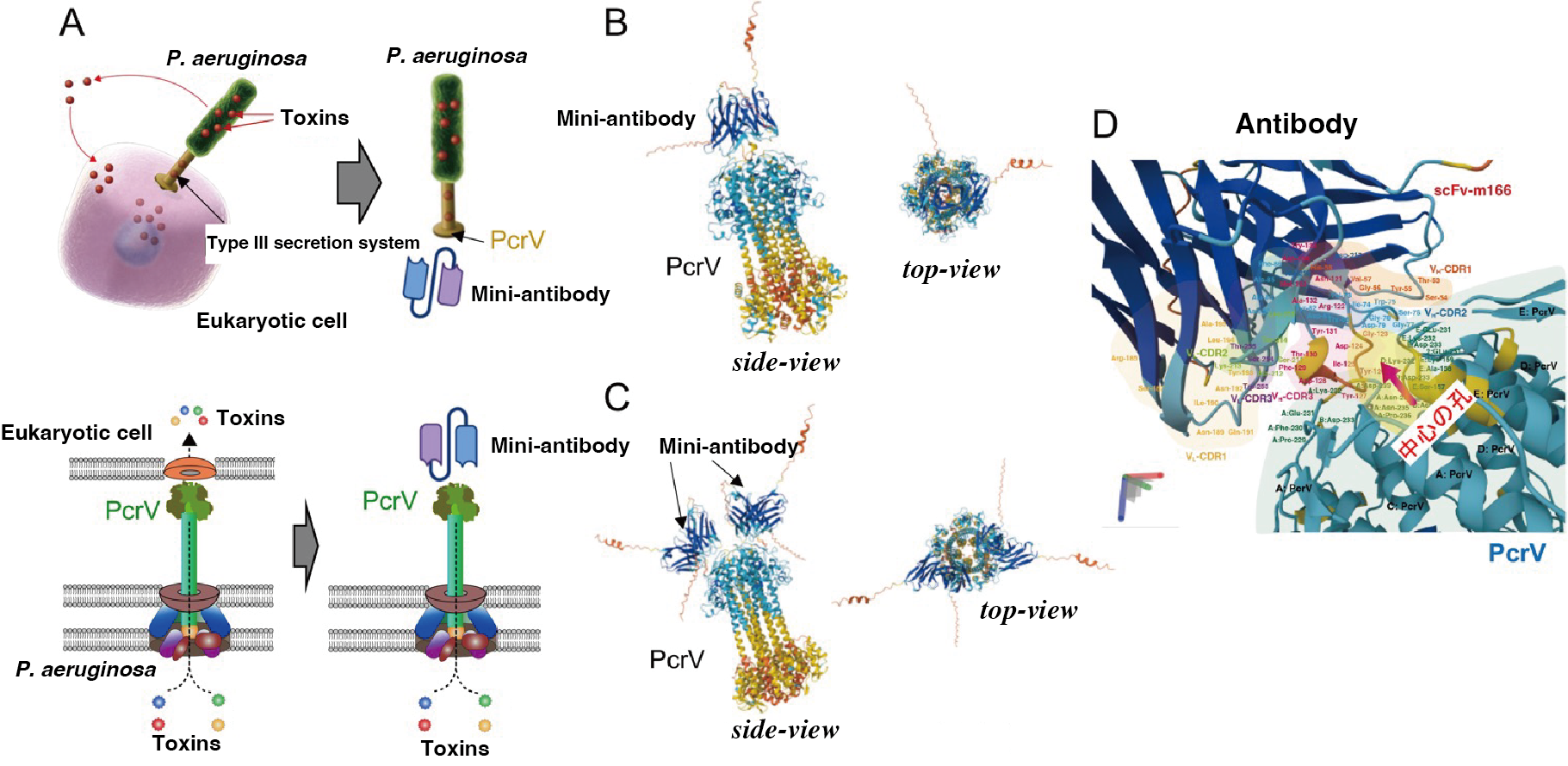
Figure 2: Antibody mechanism of action. (A) Schematic overview: P. aeruginosa injects toxins into host cells via the Type III Secretion System (T3SS). The antibody binds to PcrV at the tip of the T3SS, blocking toxin injection. (B) A model in which one antibody molecule binds to PcrV. (C) A model in which two antibody molecules bind to PcrV. (D) High-accuracy structural prediction using AlphaFold3.
2.3 Successful Treatment of P. aeruginosa Pneumonia in Mice
Next, we evaluated the efficacy of an mRNA platform for the in vivo production of our engineered antibody fragment. We first established an immunocompetent mouse model by intratracheal inoculation with P. aeruginosa, followed by mRNA administration 30 minutes post-infection (Figure 3A). While all untreated control mice succumbed to the infection within 24 hours, 80% of the mRNA-treated group survived for at least one week, demonstrating potent therapeutic efficacy (Figure 3B). These results suggest that this platform could offer a novel and effective therapeutic option for patients with severe, drug-resistant P. aeruginosa infections, particularly those at high risk in clinical settings, such as immunocompromised individuals.
To simulate high-risk clinical scenarios, such as multidrug-resistant (MDR) infections in immunocompromised patients, we challenged immunodeficient mice with a hypervirulent, MDR clinical isolate. While untreated mice died within three days, nearly 50% of the mRNA-treated mice survived through the one-week observation period (Figure 3C). Notably, our mRNA therapy outperformed colistin—a last-resort antibiotic—which yielded a survival rate of less than 20%. Furthermore, combining mRNA therapy with conventional antibiotics achieved 100% survival, indicating a strong synergistic effect and highlighting its potential as a complementary adjunct to current standards of care.
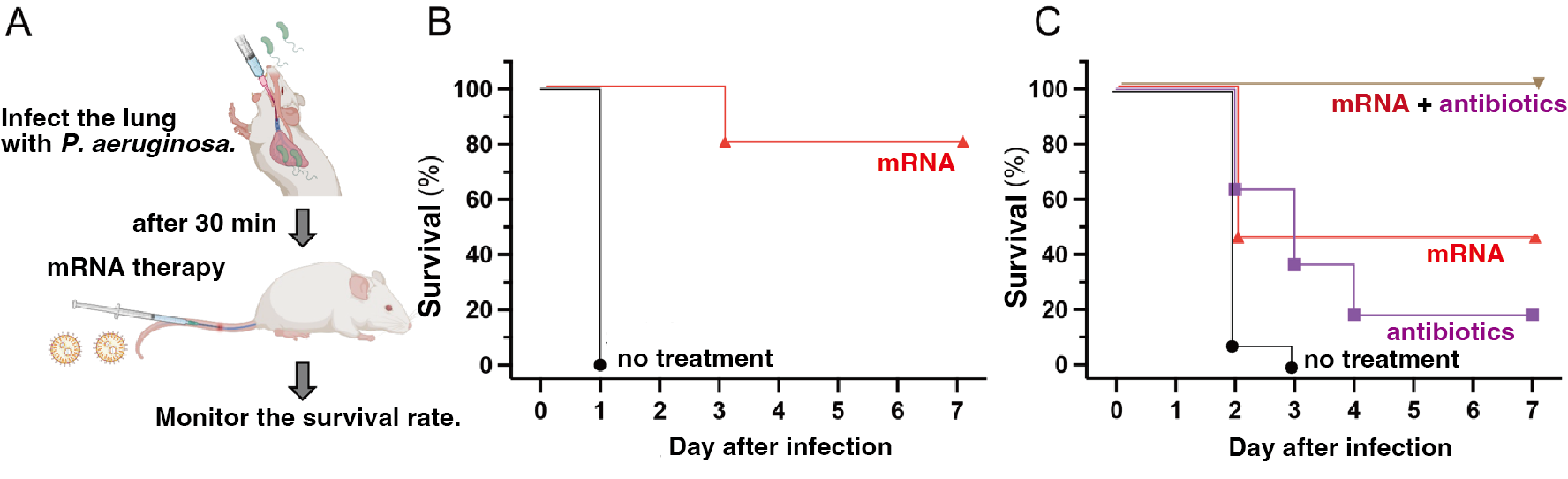
Figure 3: Mouse therapeutic experiments. (A) Study design overview. (B) Survival in an immunocompetent mouse infection model. (C) Survival in immunodeficient mice infected with a multidrug-resistant, hypervirulent P. aeruginosa strain.
Mechanistic analysis revealed that mRNA treatment significantly reduces pulmonary bacterial burden (Figure 4A) and alleviates severe pneumonia (Figure 4B). Histological examination showed widespread inflammatory cell infiltration in untreated lungs, whereas mRNA-treated lungs exhibited minimal inflammation.
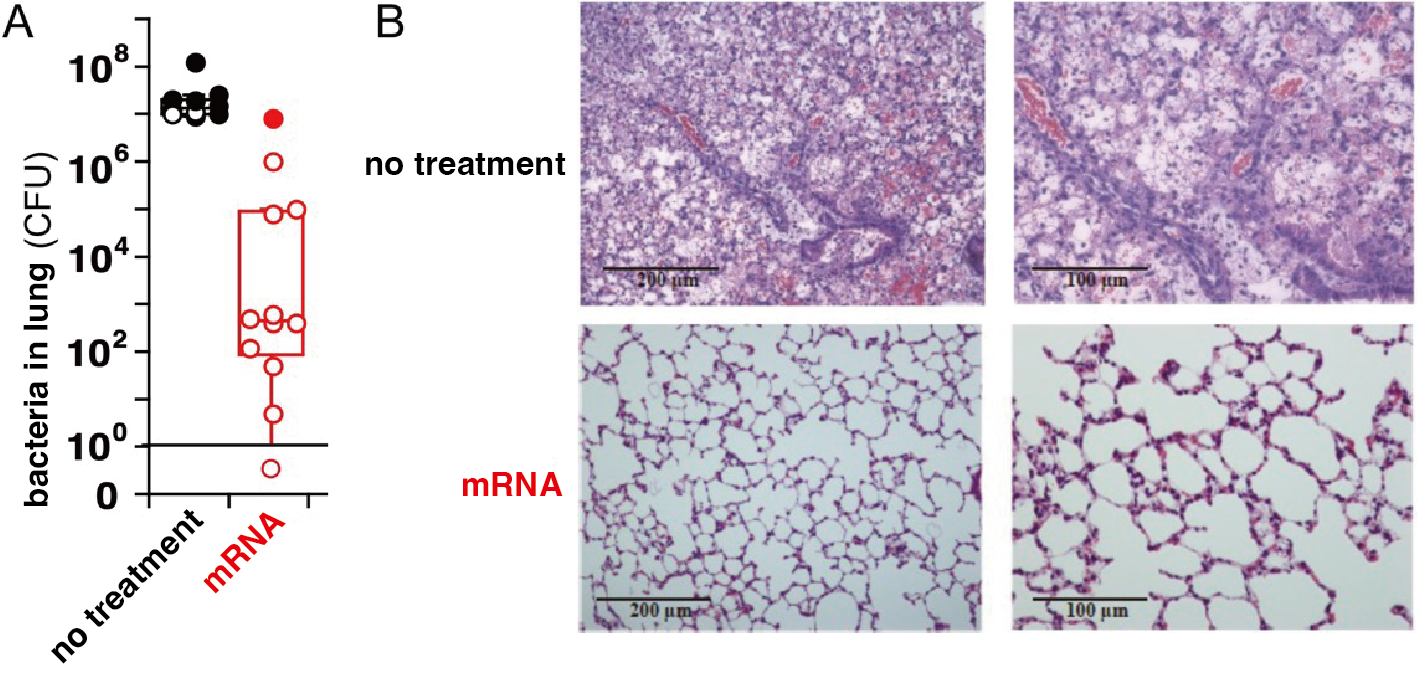
Figure 4: Mechanism of antibody therapy.(A) Pulmonary P. aeruginosa burden. (B) Representative lungs after infection.
Finally, we assessed the impact of antibody miniaturization. We compared our small antibody fragment to a conventional full-sized antibody. Across multiple models, the smaller format consistently achieved higher survival rates (Figure 5A and 5B). This superior performance is attributed to enhanced delivery to the lung epithelium, the primary site of infection, where the smaller molecule demonstrated significantly greater accumulation than the conventional format (Figure 5C).
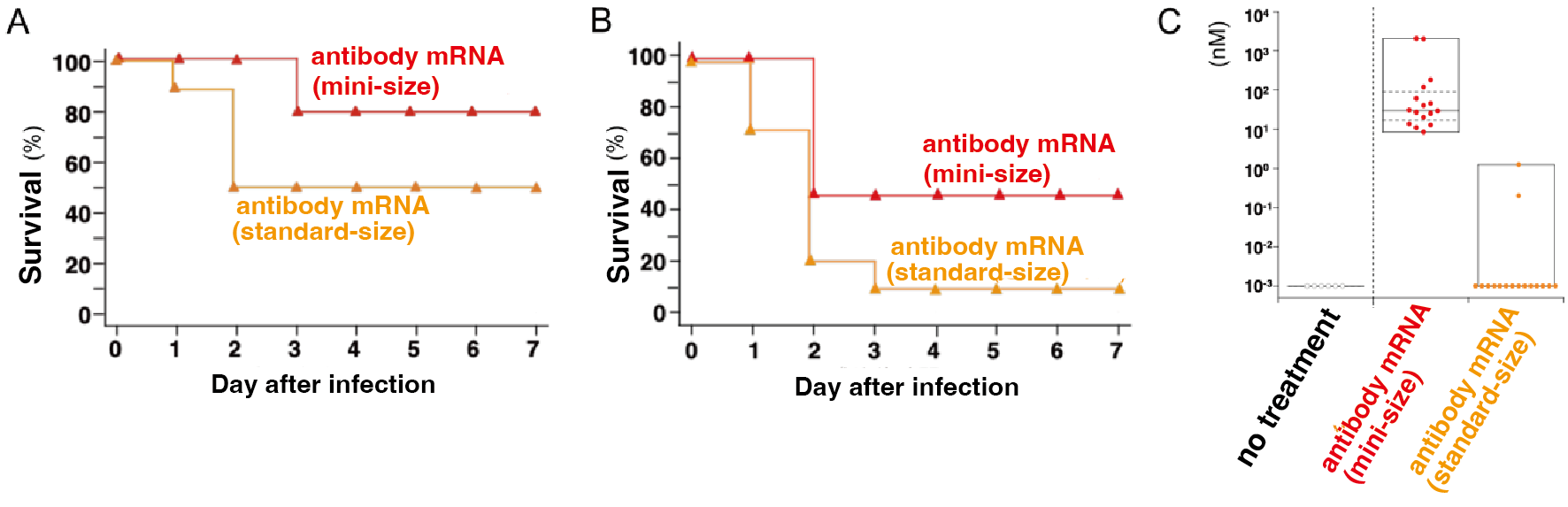
Figure 5: Effects of antibody miniaturization. (A) Survival in an infection model using immunocompetent mice. (B) Survival in immunodeficient mice infected with a multidrug-resistant, hypervirulent P. aeruginosa strain. (C) Antibody levels on the epithelial side of the lung, the primary site of infection.
2.4 What Makes This Study Groundbreaking
▪ Antibody mRNA Therapy Against Multidrug-Resistant Bacteria
While monoclonal antibodies (mAbs) are established staples in oncology and immunology, their application in infectious diseases has been constrained by high manufacturing costs and logistical barriers. This study demonstrates the transformative potential of mRNA-encoded antibody therapy. By significantly reducing production costs and simplifying delivery, mRNA platforms could democratize access to antibody-based anti-infectives, providing a critical countermeasure against the escalating global threat of multidrug-resistant (MDR) bacteria.
▪ Advantages of Antibody Miniaturization
To date, most mRNA-based antibody studies have utilized conventional, full-length immunoglobulin formats. This study is the first to establish the physiological advantages of antibody miniaturization. Although smaller antibody fragments often undergo rapid systemic clearance, our approach leverages mRNA to enable sustained in vivo production. This effectively bypasses the pharmacokinetic hurdles of protein-based delivery, making the therapeutic use of high-penetrance mini-antibodies clinically viable for the first time.
▪ Validation in Clinically Relevant Models
MDR P. aeruginosa infections in immunocompromised patients represent a significant clinical challenge with high mortality rates. By achieving potent therapeutic efficacy in a model that recapitulates these exact clinical conditions, our results provide a robust foundation for translational development. These findings strongly support the transition from preclinical proof-of-concept to human clinical trials.
▪ Structural Insights into Molecular Mechanisms
Our high-resolution structural modeling using AlphaFold3 suggests that the antibody binds to the PcrV protein, effectively occluding the pore of the Type III Secretion System (T3SS) needle. By physically blocking the pathogen's ability to inject toxins, the antibody disarms the pathogen at the molecular level. This mechanistic framework serves as a blueprint for developing similar "cap-and-neutralize" strategies against a wide array of other needle-bearing bacterial pathogens.
3. Future Outlook and Key Messages
Addressing the global crisis of multidrug-resistant (MDR) bacteria, this study establishes a next-generation mRNA-powered antibody therapy platform. By directly neutralizing the bacterial toxin-injection apparatus, this strategy offers a robust countermeasure to the projected surge in MDR-related mortality by 2050.
A key innovation of this work is the use of mRNA to overcome the pharmacokinetic limitations of miniaturized antibodies. While such fragments are typically cleared too quickly to be effective, our mRNA-based delivery ensures sustained in vivo production, achieving a therapeutic window previously unattainable for small-molecule biologics. Furthermore, this platform promises to significantly reduce manufacturing costs and stabilize global supply chains compared to conventional protein-based drugs. The proven success and scalability of mRNA vaccines during the COVID-19 pandemic provide a strong regulatory and logistical foundation for the clinical adoption of this technology.
Beyond P. aeruginosa, this "cap-and-neutralize" approach is broadly applicable to other major pathogens utilizing similar secretion systems, such as Salmonella and Shigella. Our results in clinically relevant models serve as a powerful catalyst for human trials. Ultimately, we envision this technology as "next-generation medical infrastructure"—a digital-to-biological bridge that can rapidly deliver life-saving therapeutics to the most vulnerable patients, thereby democratizing access to advanced biologics and driving the digital transformation of global healthcare.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Healthy Aging
Publishing Model: Open Access
Deadline: Dec 31, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in