Phage therapy is back, it’s promising and it’s going to take structural support to keep it going.
Published in Microbiology, Biomedical Research, and General & Internal Medicine
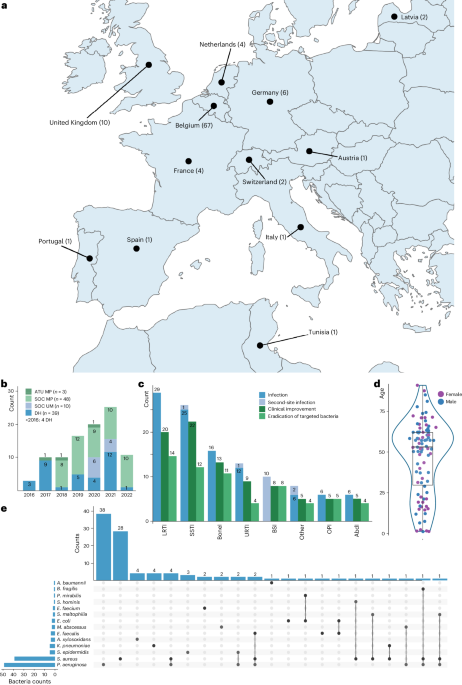
This monumental effort of providing phage therapy for 100 cases of difficult-to-treat infections was led by the group at the Queen Astrid Military Hospital (QAMH) located in Brussels, Belgium. A Belgian consortium consisting of QAMH, KU Leuven and Sciensano, the public health institute for Belgium, worked together to facilitate these treatments.
This was a collaborative effort, involving eight institutes from six different countries to provide phages for patients. Also, 63 providers from around the world to administer these treatments. For these treatments, 14 bacterial species were targeted using 26 individual phages and six defined phage cocktails. Notably, 13 phages were pre-adapted using a method named “Appelmans protocol,” a titration protocol first developed in the 1920s by René Appelmans, a Belgian physician. This co-evolution approach between phage and bacteria is used to increase the activity of the phage, expand its host range, and reduce the emergence of resistance (1).
In terms of clinical outcomes, a reported 77.2% of targeted infections showed clinical improvement, while eradication of the targeted bacteria was observed in 61.3% of infections with sufficient follow-up data. A statistical analysis indicated that among all categorical variables the presence of concomitant antibiotics was the most predictive factor, with bacterial eradication being more probable with concomitant antibiotic use. Further, many synergistic interactions were identified between various phages and antibiotics in supporting assays suggesting again that phage and antibiotic combination is an optimal strategy. It’s crucial to note that no major adverse effects were reported with these treatment protocols, emphasizing their safety and tolerability.
Moreover, these successful outcomes are the result of an array of clearly outlined strategies for success, that range from phage characterization & selection and phage production to clinical practice and downstream analysis of the patient outcome.

Also pictured is lead author Dr. Jean-Paul Pirnay (1st from left), phage researcher Nino Grdzelishvili (3rd from left), Khadidja Rezig (mother of patient, 2nd from left) and Dr. Patrick Soentjens (far right).
Picture used with permission from the website https://www.thomashausler.com/category/phage-therapy/. Picture was from Sofiane Rezig.
Before treatment, the carefully selected and produced phage products were thoroughly assessed for quality and safety by Sciensano, in accordance with the bacteriophage API monograph (2). Various parameters are checked including potency, bioburden, pH and endotoxin levels. Phage genome sequencing is also performed to ensure phages genomes contained no genetic determinants known to confer lysogeny, toxicity, virulence or antibiotic resistance. Host bacteria were assessed to ensure production strains were as safe as possible.
Each phage was assigned a passport which allows it to be used as an API (Active Pharmaceutical Ingredient) for personalized phage preparations.

Some major concerns that may arise during phage therapy include the occurrence of resistance and antibody neutralization of the phages. These are addressed in this paper using supporting assays.
As for the isolation of bacterial resistors, in vivo selection of phage-insensitive phenotype was observed in seven of 16 patients for which adequate follow-up samples were available. Genome sequencing of bacterial isolates from several patients, performed at KU Leuven, revealed single-nucleotide polymorphism (SNP) mutations within the bacterial receptor genes as the basis of the resistance phenotype (5 cases). In one of these patient cases, a multidrug-resistant Pseudomonas aeruginosa developed resistance to three different phages, each targeting distinct bacterial receptors. Despite the development of resistance, there were accompanying fitness trade-offs observed, including reduced virulence and re-sensitivity to fluoroquinolones. The latter was likely due to an accompanying mutation within a gene encoding a major multidrug efflux pump, a possible receptor for the phage used. This phenomenon confirms the value of a directed strategy for phage therapy: utilizing phages that target these efflux pumps, so that in the event of resistance, the once-resistant bacteria regain sensitivity to antibiotics. Observing this strategy in action during phage therapy in a patient provides valuable insight into its real-world applicability and efficacy in treatments.
Given the potential immunogenicity of phages, a phage neutralization assay was performed on serum samples of 13 screened patients, using the classical neutralization assay by Mark H. Adams (3). Neutralization activity was detected in samples from five patients. This was likely due to the fact that they received invasive phage therapy (e.g. intravenous and/or intralesional). However, this activity did not seem to affect treatment outcomes, as 4 out of 5 patients from this group showed clinical improvement and successful eradication of the targeted bacteria.
Despite this publication being a decade in the making, my involvement at KU Leuven began just two years ago. Previously, I worked in Houston, Texas helping establish a phage center aimed at aiding patients in the U.S. Our goal was similar to the group in Belgium, providing phage therapy to patients with challenging infections. Our efforts were met with significant obstacles primarily due to the stringent regulatory landscape in the U.S. In the U.S., the Food and Drug Administration (FDA) can allow for investigational medical products to be used for patients with severe or immediately life-threatening infections through their expanded-access program. This pathway can be a laborious and time-consuming process which can significantly delay treatment as demonstrated in our 12 case report(4). In contrast, since 2018 Belgium has allowed the delivery of phages through magistral preparations (also referred to as "compounding" preparations) under the direct responsibility of doctors and pharmacists(5). This regulatory flexibility enables personalized phage treatments for patients with difficult-to-treat infections and extends to anyone seeking phage therapy. This paper underscores that with regulatory flexibility, safe and effective treatments can be provided to patients in need. Recently the UK Parliament discussed the use of phages as a way to tackle the antimicrobial resistance problem signaling that other countries are looking into this therapy as well.
This paper also underscores the substantial challenges in manufacturing and coordinating these personalized treatments. Further innovations in manufacturing may be needed to make these treatments more attainable for a broader set of patients at a global scale. In my own country I imagined that phages could be produced by hospitals, and administered by medical personnel within the hospital, as a way of controlling drug-resistant pathogens from spreading and also helping the most vulnerable patients. This paper describes the possibility of the development of a device that could provide instant, on-site production of phages using synthetic biology. This type of device would be very useful in a hospital setting.
While phage therapy may not single-handily halt antibiotic resistance, it offers immediate help to patients who cannot afford to wait years for a new antibiotic to navigate through lengthy clinical trials to come to market. This paper not only presents compelling evidence of the efficacy of concomitant phage and antibiotic therapy but also outlines the standards and protocols essential for phage therapy implementation. Now it is up to the rest of the world to wake up and realise that time is limited, and we need to support the development of this treatment for people in dire need of alternative treatments.
Poster image taken with permission. Credit to Prof. Ilari Maasilta from Nanoscience Center at University of Jyvaskyla in Finland. Picture shows helium ion microscopy image taken of phages and bacteria.
- M. Merabishvili, J. P. Pirnay, D. De Vos, Guidelines to Compose an Ideal Bacteriophage Cocktail. Methods Mol Biol 1693, 99-110 (2018).
- J. P. Pirnay et al., The Magistral Phage. Viruses 10, (2018).
- M. H. Adams, Bacteriophages. (Interscience Publishers, New York, 1959), pp. 1-8.
- S. I. Green et al., A Retrospective, Observational Study of 12 Cases of Expanded-Access Customized Phage Therapy: Production, Characteristics, and Clinical Outcomes. Clin Infect Dis 77, 1079-1091 (2023).
- J. P. Pirnay, G. Verbeken, Magistral Phage Preparations: Is This the Model for Everyone? Clin Infect Dis 77, S360-S369 (2023).
Follow the Topic
-
Nature Microbiology

An online-only monthly journal interested in all aspects of microorganisms, be it their evolution, physiology and cell biology; their interactions with each other, with a host or with an environment; or their societal significance.
Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in