Rhodium is a paradox. It is one of the most valuable and technologically essential metals, critical for automotive catalysis and fine chemical synthesis, yet it is also one of the most difficult to recover. Compared to its platinum group neighbors, palladium and platinum, rhodium resists most conventional approaches. In aqueous chloride media, it typically exists as kinetically inert and thermodynamically stable chloro-complexes such as [RhCl6]3-, which rarely undergo ligand exchange and interact only weakly with traditional adsorbents.
Our group had previously developed a series of porous organic polymer (POP) based “nanotraps” capable of efficiently capturing palladium and platinum. Naturally, we assumed rhodium would behave similarly. It did not. Early experiments revealed low uptake capacities and slow kinetics, clearly indicating that rhodium follows a fundamentally different coordination chemistry. This initial setback ultimately became the true starting point of this work.
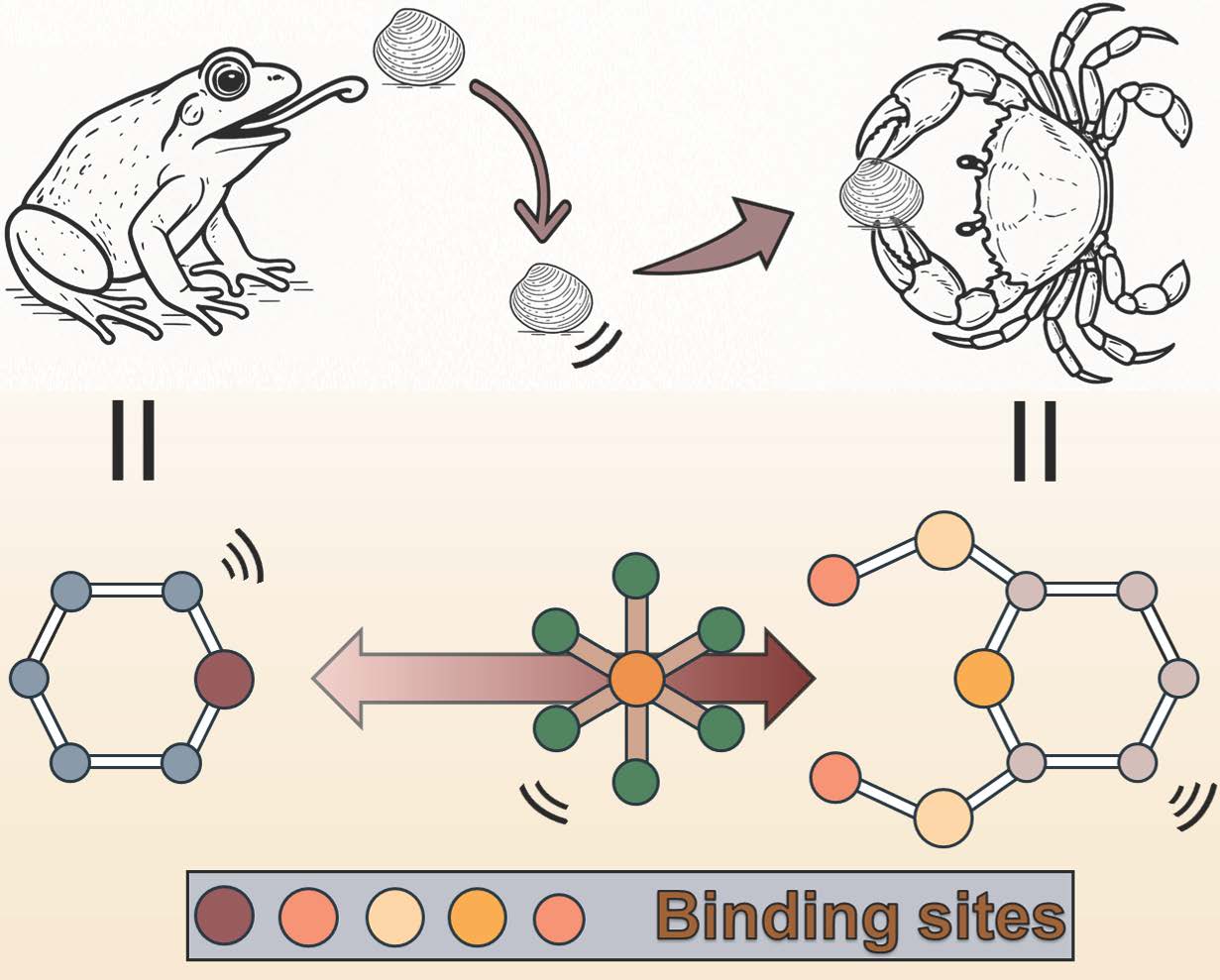
We then asked a simple but critical question: if rhodium does not readily bind to existing adsorption sites, can we redesign the binding environment to actively engage it? This led us to pincer-type chelators. Unlike traditional monodentate ligands, pincer ligands feature rigid, preorganized, multidentate coordination motifs that can bind a metal center through multiple donor atoms simultaneously (Fig.1). This provides both enhanced thermodynamic stability and improved kinetic accessibility, features that are essential for overcoming the inertness of Rh(III) chloro-complexes.
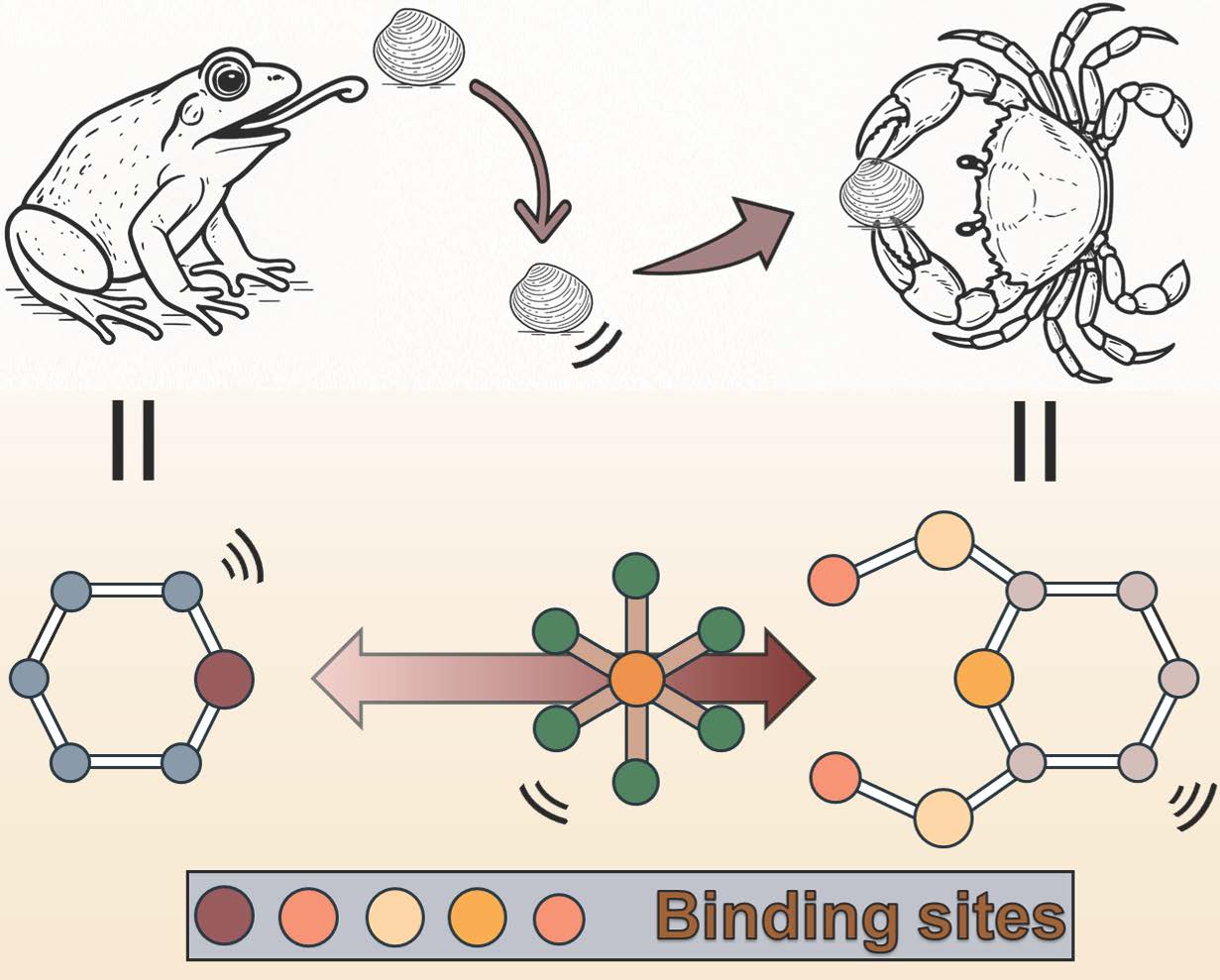
Fig. 1 | Schematic view of metal coordination with pincer ligand vs. monodentate ligand. Pincer ligands coordinate to the metal center through multiple donor atoms simultaneously, forming chelating coordination that results in stronger binding affinity and higher complex stability compared to monodentate ligands, which bind through only a single donor atom and are more prone to ligand exchange or dissociation. The rigid and preorganized geometry of the pincer ligand enforces an optimal spatial arrangement of donor atoms for metal coordination, enhancing metal–ligand interaction strength, reducing structural flexibility, and improving both thermodynamic stability and resistance to competitive coordination.
Integrating such sophisticated ligands into solid adsorbents, however, is far from straightforward. Conventional solution-based synthesis is often slow, solvent-intensive, and incomplete in polymer systems. To address this, we turned to mechanochemistry. Using a ball-milling approach under liquid-assisted grinding (LAG) conditions, we were able to install pincer functionalities into the POP framework through rapid imine bond formation (Fig.2). Reactions that would typically take hours and still remain incomplete in solution were finished within 30 minutes of milling. Equally important, this method enabled a dense and homogeneous distribution of pincer sites, effectively creating a high concentration of well-defined “nanotraps” throughout the material.
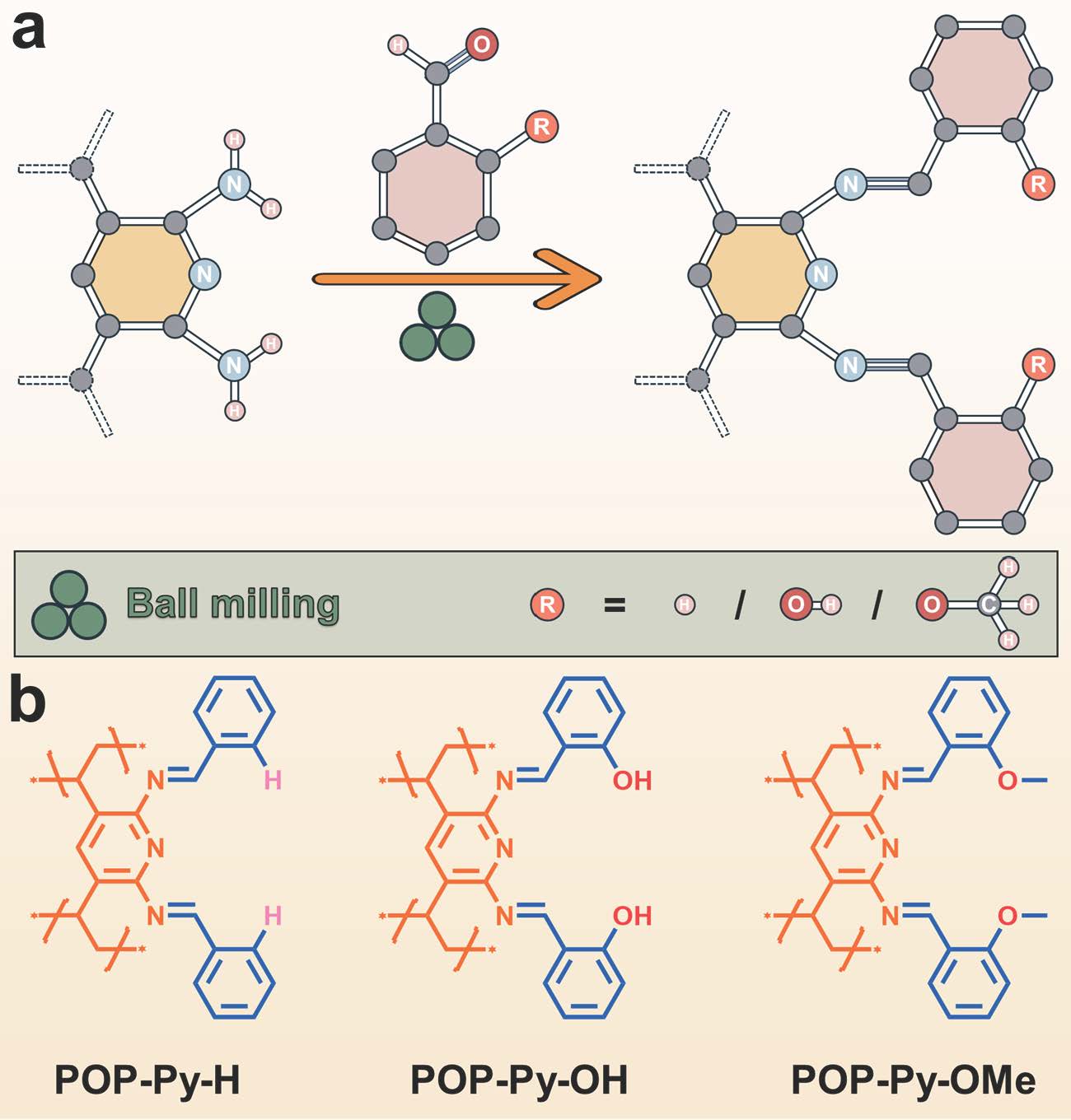
Fig. 2 | Preparation of the pincer nanotraps for rhodium recovery. (a) Schematic illustration of the solvent-free mechanochemical synthesis used to incorporate pincer-type chelators into POPs. (b) Representation of the porous frameworks functionalized with rhodium-specific pincer ligands, along with the molecular structures of the resulting pincer-functionalized POPs investigated in this study.
One particularly striking observation was the dramatic performance difference among three nearly identical materials that differ only by a single substituent: H, OH, and OMe (Fig.2). Among them, the methoxy-functionalized material (POP-Py-OMe) consistently showed the highest uptake capacity, fastest kinetics, and strongest binding affinity. Spectroscopic studies and DFT calculations revealed that the electron-donating OMe group enhances electron density at the binding site, strengthening Rh–ligand interactions. In contrast, the OH group, although potentially electron-donating, is partially engaged in hydrogen bonding and solvation, which reduces its effective coordination ability. This finding highlights how subtle electronic effects can translate into significant functional differences.
Capturing rhodium is only part of the challenge. In realistic waste streams, such as those derived from spent automotive catalytic converters, Pt, Pd, and Rh coexist alongside many competing ions. A single material proved insufficient for clean separation. Instead, we designed a modular strategy by arranging three different POP materials in sequence, each with distinct selectivity. This enabled stepwise capture, allowing platinum, palladium, and finally rhodium to be separated with high purity. Rather than relying on a single “perfect” material, this approach leverages complementary functionalities to solve a complex separation problem.
Sustainability was a key consideration throughout this work. While strong binding is beneficial for rhodium capture, it makes desorption challenging. Conventional elution methods were ineffective. We therefore took advantage of the dynamic nature of imine bonds: under acidic conditions, the pincer structures can be selectively hydrolyzed, releasing the bound rhodium and reverting the material to its precursor form. A subsequent mechanochemical step restores the active nanotrap. This degradation–reconstruction cycle proved both effective and robust, maintaining performance over multiple cycles.
This study began with a seemingly simple but persistent problem, poor rhodium adsorption. Addressing it required us to rethink adsorbent design at a fundamental level. By combining pincer coordination chemistry with mechanochemical synthesis, we developed a platform capable of efficiently capturing rhodium while integrating into a broader strategy for full PGM recovery. More broadly, this work illustrates how tackling a particularly challenging element can drive innovation in materials design. Rhodium’s resistance forced us beyond conventional approaches, ultimately opening new directions for designing task-specific adsorbents for complex separations. Sometimes, the most stubborn problems lead to the most rewarding solutions.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in