Until 2006, four infectious human retroviruses had been identified: HTLV-1, HTLV-2, HIV-1, and HIV-2 [1] (Note: Coronavirus is an RNA virus but not a retrovirus). Then an article published by Bob Silverman et al sounded like a long-waiting breakthrough: in a long-established human prostate cancer cell line, they identified a new retrovirus that was highly similar to mouse viruses that cause leukemia. They gave this virus the rather ungainly name, "xenotropic murine-leukemia virus-related virus" (XMRV).
This caught the attention of Dr. John Coffin, a Professor at Tufts University in Boston.
John is a virologist whose research focuses on HIV. However, he is also very interested in how retrovirus impacts the evolution of the host genome. Over the years these studies made him a renowned expert on mouse endogenous retroviruses. The similarity of XMRV with murine leukemia virus (MuLV) suggested its origin as the endogenous virus in mice, and "xenotropic" indicated that it can infect other species, such as humans, but not those of its natural host.
The straightforward connection between XMRV and human disease came much sooner than John expected. In 2009, a study published in Science reported that XMRV was detected in the blood of patients with a mysterious disease, Chronic Fatigue Syndrome (CFS), but not in most healthy donors. Bob Silverman was one of the authors, and another was Francis Ruscetti, a viral immunologist in NCI Frederick. The corresponding author, however, was a new name in the field of viral infection, Judy Mikovits.
In the long history of medicine there are many diseases which, like ghosts, haunted people but no one could find their origin. Chronic Fatigue Syndrome (CFS) is one such ghost. Following ordinary minor physical or mental activity, the patients suddenly have lengthy exacerbations or flare-ups of sickness. Their capacity to accomplish tasks is greatly diminished, and constantly troubled by unknown pain. These patients become dysfunctional, either physically or mentally, and their daily life is just a mess.
Now suddenly a new virus was connected to this mystical disease and to prostate cancer. It was much more than that, actually. Mikovits' report found that XMRV was also detected in the blood of some healthy donors. If this was true, donated blood had been contaminated with this disease-causing virus that no one was aware of. This news shocked everyone like those days when HIV was identified. This paper sparked a wildfire of enthusiasm for finding XMRV in more different diseases. NIH even called for a national meeting to discuss the new blood screen policy.
If all were confirmed, XMRV would be the fifth retrovirus that could cause disease in humans, and the virology textbooks needed to be updated. Moreover, its similarity with murine retrovirus suggested that we should be able to find its origin in the mouse genome. It might give important implication in the evolution of disease-causing retroviruses, John thought. He asked his graduate student Oya Cingöz to take a look. It is good practice for a graduate student, John thought.
Not before long, the disease-related XMRV studies became controversial. Initially, several attempts, especially those from Europe, failed to reproduce Mikovits’ finding in the blood of CFS patients. In contrast, a few studies had confirmed the presence of XMRV in the genome of human prostate carcinoma. Later, it was shown that XMRV replication could be stimulated by androgen, which implied that its preference for prostate tissue over blood cells. Moreover, in July of 2010, two publications from three U.S. government laboratories contradicted each other. A study by NIH and FDA groups, led by Dr. Harvey Alter (2020 Nobel Laureate), found MuLV-related virus gene sequences in blood of CFS patients, but not in healthy donors. This result showed the existence of a mixture of related viruses in CFS samples, but it was enthusiastically, but incorrectly, interpreted to directly confirm Mikovits’ finding. On the other hand, the results from a similar study led by William Switzer at CDC were virus negative [2]. What made the whole controversy more complicated was that a small group of CFS activists, who firmly believed XMRV as the long-awaited cause of disease, were so furious about the negative results. In UK, one of the authors of a paper even received death threats from these zealots. Mikovits fought back the negative results by blaming their PCR methods, which did not help to clarify the situation.
In John's laboratory, XMRV was still a scientific question rather than a public health controversy. Oya searched the mouse genome patiently (by designing a highly-specific PCR that was stringent enough to amplify the XMRV sequence, but not the other endogenous MLVs). She took a lot of time to check many strains- almost 100- but finally did find her target. Precisely speaking, she dug out a DNA fossil. The genome of two particular mouse strains had a unique "provirus"- the genetic sequence of retrovirus that had infected mice but lost its infectivity, usually through mutations in the critical viral genes, and stayed in the host genome a long, long time ago. The provirus sequence, Oya found, was 90% identical to XMRV on average, and several mutations made it unable to become a virus again. Some virologists may shrug off the discovery. It is just not a surprise that a related type of virus left a trace in the host genome. However, the more details that she checked, the more it puzzled her. Half of the sequence was nearly identical to the XMRV, and it contained functional viral genes. However, where was the other half of the sequence to make the XMRV? She wondered how this project might proceed.
One day John called a friend, Dr. Vinay Pathak, at the National Cancer Institute, Frederick, Maryland. Vinay is also a virologist but focused more on the basic aspects of retrovirus replication. As Vinay told John, Silverman's discovery of XMRV in a prostate cancer cell line intrigued him very much. He mentioned these cells to his wife, Dr. Wei-Shau Hu, who is also his colleague on the same floor. By coincidence, Wei-Shau's brother-in-law, Dr. Hsing-Jien Kung, a renowned prostate cancer researcher, had acquired these cells lines and xenografts and used them for his study for many years. She connected Vinay with Hsing-Jien, so the latter sent the former the DNA from the cell lines and the tumors from which they were derived to. These materials were generated through a complicated process. When the biopsy of the tumor was taken from the patient, its pieces were implanted into a few mice lacking normal immune function (immunocompromised "nude" mice), so they could grow without being rejected by mouse immunity. As the tumor grows, it will recruit mouse cells to form blood vessels and supporting structures and thus become a mixture of human and mouse cells. When reaching a certain size, to keep it alive, the tumor will be resected and transplanted to a few new mice. Such xenotransplantation is called one passage between mice. The tumors can also be cultured in Petri dishes instead of being transplanted in mice. In that case, the supporting cells will die out eventually, and only cancer cells can survive to form a "cell line".
Vinay's first question was how these cell lines got infected. Every mammalian cell has a defense system against viral infection. One of its components is APOBEC, an enzyme that extensively mutates viral DNA. It exists at high level in blood cells where XMRV was detected. How could it escape from the enzyme? Vinay asked his postdoctoral fellow, Tobias Paprotka, to take a look. Tobias found XMRV can only proliferate in cells with low APOBEC expression, like prostate and other cancer cell lines, including those requested from Silverman. In contrast, blood cells were very resistant to XMRV. Even when it could proliferate in some blood cells, its genome was badly damaged by APOBEC. The short answer to Vinay’s question is that XMRV was not capable of infecting the blood cells. Both Vinay and Tobias were alarmed that something could be wrong in the report of detecting XMRV in the blood of CFS patients.
At this moment, Mikovits was caught in the middle of a storm. More and more studies failed to reproduce her results. However, she got support from a small group of highly militant CFS activists, who believed an absurd, but effective, conspiracy theory: sinister forces hid the fact that XMRV was killing the population. They even sent death threats to those scientists who could not detect XMRV in patients' blood. The Whittemore Peterson Institute, which was founded by a wealthy family frustrated by CFS and hired Mikovits as its first Director, could not wait to offer diagnostic tests for “XMRV status”. Meanwhile, with the concern of XMRV contamination in blood products, NIH called for a large-scale study to address reproducibility of XMRV in CFS samples. Mikovits seemed to enjoy the spotlight of the news media very much during the chaotic year.
Vinay and his staff quietly moved the test forward. Could XMRV first infect the prostate cancer cell in Silverman’s laboratory by accident, and then contaminate the blood samples when the cell lines were sent to Mikovits’ laboratory? Vinay decided to trace the source of XMRV in the prostate tumors and cancer cell lines that he requested from Hsing-Jien. He received six tumors from the previous four passages and two cell lines made from tumors of the most recent passage. If XMRV was detected only in tumors from recent passages, not in earlier ones, it might indicate laboratory contamination.
Tobias used PCR to detect an XMRV gene (env) in the cells. It was detected at a very high level in the two cell lines and tumors of later passages, as it should be. However, a very low level of env was also present in the tumors of earlier passage, so Vinay and Tobias could not say that XMRV was simply an exogenous contamination. PCR results from the tumor showed that the env gene was from a fragment of XMRV DNA. Vinay and Tobias scratch their heads wondering what that meant. Vinay recalled what John told him about Oya's finding… could this come from the small number of mouse cells mixed in the tumors of earlier passages? He asked Tobias to check the abundance of mouse cells in the earlier-passage tumors. It was an amazing match of the level of env genes in the tumors: 1%. They had to keep tracking, so Tobias and his lab mate Krista Frankenberry went on to detect the proviruses in genomes of dozens of mouse strains by PCR. They found a provirus similar to XMRV in a few strains. Like Oya's finding, some mutations disabled the viral function, but half of the sequence, which contained another set of functional viral genes, was nearly identical to XMRV. Vinay thought he should talk to John.
Almost as soon as he hung up the phone call with Vinay, John flew to DC from Boston carrying Oya's data with him. They sat together, pulling out the data from their files, and got totally stunned. The two partial XMRV-identical sequences, found by these two teams independently, were the two complementary parts of XMRV, bringing all the required genes to make a virus together. John recalled:
"I will never forget the day Vinay and I sat down and compared the two sequences. They snapped together like two pieces of a puzzle to make XMRV exactly. A remarkable moment."
They named the two proviruses found by Vinay’s and John’s groups XMRV-1 and -2, respectively. When putting the mouse genome sequencing data from two groups together, among 94 strains of mice they screened, only three have both XMRV-1 and -2 sequences in their genomes. Two of them are the nude mice- the immunodeficient mice used for growing human prostate tumor tissue in Silvermann’s laboratory. The two parts of torn maps had now been one piece, and it pointed out where the treasure was- XMRV must have been created during the tumor passages in nude mice!
Like the detectives who rebuild murder scenes, Vinay and John could deduct how this scenario had unfolded. XMRV-1 and XMRV-2 are the relic sequences left in the mouse genome by two related but different retroviruses. By chance, cells of these mice synthesized the RNA genomes of preXMRV-1 and preXMRV-2 and packed them into the same viral particle (virion). When such heterozygous virus infects another cell, then reverse transcriptase, in the course of making proviral DNA, crosses back and forth from one genome RNA to the other- the outcome of this process is so-called "recombination". In some cases, all the functional genes required for making a virus was put together, and the XMRV was born (Figure 1). Rreading this part in the paper always brought the "Dawn of Men" scene of the movie "2001: the Space Odyssey" in my brain".
However, if this is true, why was XMRV was never found in mouse cells? The two teams recollected a well-known fact concerning the virus-binding receptors in mice and humans. Interestingly, the mouse receptor has a few amino acids difference from the human counterpart, making it unable to bind to XMRV. That is, even if XMRV forms in mouse cells, it could not infect other mouse cells. The human cells are required for the dissemination of XMRV. In other words, XMRV is an artifact produced from the experimental setting: xenograft of human cells in mice strains that harbors preXMRV-1 and -2 (Figure 2). The XMRV detected in the blood of CFS patients can only be from one cause: laboratory contamination.
Vinay and John submitted their manuscript to Science, and it was published in May 31, 2011 [3]. The title had only six words but read like a verdict : “Recombinant Origin of the Retrovirus XMRV”. The controversy was all over. Vinay’s and John’s teams put the last nail in the coffin of XMRV as the cause of CFS or any other disease. In the following few years, many XMRV-related papers were retracted, some of them just for avoiding further controversy, including Dr. Alter’s paper in PNAS (even though it did not validate Mikovits’ result directly).
While the manuscript was in the review process, two large-scale studies including more CFS patients were underway, too. All of them gave negative results, and Mikovits was a co-author in both reports when published. Although she made many mistakes, these negative results may not necessarily to be a total failure. The whole XMRV saga still has biological implication and lessons for biomedical scientists, including how virological testing should be performed to avoid contamination and bias, and the danger of over-promise. Many CSF patients steadily believed that Mikovits had found them the cause and a cure was coming- some still do even today. However, she did not end the saga in a meaningful manner. After being revealed that she doped XMRV DNA into patient blood samples [4], Mikovits has turned herself into a champion in medical conspiracy theories, basing claims linking the XMRV to autism and cancer on those retracted papers, and claiming she had been the victim of the influence of the deep state and Big Pharma. Since the pandemic started, she gained lots of attention from social media for her anti-vaccine, anti-mask rhetoric.
However, this story should not be focused on a conspiracy theorist. This story is about a group of scientists who were interested in truth and worked relentlessly like a team of detectives. They took the case because of scientific curiosity and a sense of duty to apply their expertise to a potential public health crisis, rather than the motivation for getting a glorious publication. They paid attention to the tiniest clues and looked for suspects based on evidence, not toward the one attached with the biggest bounty. More importantly, they have shown us the meaning- and the fun- of doing scientific research.
Epilogue
Two years after the publication of XMRV study from John’s and Vinay’s teams, a graduate student found an additional xenotropic MLV in the human prostate cancer cell line (LNCaP) xenografted in nude mice. It looked like that other fancy RNA viruses could be born out of the path of XMRV generation. Moreover, she showed that the viral infection promoted the growth of the xenografted tumors in mice. This sent an alarm to all the cancer xenograft experiments: XMRV was not an isolated incident. RNA viruses could arise frequently and spread through different species when they get contact with each other. Retrospectively, this study not only showed the risk of unintended contamination that altered experiment results, but also a far cry to people of what was coming in 2020.
The student’s name is Meera Murgai [5]. She is my colleague now.
Acknowledgment
I want to give special thanks to Drs. John Coffin and Oya Cingöz for their wonderful help in fact check and text editing.
Reference
- https://journals.asm.org/doi/10.1128/MMBR.00033-07
- https://www.science.org/doi/full/10.1126/science.329.5995.1000
- https://www.science.org/doi/full/10.1126/science.1205292
- https://www.liebertpub.com/doi/10.1089/aid.2020.0095
- https://retrovirology.biomedcentral.com/articles/10.1186/1742-4690-10-34
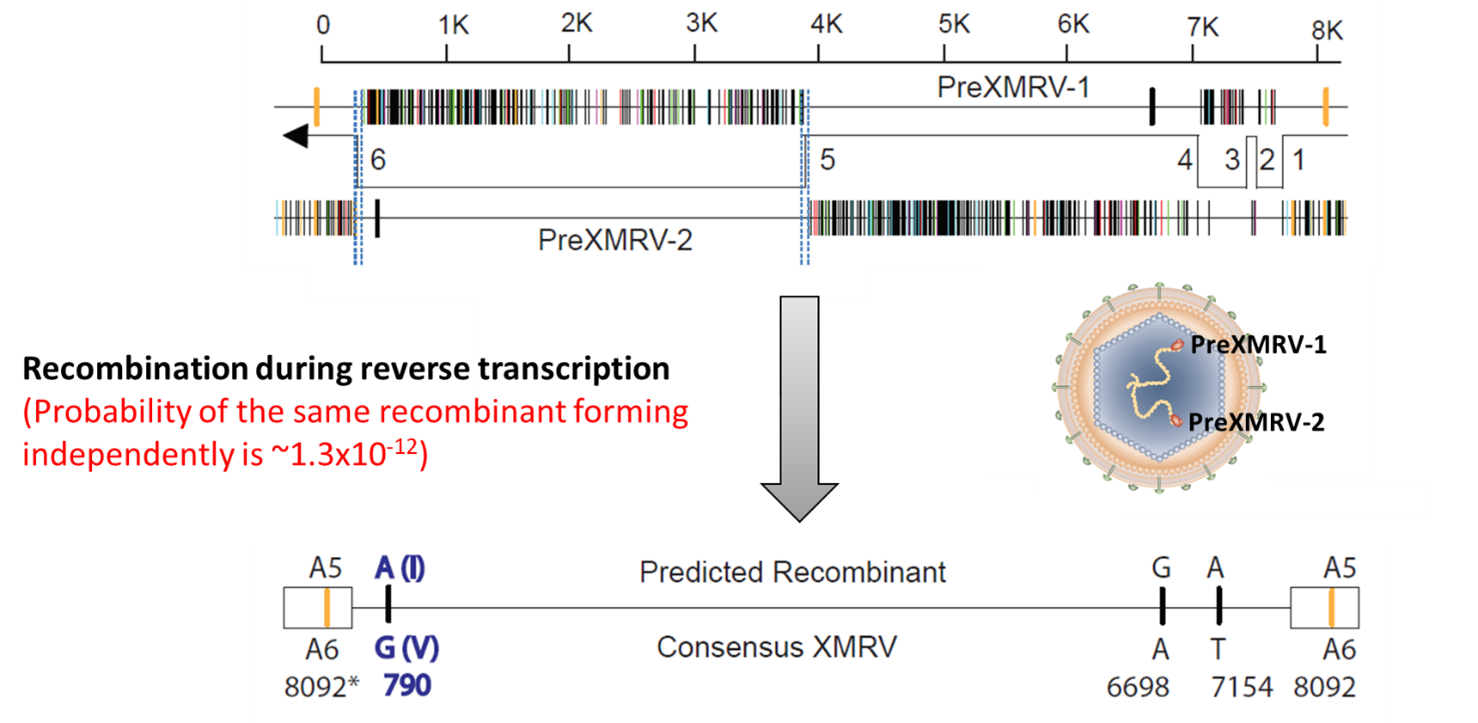
Figure 1. The hypothetical recombination event that generates XMRV. Top panel, the sequence diagrams of preXMRV-1 and preXMRV-2. The short vertical lines mark the bases different from those in XMRV. Therefore, each of the preXMRVs exhibits about 50% of sequence identical to XMRV. The recombination sites are marked by the “elbows” of the arrow. Middle panel, the viral particle formed in the mouse cells packs the RNA genomes of preXMRV-1 and -2 together, followed by the recombination event. Bottom panel, the recombination product that generates XMRV.
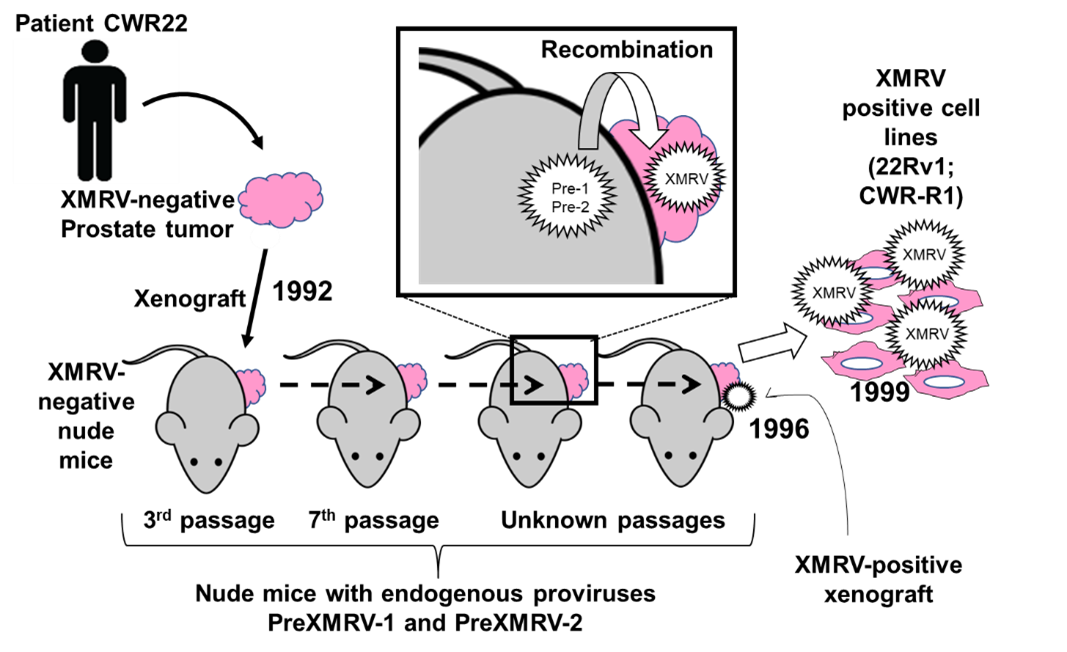
Figure 2. The putative process of the formation of XMRV in the mouse strains and its infection of the prostate cancer cell lines during the passage of xenografts.
Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in