Sustainable Ethene Production: Solar Selective Catalytic Dehydrogenation of Ethane
Ethylene, the world's most widely used organic compound and the most fundamental building block for the chemical industry, is predominantly produced through high-temperature steam cracking of ethane derived from fossil fuels. This energy-intensive process contributes significantly to greenhouse gas emissions. In consideration of this challenge, our recent work presents a sustainable alternative pathway for the selective conversion of ethane to ethylene and a bonus equimolar co-product, hydrogen, driven solely by sunlight and operating under ambient conditions. This solar-driven catalytic process offers a renewable and environmentally friendly route for ethylene and hydrogen production, circumventing the need for fossil fuel-based feedstocks and the associated carbon footprint of conventional steam cracking (Nature Energy, DOI:1038/s41560-024-01541-7).
In this work, we designed an isomorphic copper-substituted lanthanum manganese oxide perovskite (LaMn1-xCuxO3) photocatalyst and demonstrated its ability to selectively convert ethane into ethylene and hydrogen, two valuable and readily separable green commodity chemicals, through a non-oxidative photocatalytic dehydrogenation process. This perovskite oxide possesses redox-active Lewis acid sites comprising Mn(III) and Mn(IV), and redox-active Lewis base sites, comprising O(-II) and OH(-I), collectively dubbed surface frustrated Lewis pairs (SFLPs). Under light illumination and without external heating, the catalyst achieved an impressive ethylene production rate of 1.1 mmol/g/h with an ethane conversion of 4.9%. The similarity in surface temperatures between copper-substituted photocatalyst and unsubstituted control indicates significant photochemical contributions to the photocatalytic reaction with minor contribution from photothermal conversion reaction pathway. Notably, a rapid increase in the ethylene production rate was observed only above 600°C in dark for both samples, supporting the photochemical role of light illumination during the reaction. Remarkably, the activation energy (Ea) exhibited a drastic reduction with light irradiation compared to dark reaction conditions, emphasizing the solar advantage of the photo-dehydrogenation of ethane compared to the thermal process (Figure 1). This observation begs the question of how photo- and photothermal chemistry enable the photocatalytic dehydrogenation of ethane to ethene and hydrogen?
To answer this question, in-depth investigations using diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) and nuclear magnetic resonance (NMR) were implemented to provide insights on the surface reaction pathway and evidence for the role of light-mediated SFLP in the observed ethane conversion. Temperature-programmed desorption (TPD) experiments with ethane under dark and light conditions provided strong and direct evidence for light-enhanced chemisorption of ethane, further supporting the photochemical role of light in addition to the photothermal contribution. Detailed analyses reveal that this catalytic process over the copper-doped lanthanum manganese oxide perovskite operates in the photoexcited state, and benefits from the tailored SFLP produced by photogenerated electron-hole pairs. Additionally, non-radiative electron-hole recombination can produce local heating through phonon confinement, introducing a photothermal advantage to the overall photochemical process. During the revision process of this article, one of the reviewers for the article have posed a challenging yet insightful question during the revision process, of whether radicals induced by the traditional photoinduced charge transfer process could have participated in the reaction. Rising to this challenge, we conducted additional experimental and theoretical analyses, which suggested that while the conventional radical mechanism could occur in our system, it was less likely to be responsible for the observed light-enhanced activity.
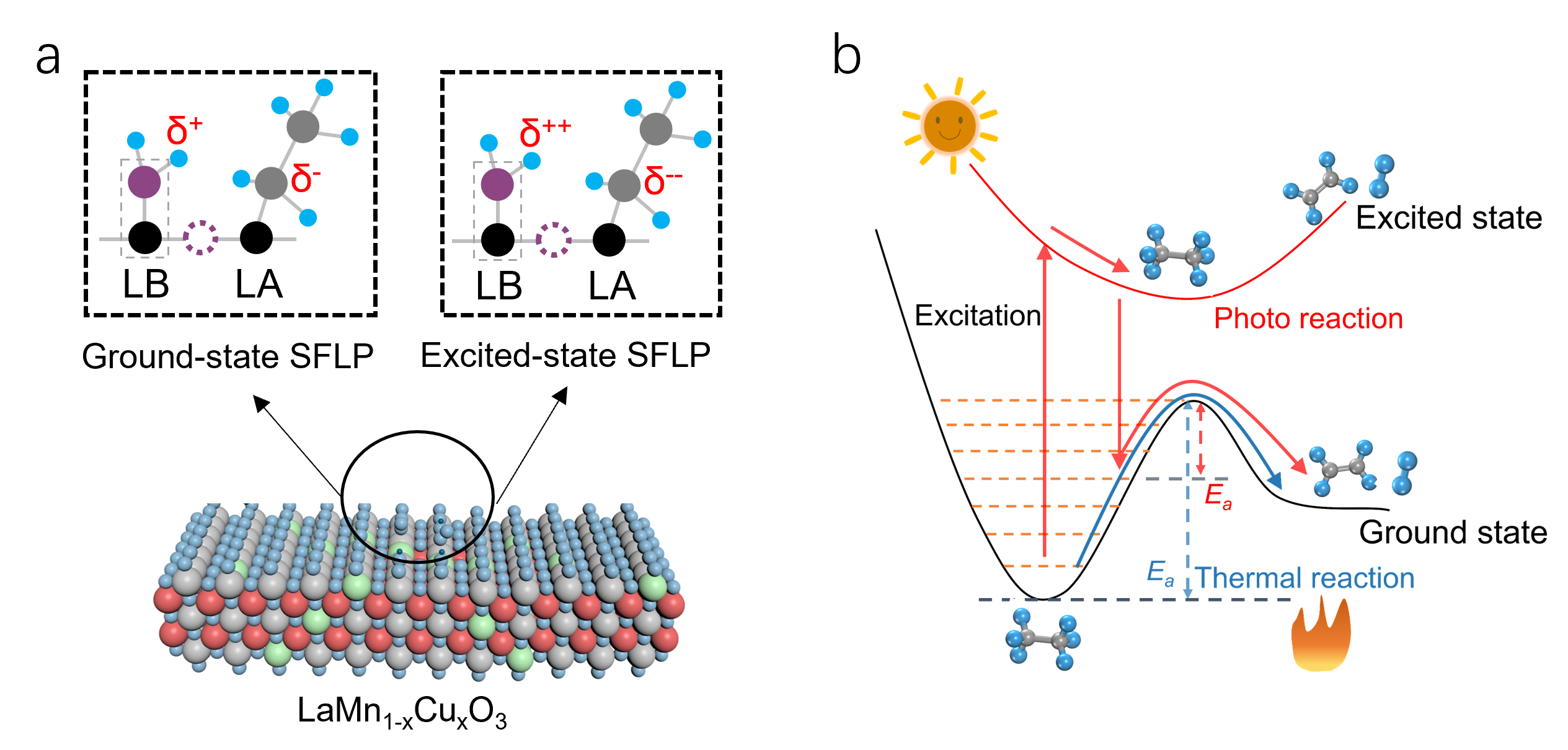
Figure 1. a. Illustration of ground-state and excited-state SFLP in LaMn1-xCuxO3. b. Energy schematic of a photocatalytic compared to a thermocatalytic ethane to ethene dehydrogenation reaction over LaMn1-xCuxO3.
To demonstrate the practical viability and scalability of this solar-driven ethane-to-ethylene/hydrogen process, a rooftop prototype reactor was constructed and tested. Its successful operation illustrates the potential utilization of solar energy in our system for ethylene and hydrogen production. Furthermore, a comprehensive techno-economic analysis was conducted to evaluate the energy and economic potential of an industrial-scale, continuous 24/7 process powered by light-emitting diodes (LEDs). This analysis reveals that the use of LEDs can overcome the intermittency and cloud fluctuations in solar light intensity, enabling consistent and reliable ethylene production. The techno-economic evaluation highlights the potential of this sustainable, solar-driven approach to be implemented on an industrial scale, offering an economically viable, energy- and carbon-efficient alternative to conventional fossil fuel-based ethylene production routes.
This work unveils a novel photochemical and photothermal pathway that harnesses both the light and heat components of solar energy, which marks a key advantage for solar irradiation in gas-phase heterogeneous photocatalysis. Out results represent a significant advancement with promising potentials for the development of a practical, sustainable solar ethylene and hydrogen production process from the ethane component of abundant natural gas. By circumventing the need for fossil fuel-based feedstocks and leveraging the renewable energy nature of sunlight, the newfound ability to photo catalytically dehydrogenate ethane to ethene and hydrogen can greatly reduce the energy consumption and carbon footprint associated with conventional industrial ethylene production processes. This advance bodes well for a more environmentally friendly and sustainable source of two of the most important chemical and energy vectors, ethylene and hydrogen.
In retrospect of this research journey, I am profoundly grateful to Professor Ozin for his unwavering guidance, invaluable insights, and the confidence he instilled in me throughout this project. Our group has long been working on surface frustrated Lewis pairs for heterolytic hydrogen splitting. When I shared the results with Professor Ozin, he incisively suggested that a similar SFLP heterolytic process might be responsible for ethane activation. Discussions with Prof. He and Prof. Zhang, to whom I am sincerely thankful for the supervision and the ideas provided, have further educated me on and convinced me of the SFLP-mediated mechanism. These hypotheses and ideas were next evidenced through communication and collaboration with Dr. Guanshu Zhao for density functional theory (DFT) studies, aiming at obtaining evidence on the effect of SFLP towards the heterolytic splitting of ethane. Combining our experimental evidence and theoretical results, we were pleasantly surprised at the proposed reaction pathway. I express my sincere gratitude to all collaborators for their invaluable help and support, as well as the contributions from the reviewers and editors, which significantly improved the quality and impact of this work.
This research would not have been possible without the strong and sustained financial support of the National Natural Science Foundation of China, Natural Sciences and Engineering Research Council of Canada, and Hydrofuel Canada Incorporated.
Follow the Topic
-
Nature Energy
Publishing monthly, this journal is dedicated to exploring all aspects of this on-going discussion, from the generation and storage of energy, to its distribution and management, the needs and demands of the different actors, and the impacts that energy technologies and policies have on societies.
Related Collections
With Collections, you can get published faster and increase your visibility.
Microgrids and Distributed Energy Systems
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in