Water oxidation on a mononuclear manganese heterogeneous catalyst
Published in Chemistry
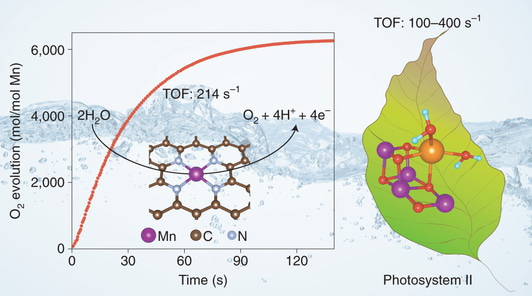
When Professor Can Li asked me this question and assigned me to take this project four years ago, firstly I studied the previous literature and found that the turnover frequency (TOF) of synthetic Mn-based water oxidation catalysts is between 1*10-5 and 6*10-4 s-1, which is at least five orders of magnitude lower than that of PSII. I thought that it was almost impossible to fabricate a Mn-based catalyst with water oxidation activity comparable to that of PSII, considering that Mn shows the lowest activity in electrochemical oxygen evolution reaction among Mn, Fe, Co, Ni, and Cu.
I tried some polyoxometalate compounds, but found that they were inactive using diammonium cerium nitrate as the oxidant. Therefore, I turned to synthesize heterogeneous catalysts, such as CoOx, MnOx, RuOx, and IrOx. I found that the TOF increases with decreasing the particle size of metal oxides, and was pleasantly surprised to find that subnanometric iridium oxide showed water oxidation activity comparable to that of Ir-based molecular catalysts. It stimulated me to explore the synthesis of nanoclusters with few atoms and even single atom. However, the controlled synthesis of heterogeneous cluster catalysts with regular metal sites is very difficult. As far as I know, it is still a technically challenging issue to synthesize heterogeneous cluster catalysts with regular active sites up to now.
It was surprised to find that all the Fe-, Co-, Mn-, Ru-, and Ir-based N-doped graphene materials showed very high water oxidation activity. However, after I further checked the materials carefully, I found that the high water oxidation activity came from the residual Mn in the synthesis of graphene oxide (GO) by using potassium permanganate as the oxidant. After carefully removing the residual Mn in the GO precursor, the water oxidation activity decreased or even down to zero. Therefore, it is confirmed that the Mn should be responsible for water oxidation and then we speculated that the single-site Mn should be highly active. However, many people disagreed with the conclusion because it is believed that the water oxidation reaction needs multiple reaction sites as that in Mn4CaO5 cluster in PSII.
So we began a long journey to verify if the mononuclear Mn is the active site. We found that the mononuclear Mn coordinated with O in the graphene cannot chemically catalyze water oxidation reaction, but Mn coordinated with four N atoms in the graphene can efficiently catalyze water oxidation reaction by chemical and electrochemical manners. By DFT calculations, we found that the MnN4-G site is much more active than other Mn-based catalyst, mainly owing to the four N coordination to Mn in favour of the formation of MnIV-oxo species and the further nucleophilic attack of the second water molecule to MnIV-oxo to evolve O2 with low activation energy via the MnIII-OOH intermediate.
The corresponding paper in Nature Catalysis is here: https://www.nature.com/articles/s41929-018-0158-6
Jingqi Guan Associate Professor of Chemistry, Jilin University
Jingqi Guan worked as a postdoctoral researcher in Prof. Can Li’s group in the past four years and studied the water oxidation reaction, and currently works at Jilin University and further investigates electrocatalytic water oxidation.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in