SpFN+ ALFQ vaccine induces the rapid generation of SARS-CoV-2 spike-specific Tfh cells and binding and neutralizing antibody responses
Published in Healthcare & Nursing
Two major events, the September 11, 2001 attack (9/11) and the emergence of the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in 2020 completely changed the world and our outlook towards life in general. Both events showed our vulnerability, hostage to terrorist attacks in the former case and to a deadly virus in the latter scenario. The COVID-19 pandemic, the staggering number of deaths, and COVID related issues for those who recovered from COVID infection highlighted the importance of rapid development of vaccines. In the U.S., two vaccines based on the mRNA platform were produced in record time and were the first ones to be authorized for emergency use at the height of the pandemic which saved millions of lives.
It is well established that neutralizing antibodies play a key role in preventing infection. The global emergence of circulating new SARS-CoV-2 variants of concern (VOC) continues to cause surges in COVID-19 infections and deaths. Neutralizing antibodies have been impacted by mutations in the spike and RBD domains in VOC, resulting in escape mutants. While the current mRNA vaccines are effective in reducing the severity of the infection and the death rates, they do not induce durable cross-protective neutralizing antibody responses, thus requiring frequent booster vaccinations. These unique challenges necessitate the need for next generation vaccines that could induce durable cross-protective neutralizing antibody responses and currently this is an active area of research.
The four major structural proteins of SARS-CoV-2 consist of the spike glycoprotein (S), which is further subdivided to S1 and S2 subunits, nucleoprotein, envelope protein, and membrane protein . The S1 subunit consists of the receptor-binding domain (RBD) that is involved in virus entry by binding to the human angiotensin-converting enzyme 2 (hACE2) receptor, while the S2 subunit mediates the subsequent membrane fusion (1).
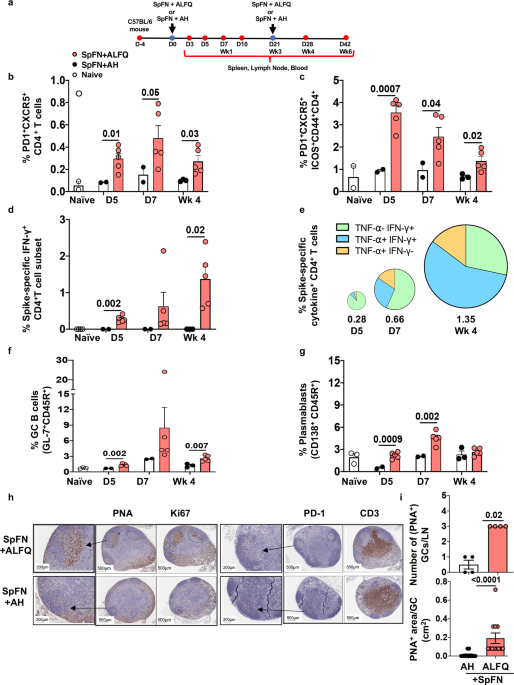
We recently developed a self-assembling SARS-CoV-2 sub-unit vaccine based on a ferritin nanoparticle platform that displays 8 copies of a prefusion stabilized spike glycoprotein trimer of the Wuhan-Hu-1 strain of SARS-CoV-2 on its surface in an ordered array (SpFN) (2). We adjuvanted this nanoparticle with either aluminum hydroxide (AH) or with a potent Army Liposomal Formulation, ALFQ that contains 2 immunostimulants, synthetic monophosphoryl lipid A (3D-PHAD) and the saponin QS-21 in addition to saturated phospholipids and 55 mole% cholesterol (3, 4). We previously demonstrated that SpFN+ALFQ vaccine induced enhanced recruitment and activation of antigen-presenting cells, polyfunctional T cell responses, high titer antibody responses, leading to durable SARS-CoV-2 specific adaptive and protective immune responses in several animal models (5, 6).
It has been documented that adjuvants play a critical role in the type and durability of immune responses generated. In the current study, we investigated the influence of adjuvants on the immune response, specifically the impact of two adjuvants (SpFN +ALFQ vs SpFN + AH) on the development of T follicular helper (Tfh) and B cells, and their influence on antibody responses in mice. Tfh cells are specialized cells that help in the differentiation of B cells into plasmablasts to produce class-switched antibodies within secondary lymphoid organs. Following a single vaccination, our findings highlight the importance of SpFN + ALFQ compared to SpFN + AH vaccination in orchestrating the rapid induction of (i) higher frequency of spike-specific IL-21-producing Tfh cells and germinal center (GC) B cells; (ii) significantly higher titers of S-2P-specific IgM and IgG antibodies as early as day 5 post vaccination; (iii) robust cross-neutralizing antibodies against the parental SARS-CoV-2 strain and VOC (Alpha, Beta, Delta) as early as day 7. The early engagement of S-2P specific naive B cells and high titer IgM antibodies shape the development of long-term neutralization breadth as seen in this study. Notably, subsequent vaccinations induced high levels of neutralizing antibodies against the Omicron variant as well. In contrast, no cross-neutralizing antibodies against Omicron were induced with SpFN +AH. The other hallmarks of the study were the induction of high avidity binding and functional antibodies and long-lived plasma cells in the bone marrow following vaccination with SpFN + ALFQ (Graphical Figure 1).
These data as well as other preclinical studies demonstrating the efficacy and potency of this vaccine led to the safety and immunogenicity evaluation of SpFN + ALFQ vaccine in a phase 1 clinical trial (ClinicalTrials.gov Identifier: NCT04784767). The vaccine was found to be safe with no serious adverse events. The trial has been completed and the safety and immunogenicity results will be published soon. Since the antibody responses are not sustained over a period of time with the current COVID vaccines and ALFQ has proven to be an extremely potent adjuvant, it is tempting to speculate that a protein boost with ALFQ as the adjuvant following mRNA prime might increase the durability and the breadth of the neutralizing antibody responses and could form the basis for a pan-coronavirus vaccine.
For more detail on the experiments and results, please read our paper.
npj Vaccines (2023) 8:43; https://doi.org/10.1038/s41541-023-00638-6
References
- Hoffmann M, A, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–80.
- Joyce, M. G. et al. SARS-CoV-2 ferritin nanoparticle vaccines elicit broad SARS coronavirus immunogenicity. Cell Rep. 37, 110143 (2021).
- Rao M, Peachman KK, Alving CR. Liposome Formulations as Adjuvants for Vaccines. Curr Top Microbiol Immunol. 2021;433:1-28.
- Alving CR, Rao M, Matyas GR. Similarities and differences of chemical compositions and physical and functional properties of adjuvant system 01 and army liposome formulation with QS21. Front Immunol. 2023 Jan 25;14:1102524.
- Carmen, J. M. et al. SARS-CoV-2 ferritin nanoparticle vaccine induces robust innate immune activity driving polyfunctional spike-specific T cell responses. NPJ Vaccines 6, 151 (2021).
- Joyce, M. G. et al. A SARS-CoV-2 ferritin nanoparticle vaccine elicits protective immune responses in nonhuman primates. Transl. Med. 14, eabi5735 (2022).
Follow the Topic
-
npj Vaccines
A multidisciplinary journal that is dedicated to publishing the finest and high-quality research and development on human and veterinary vaccines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Therapeutic HPV vaccines
Publishing Model: Open Access
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in