Sticking around for the hard part
Published in Bioengineering & Biotechnology, Microbiology, and Cell & Molecular Biology

Millions of years of evolution have produced bacteria that can survive in almost every imaginable niche, frequently in a biofilm state. Biofilms can conquer surfaces almost everywhere in nature and our everyday life. Biofilms have the capacity to colonize hard surfaces, such as rocks near the river, the basin in the kitchen, orthopedic implants, and our teeth (as you may know to your chagrin as your dental hygienist gently chastises you). Biofilms can also colonize soft surfaces such as living tissues and contact lenses (which is why good lens and case hygiene is very important if you use contacts).
However, despite the near-ubiquity of biofilms on all kinds of surfaces, very little is known about the interplay of surface mechanics with biofilm formation. Open questions include: Does the surface hardness or softness matter in the bacterial “decision” to form a biofilm, initiated by the attachment and accumulation of planktonic bacteria onto surfaces? If yes, how do bacteria perceive surface stiffness? And what are the underlying mechanisms? For the biofilm-forming pathogen Pseudomonas aeruginosa, there are at least two possible mechanosensory elements relevant to surface attachment; these are the type-IV pili that drive twitching and the cell-surface-exposed protein PilY1. Are either, or both, of these elements involved in bacterial mechanosensing of surface stiffness and consequent transition to the biofilm phenotype?
To address these questions, making surfaces with different stiffness but with the same adhesivity and chemistry is the first challenging step. Inappropriate fabrication has the very real risk of introducing unintended changes to other surface properties, such as surface energy, porosity, or the density of adhesion sites, and thereby impacting adhesivity. In such a case, changes in adhesivity would be a confounding factor that made it difficult or impossible to disentangle the effects of adhesivity from those of surface stiffness.
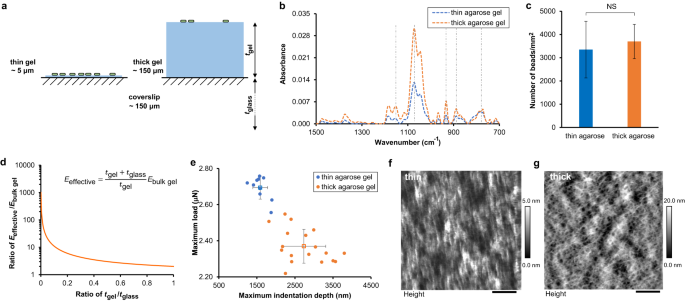
Therefore, we fabricated surfaces consisting of a layer of hydrogel coated on a glass coverslip. By varying the thickness of hydrogel by an order of magnitude, we achieved surfaces with different effective stiffness while the surface chemistry remained unchanged. Consider an everyday-life example of a thin yoga mat versus a thick yoga mat; on a thin mat, you strongly feel the underlying studio floor. Or consider the anti-fatigue mats some people have in their kitchen – if the mat material is the same, a thicker mat will feel softer underfoot than will a thinner mat. Since the material of the hydrogel was the same regardless of thickness, adhesivity was unchanged.
What mechanical cues do bacteria perceive as they attach to surfaces with different stiffness? From equilibrium mechanics, we expect that, if adhesivity is unchanged, the bacteria attached to stiffer surfaces would undergo greater cellular deformation than those attached to softer surfaces. Greater deformation means larger changes in the mechanical stress and strain states of bacterial cell envelopes. This expectation is confirmed by our observation that the activity of mechanosensitive ion channels on bacterial cell membrane increases upon surface attachment and the increase is greater on thin gels, which act as a stiffer surface than thick gels.
How, then, do bacteria respond to surface mechanics and how might this be linked to biofilm development? Using quantitative microscopy, we found that bacteria accumulate more on stiffer surfaces, EXCEPT for those bacteria that lack the PilY1 protein. Surface thickness does not impact the likelihood of bacteria encountering and initially interacting with the surface. However, bacteria on thin surfaces increase levels of intracellular cyclic-di-GMP much more than do bacteria on thick surfaces. Cyclic-di-GMP is a second messenger that controls biofilm initiation in a wide range of species. Cyclic-di-GMP downregulates motility, and therefore higher levels of cyclic-di-GMP on thin surfaces than on thick surfaces results in a lower probability of bacteria detaching from the thin surface than the thick surface. This results in greater accumulation of bacteria on thin surfaces than on thick surfaces.
Mutants lacking PilY1 have very similar levels of cyclic-di-GMP, motility, and detachment regardless of surface thickness, and as a result the accumulation of DpilY1 mutants is the same on both thick and thin surfaces. Thus, the bacterial “decision” to remain on the surface arises from bacterial mechanosensing and is mediated by the cell-surface-associated protein PilY1.
We suggest that changes in the mechanical stress and strain state of the cell envelope will occur any time bacteria stick to a surface, and that these changes will be more pronounced for gram-negative bacteria than for gram-positive. Given the near-ubiquity of biofilm formation and their importance as a problem in healthcare and in the built environment, this suggests that bacterial mechanosensing of surface mechanics, and links to biofilm initiation, is likely vastly understudied. Such studies have the potential to give rise to new approaches to biofilm prevention.
Follow the Topic
-
npj Biofilms and Microbiomes
The aim of this journal is to serve as a comprehensive platform to promote biofilms and microbiomes research across a wide spectrum of scientific disciplines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Natural bioactives, Gut microbiome, and human metabolism
Publishing Model: Open Access
Deadline: May 20, 2026
Harnessing plant microbiomes to improve performance and mechanistic understanding
Publishing Model: Open Access
Deadline: Jun 01, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
Very nice work in biofilms. Thanks for sharing. Please check my work that was recently published online in nature communications.
https://go.nature.com/3RduzBM