Sustaining gut health with specialized binding proteins
Published in Bioengineering & Biotechnology, Public Health, and Agricultural & Food Science
Food- and waterborne diseases are often caused by common enteric infections, which are transmitted via the fecal-oral route. These diseases typically involve harmful viruses, microbes, and/or toxins and may give rise to clinical symptoms ranging from mild diarrhea (e.g. traveler’s diarrhea) to severe diarrheal disease that necessitates hospitalization and can be fatal. They affect animals and humans alike, and all of us will encounter these diseases during our lifetime.
According to the World Health Organization (WHO), 31 foodborne hazards (e.g., bacterial, viral, or parasitic) caused 600 million clinical incidences and 420,000 deaths worldwide in 2010 [1]. Furthermore, diarrheal diseases remain the second leading cause of death in children under 5 years of age, with approximately 1.7 billion cases of childhood diarrhea and 525,000 child deaths every year [2]. The areas typically most affected are developing regions, yet diarrheal disease still have an enormous impact on gut health in developed countries as well. Indeed, in the United States alone, 1 in 6 people contract a foodborne disease each year, incurring 3,000 deaths [3].
Post-weaning diarrhea in piglets: A significant industrial challenge and a model for human gut health
In our newly published article, we demonstrate how piglets that are fed with specialized binding proteins (i.e., single-domain antibody constructs) are protected against infections from enterotoxigenic Escherichia coli (ETEC) after weaning. This is an important result, since weaning is a highly vulnerable phase of life for piglets, as they are transitioning from milk to feed, have an immature gastrointestinal (GI) tract and immune system, and are stressed due to their new life without the mother sow. The incidence of post-weaning diarrhea (PWD) is therefore high, as ETEC strains present in the environment have perfect conditions for colonizing the intestine of the piglets. This consequently results in reduced animal welfare, low productivity, and high usage of antibiotics in this group of animals. Therefore, better and more sustainable ways of managing PWD are greatly needed.
The reason why ETEC strains are pathogenic is because they carry a range of virulence factors, including fimbrial adhesins and toxins, which are critical for establishing infection and proliferating in the GI tract of piglets. While still suckling, piglets are protected against these virulence factors by maternal antibodies in the sow’s milk, which thus protect the piglets through passive immunity. By designing binding proteins, which block key ETEC virulence factors, we can emulate this passive immune protection and sustain health in piglets during weaning, while their own immune system and gut microbiota mature (Figure 1). In our study, we show this by using daily supplementation of two binding proteins that block an ETEC toxin and an adhesin, by which we strengthen the GI tracts of the piglets against an ETEC challenge. Specifically, the presented research shows how daily supplementation of specialized binding proteins can:
- Reduce ETEC proliferation in the piglet GI tract after an oral challenge with the bacterium.
- Support healthy gut microbiota maturation, seen as an increase in beneficial bacterial genera for feed digestion and utilization
- Bind to ETEC adhesins and toxins without affecting ETEC growth
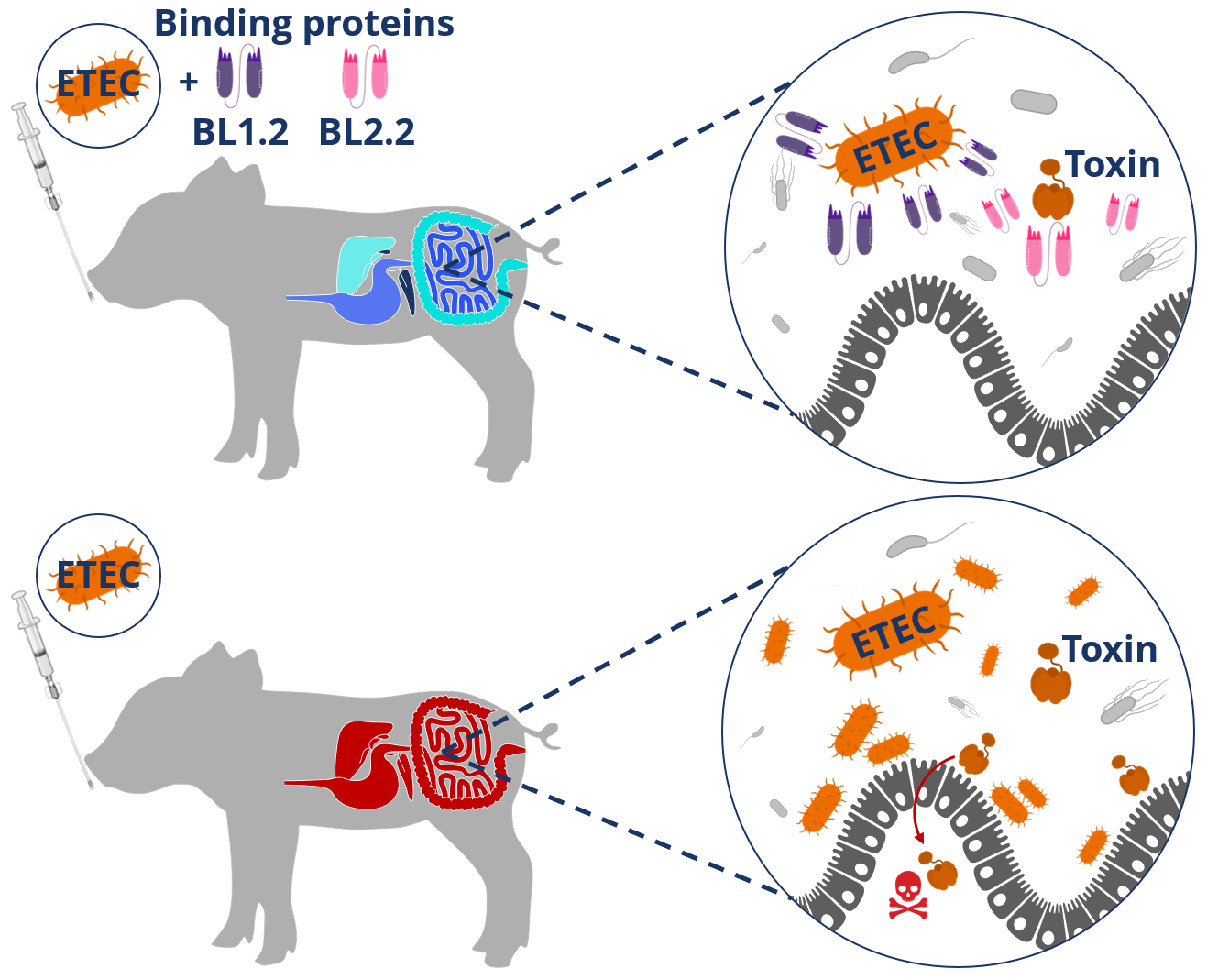
Figure 1. Schematic representation of the protective effect of two binding proteins (BL1.2 and BL2.2) in the piglet GI tract (upper section) versus a gut disrupted by ETEC (lower section) after ETEC challenge.
The presented research thus exemplifies how binding proteins can be used to provide passive immunity to piglets, which could be useful for industrial pig production to support better animal health and productivity. Yet, the research also points towards applications in human health, as ETEC infections remain a significant global challenge also for us. In fact, human ETEC infections are facilitated by very similar virulence factors as those related to PWD in piglets.
Sustaining global gut health in the face of climate change
ETEC infections in humans are mostly recorded as food- and waterborne diseases in developed countries, accounting for a significant part of bacterial traveler's diarrhea as well as domestic food poisoning. Traveler's diarrhea impacts 10-40% of travelers worldwide and results in millions of lost travel days and absence from work [4]. Deployed personnel, like military troops, report incidence rates up to 1 case per 2.7 person-months [5], which leads to the loss of a high number of active duty days. Traveler’s diarrhea mediated by bacteria is primarily caused by ETEC, enteroaggregative E. coli (EAEC), and Campylobacter, which combined account for 62% of cases in deployment scenarios. ETEC is, however, the predominant contributor to this high number [5]. In developing countries, ETEC is endemic in large regions, with seasonal outbreaks following monsoon season, similar to cholera [6]. ETEC-mediated diarrhea can range from self-contained cases to severe dehydration in some adults and in many children. Overall, this contributes to significant morbidity and mortality in resource-poor settings [7]. The problem is in fact so grand that ETEC and cholera alone are estimated to account for up to 50% of all cases of diarrhea in Bangladesh, and co-infection is often recorded [8].
Looking to a different region, such as Zambia, a disastrous cholera outbreak caused by heavy rainfall and flooding is currently ongoing [9]. This is noteworthy, as it could indicate that infectious diseases are likely to cause increased human suffering in areas that are not prepared for climate change. In fact, researchers estimate that over half of all known human pathogenic diseases will be aggravated by climate change [10] – this includes ETEC.
Since 1990, deaths and disease burden from enteric infections have decreased significantly, with approximately 50% [11]. However, the number of incidences and individuals affected remains alarmingly unchanged [11]. This highlights that while the treatment of disease has improved (mostly driven by increased use of antimicrobials), our ability to maintain health and prevent enteric infections has not. This is a critical gap in global management of gut health and a large unmet need. Addressing the root causes of enteric infections, and enhancing overall health, can significantly decrease both their incidence and prevalence. The specialized binding proteins developed and presented in this newly published article, as well as other strategies that involve protection by passive immunity, will not solve all these problems caused by infectious pathogens. However, we believe that the approach presented in our newly published article could help protect animals and humans against many of these threats by maintaining and supporting health before we get sick, as exemplified here on ETEC-mediated PWD in piglets.
References
- Havelaar, A. H. et al. World Health Organization Global Estimates and Regional Comparisons of the Burden of Foodborne Disease in 2010. PLoS Med 12, e1001923 (2015).
- WHO, Diarrhoeal disease. https://www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease
- Scallan, E. et al. Foodborne illness acquired in the United States--major pathogens. Emerg Infect Dis 17, 7–15 (2011)
- Leung, A. K. C., Leung, A. A. M., Wong, A. H. C. & Hon, K. L. Travelers’ Diarrhea: A Clinical Review. Recent Pat Inflamm Allergy Drug Discov 13, 38 (2019)
- Olson, S., Hall, A., Riddle, M. S. & Porter, C. K. Travelers’ diarrhea: update on the incidence, etiology and risk in military and similar populations – 1990-2005 versus 2005–2015, does a decade make a difference? Tropical Diseases, Travel Medicine and Vaccines 2019 5:1 5, 1–15 (2019)
- Chowdhury F, Rahman MA, Begum YA, Khan AI, Faruque ASG, Saha NC, et al. Impact of rapid urbanization on the rates of infection by Vibrio cholerae O1 and enterotoxigenic Escherichia coli in Dhaka, Bangladesh. PLoS Negl Trop Dis. (2011)
- Sarker MHR, Moriyama M, Rahman MM, Das SK, Uzzaman MN, Das J, et al. Characteristics of rotavirus, ETEC, and Vibrio cholerae among under 2-year children attending an urban diarrheal disease hospital in Bangladesh. J Prim Care Community Health. (2021)
- Chowdhury F, Begum YA, Alam MM, Khan AI, Ahmed T, Bhuiyan MS, et al. Concomitant enterotoxigenic Escherichia coli infection induces increased immune responses to Vibrio cholerae O1 antigens in patients with cholera in Bangladesh. Infect Immun. (2010)
- Fighting the Cholera Outbreak in Zambia | UNICEF Zambia
- Mora, C., McKenzie, T., Gaw, I.M. et al. Over half of known human pathogenic diseases can be aggravated by climate change. Clim. Chang. 12, 869–875 (2022)
- Wang Y, Huang Y, Chase RC, Li T, Ramai D, Li S, Huang X, Antwi SO, Keaveny AP, Pang M. Global Burden of Digestive Diseases: A Systematic Analysis of the Global Burden of Diseases Study, 1990 to 2019. Gastroenterology. (2023)
Follow the Topic
-
npj Biofilms and Microbiomes
The aim of this journal is to serve as a comprehensive platform to promote biofilms and microbiomes research across a wide spectrum of scientific disciplines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Microbial endocrinology
Publishing Model: Open Access
Deadline: Oct 21, 2026
Microbiome and energy metabolism
Publishing Model: Hybrid
Deadline: Dec 06, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in