Serum paraprotein persistence and size determine outcome in a cohort of patients with a modern definition of plasmacytoma with up to 19 years of follow up
Published in Cancer
Solitary plasmacytoma is characterized by the presence of a localized collection of plasma cells in the absence of bone marrow or other areas of involvement. This condition, which is extremely rare, carries a risk of progression to myeloma myeloma estimated to be up to 50% in the first 5 years after diagnosis. Risk factors used to identify progression in the past have included location, serum free light chain values, paraprotein persistence, size and bone marrow involvement by flow cytometry, among others. Identification of solitary plasmacytoma patients with high risk of progression to multiple myeloma is an important step to design prevention strategies.
With this in mind, we designed a retrospective study to evaluate all consecutive patients with solitary plasmacytoma from 1986 until 2015 evaluated at our institution and create an easy to use, clinical model of progression. 71 patients [50 solitary bone plasmacytoma (SBP) patients and 21 solitary extramedullary plasmacytoma (SEP) patients] were included. All these patients had assessments with advanced imaging (CT, MRI and/or PET/CT) and evaluation of occult marrow disease by at least 4-color flow cytometry. The aim of our study was to identify factors predicting progression to myeloma. Progression to myeloma was defined as having a new plasmacytoma lesion outside the radiation area, multiple plasmacytomas and/or development of the new SLiM CRAB criteria.
Most of the patients in our cohort (94%) were treated with localized radiation with or without surgery/chemotherapy. In our analyses, we included known clinical factors such as age, LDH, gender, β2-microglobulin, plasmacytoma type (solitary or extramedullary), immunoparesis, serum monoclonal protein presence at baseline, serum monoclonal protein persistence at 1 year after treatment, involved/uninvolved serum free light chain ratio, Bence Jones proteinuria, plasmacytoma size and radiation dose. With a median follow-up time of 9.4 years (range 0.3-19.5 years), about half of patients (35/71) progressed to MM. Our results confirmed the higher rate of progression in patients with bone (56%) versus extramedullary (33%) plasmacytoma. We also observed that all extramedullary plasmacytoma patients progressed within 5 years of diagnosis whereas bone plasmacytoma patients continued to progress even after 14 years of diagnosis.
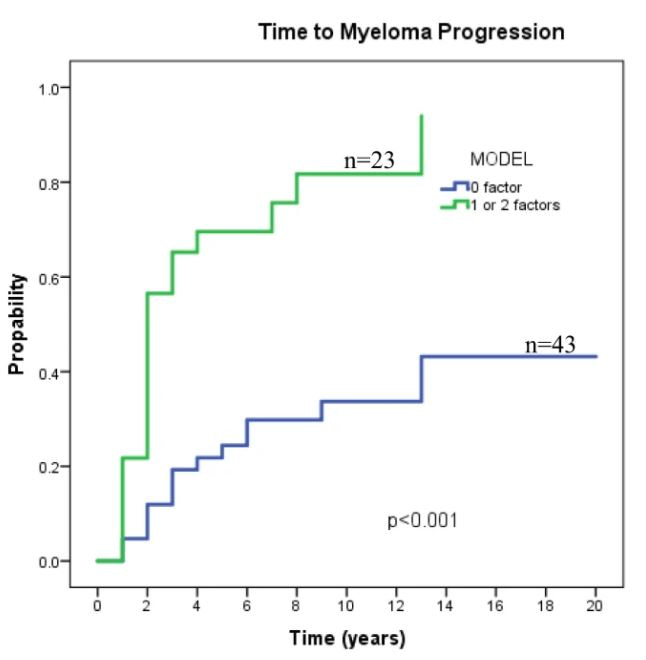
Two factors, the persistence of serum monoclonal protein one year after treatment and plasmacytoma size > 10 cm were identified in multivariate analysis backwards selection model as risk factors for progression to myeloma with hazard ratios of 4.9 (95% CI 1.9 – 12.5; p=0.001) and 4.5 (95% CI= 1.4 – 14.5; p<0.012), respectively. This model (plasmacytoma >10 cm. and persistence of paraprotein 1 year after diagnosis) predicted, in our cohort, the progression risk to MM in SP patients (19% vs 65%, 30% vs 70%, and 39% vs 82% in patients with 0 and 1 or 2 factors, respectively).
We describe an easy to use clinical model including two variables: plasmacytoma >10 cm. at baseline and persistence of paraprotein after one year after treatment to predict progression to MM and may provide insights into the progression of patients in the clinic. Given the rarity of this condition, international efforts are needed to lead adjuvant treatment studies in solitary plasmacytoma.
Follow the Topic
-
Blood Cancer Journal
This journal seeks to publish articles of the highest quality related to hematologic malignancies and related disorders.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in