Remdesivir improves biomarkers associated with disease severity in COVID-19 patients treated in an outpatient setting
Published in Healthcare & Nursing
Coronavirus 2019 disease (COVID-19), which is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), quickly progressed to a global pandemic following its identification in 2019. As of December 8, 2022, there have been 7.2 million reported deaths and 17.8 estimated deaths attributed to the disease. In the early stages of the pandemic, there was a rush to identify reliable biomarkers related to COVID-19. The goal was to identify which biomarkers were associated with the susceptibility of progressing to severe disease so that patients could be stratified by risk and resources could be allocated most effectively. Multiple risk factors are now known to contribute to severe disease in patients who contract SARS-CoV-2, including advanced age and pre-existing medical conditions, such as diabetes, hypertension, and heart disease, all of which can impact biomarker concentrations.
COVID-19 has been found to lead to changes in a variety of biomarkers, including those related to the immune system. For example, lymphopenia (reduction in white blood cell count), which can occur in elderly COVID-19 patients and in those with type 2 diabetes with high blood sugar, has been associated with poor clinical outcomes.1 Although numerous biomarkers have been identified to assist in determining which patients are most susceptible to severe disease and how COVID-19 impacts such factors, the question remains as to which biomarkers may provide insight into the efficacy of COVID-19 therapeutics, such as antiviral drugs.
Remdesivir (RDV; Veklury) is a nucleoside analog that binds to the viral RNA-dependent RNA polymerase, the enzyme that makes copies of the viral RNA, and inhibits SARS-CoV-2 viral replication. Clinical trials with RDV showed that a 3-day course of the drug could lower the risk of hospitalization and death by 87% in non-hospitalized patients who were at risk for COVID-19 progression.2 Studies in patients hospitalized with COVID-19 found that those who received RDV recovered faster than those who received placebo (median of 10 days vs. 5 days, respectively) and were discharged sooner (median initial hospital stay of 12 days vs. 17 days, respectively).3
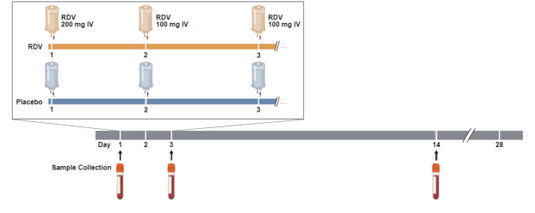
In the study presented here, we aimed to identify biomarkers associated with clinical outcomes for patients with COVID-19 and the impact of RDV treatment on these biomarkers over time. The patients included in the study had tested positive for SARS-CoV-2 and were considered at risk for severe disease because of their age (≥60 years) or the presence of other risk factors, such as chronic lung, kidney, liver, or heart disease; hypertension; diabetes; obesity; immunocompromised; cancer; or sickle cell disease. Patients enrolled in the study had a baseline blood sample taken and were then given RDV or placebo (Fig. 1). Additional blood samples were taken on days 3 and 14 after treatment to assess how blood, inflammation, and coagulation biomarkers were impacted by RDV.
One of the first observations made was that in patients who were either hospitalized or had died by day 28 of the study, several of the biomarkers that have been previously shown to correlate with severe disease were significantly elevated. This included soluble angiopoietin 2 (sAng2), a growth factor involved in the formation of new blood vessels; ferritin, an iron storage protein; interleukin 6, a cytokine produced by a variety of cells after tissue damage; lactate dehydrogenase, an enzyme involved in cellular respiration; procalcitonin, a precursor molecule involved in calcium homeostasis; and C-reactive protein (CRP), a protein produced by the liver that increases during inflammatory responses.
We next sought to determine how RDV treatment might impact expression of these biomarkers over time. Although patients in this study did have elevated CRP at baseline, levels of this biomarker were not significantly different in patients who received RDV versus those who received placebo by day 14. Other biomarkers exhibited significant changes based on the patient’s treatment regimen. For example, patients who received RDV had significantly reduced levels of sAng2 and ferritin compared with those who received placebo. One of the most pronounced differences was in a biomarker known as D-dimer, a breakdown product of blood clots that is associated with poor prognosis in patients hospitalized with COVID-19. While patients who received placebo continued to exhibit increases in D-dimer concentrations throughout the study period, patients who received RDV quickly returned to baseline levels. Expression of most of the biomarkers assessed was lower in patients treated with RDV, but some patients had increased expression compared with those in the placebo group. One example included the concentration of lymphocytes, which was significantly elevated at day 3 in patients who received RDV compared with those who did not receive the drug.
Although it might seem logical that the same biomarkers used to determine the severity and disease progression of COVID-19 could be used to measure therapeutic efficacy, this does not appear to always be the case. While some biomarkers, such as CRP, may serve as valuable predictors of disease progression, they do not always appear to be impacted by antiviral use. Conversely, other factors, such as D-dimer and lymphocyte concentrations, changed significantly with RDV and may therefore be most relevant when assessing antiviral efficacy. Overall, these findings provide important insight into how RDV impacts key biomarkers of COVID-19 severity.
References
- Tan, L. et al. Lymphopenia predicts disease severity of COVID-19: a descriptive and predictive study. Signal Transduct. Target. Ther. 5, 33 (2020).
- Gottlieb, R. L. et al. Early remdesivir to prevent progression to severe Covid-19 in outpatients. N. Engl. J. Med. 386, 305-315 (2022).
- Beigel, J. H. et al. Remdesivir for the treatment of Covid-19 — final report. N. Engl. J. Med. 383, 1813-1826 (2020).
Follow the Topic
-
Communications Medicine
A selective open access journal from Nature Portfolio publishing high-quality research, reviews and commentary across all clinical, translational, and public health research fields.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Public health and health governance in China
Publishing Model: Open Access
Deadline: Jul 31, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in