New insights into androgen deprivation therapy
Published in Cancer
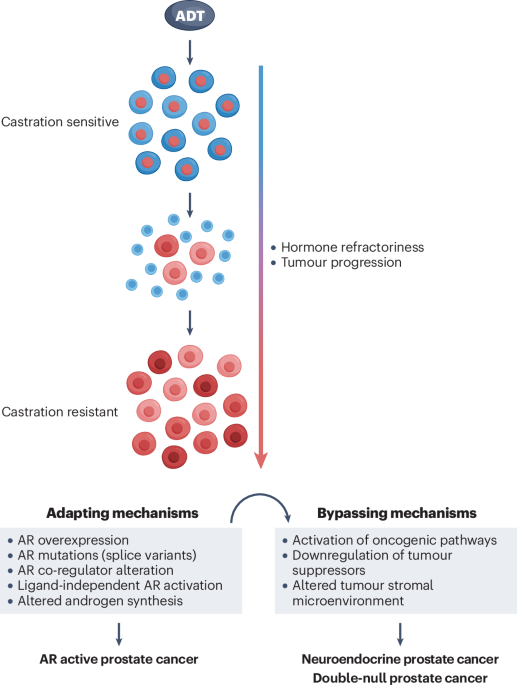
Currently, androgen deprivation therapy (ADT) that directly targets androgen receptor (AR)/androgen-mediated signaling pathways is still the first-line treatment for advanced prostate cancer. However, ADT eventually fails in almost all patients who consequently develop castration-resistant prostate cancer (CRPC). More potent AR antagonists and inhibitors for blocking androgen synthesis have been developed recently to inhibit tumor progression and CRPC development. While these second-generation antagonists/inhibitors showed some clinical responses, recent clinical data also implied that they could induce more diverse CRPC phenotypes and promote tumor progression. Specifically, a new type of CRPC, named double-null prostate cancer (DNPC), has been identified in patients treated with abiraterone and enzalutamide, featuring AR- and neuroendocrine (NE)-null prostate tumor cells, which directly contributes to the mortality of prostate cancer. Therefore, there is an urgent need to define the mechanisms underlying the ADT-induced DNPC in order to develop new therapeutic strategies to improve clinical outcomes.
In this review, we summarized the latest discoveries of the inducible role of current ADT in promoting prostate cancer progression and DNPC development. Analysis of DNPC patients’ samples showed aberrant activation of HGF/MET and Wnt/β-catenin signaling pathways. Newly developed mouse models further affirmed that activating HGF/MET and Wnt/β-catenin signaling pathways promotes tumor aggressiveness, hormonal refractoriness, and the development of prostate tumor lesions similar to human DNPC. Increased expression and activity of nuclear exporting and ribosomal biogenesis were also identified in both human and mouse DNPC samples. The review included new and emerging scientific results in combination with current and previous clinical and experimental evidence, and provided the mechanistic prospects for one of the most critical challenges in the current field of prostate cancer research. It not only discussed the new therapeutic targets and strategies for treating advanced prostate cancer but also elucidated the fresh insight into future clinical interventions for preventing lethal DNPC development.
Follow the Topic
-
Nature Reviews Urology
Nature Reviews' basic, translational and clinical content is written by internationally renowned basic and clinical academics and researchers and targeted towards readers in the biological and medical sciences, from postgraduate level upwards.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
Besides the HGF/MET and Wnt/β-catenin pathways, what other pathways are involved in ADT-induced DNPC? How are they related to each other?