When a mite is your best friend — but only sometimes
Published in Ecology & Evolution and Zoology & Veterinary Science

Why does this question matter?
Nature rarely deals in absolutes. A relationship that looks like cooperation from one angle can look like exploitation from another, and whether a symbiont is friend or foe often hinges on who else is in the room. This deceptively simple idea, that the net effect of one organism on another is shaped by the presence of a third, sits at the heart of ecological theory, yet empirical tests of it in real, multi-species systems remain surprisingly scarce (Rogalski et al. 2021; Sun & Kilner 2020).
We study burying beetles (Nicrophorus nepalensis), a charismatic insect that buries the carcasses of small vertebrates and raises their young on them, displaying a level of parental care unusual for insects. But they don't do this alone. Hitchhiking on their bodies are two uninvited guests: phoretic mites (Poecilochirus carabi), clinging to the beetles' cuticles, and parasitic nematodes (Rhomborhabditis regina), hidden deep within the beetles' genitals and intestines (Fig 1). Our question was simple to ask but fiendishly difficult to answer: do these three-way interactions shift the mites from parasites to mutualists, or back again, depending on the ecological context?

The black box problem: building an experiment from scratch
Here is something that textbooks don't tell you: knowing that an association exists is very different from being able to study it experimentally. Mite-beetle associations had been documented for decades (Pukowski 1933). Nematode-beetle associations had been noted too (Wang & Rozen 2019). But empirically dissecting the three-way interaction among mites, nematodes, and bacteria simultaneously in a controlled breeding setting had essentially never been done. We were stepping into a black box (Fig 2).
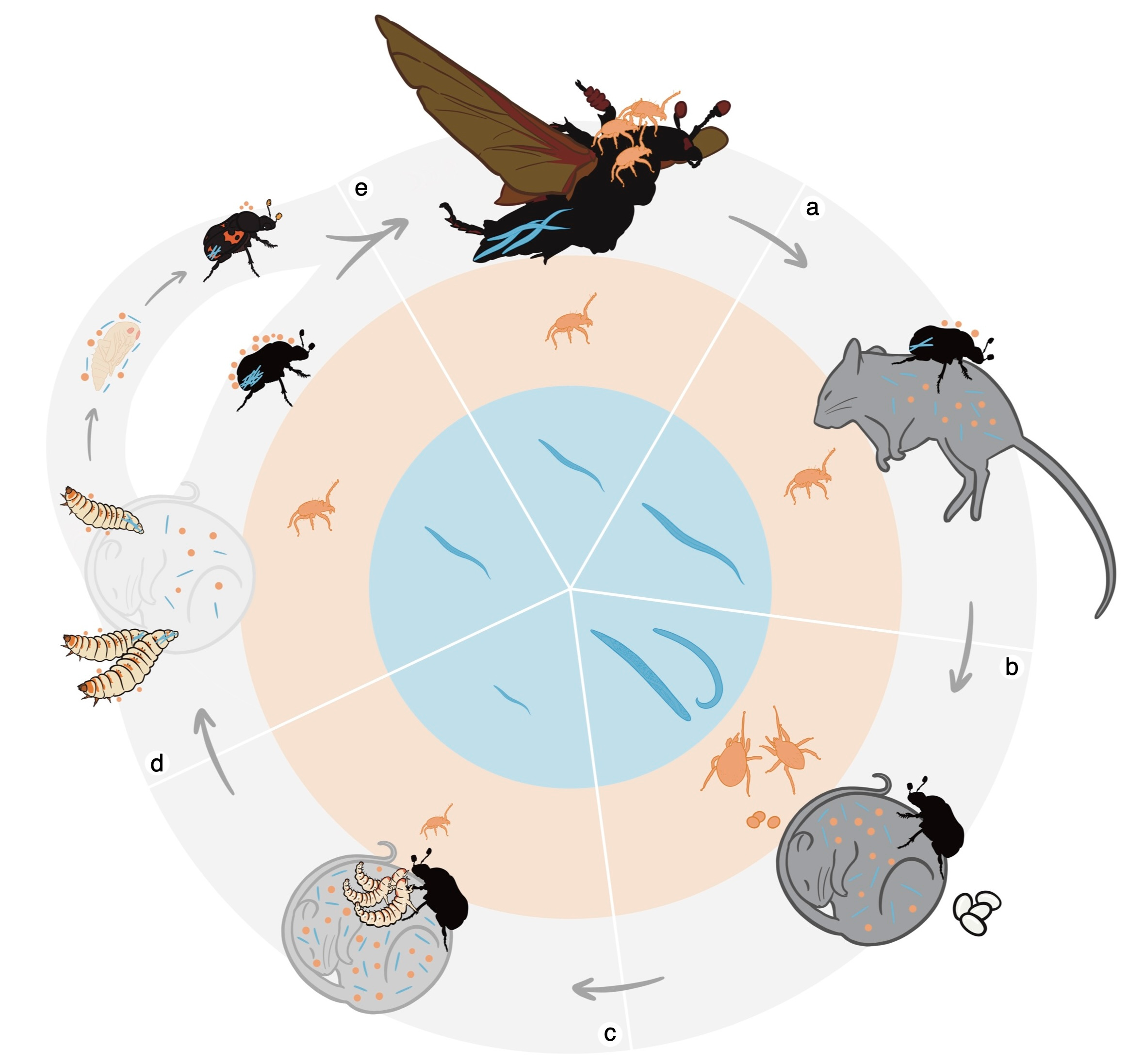
What followed was months of trial and error. How many mites per beetle is ecologically realistic? How do you standardise nematode inoculation doses across hundreds of breeding trials? Every methodological decision cascaded into new complications, and the system had no established playbook. Looking back, we think of those early months as the hidden cost of working on a genuinely understudied system: the price of novelty is uncertainty, and uncertainty is expensive in time.
Perhaps the most formidable challenge was creating a truly nematode-free beetle colony. In wild populations, essentially almost all beetles carry nematodes; they are transmitted both from parent to offspring on the carcass, and vertically through the cuticle during pupation and adult emergence. To establish a clean baseline, we had to surface-sterilise beetle eggs using an antimicrobial solution. This was a painstaking process that needed to be done egg by egg, under a dissecting microscope, at precisely the right developmental window. Even then, the work wasn't done: because the parent beetles themselves harbour nematodes, we had to raise the first generation of larvae without parental care, allowing them to feed independently on the prepared carcass to prevent re-contamination of our sterilised cohort. Anyone who has worked with burying beetles knows that separating larvae from their attentive parents goes against everything about their biology (Jarrett et al. 2018). Getting those larvae to survive and thrive without parental provisioning required yet more troubleshooting. It is the kind of challenge that never makes it into the methods section, but that quietly defines whether a project lives or dies.
What we found — and what surprised us
Once the system was running, the results were worth the effort. We manipulated nematode densities (zero, low, and high) and mite presence across hundreds of beetle breeding trials, and the pattern that emerged was striking in its context-dependence.
At high nematode densities, beetle offspring survival dropped sharply (from around 96% down to 78%) in the absence of mites. But introduce mites, and that survival penalty largely disappeared. The mites were actively preying on nematodes (we confirmed this in separate feeding assays), suppressing their transmission to beetle offspring, and in doing so, rescuing host fitness. A classic case of enemy-of-my-enemy-is-my-friend.
But here is the twist: remove the nematodes entirely, and the mites become the problem. Without nematodes to prey on, mite presence was itself associated with reduced offspring survival. The very same organism was mutualist in one context and parasite in another, with the tipping point determined by nematode density.
We also discovered something unexpected about the carcass microbiome. Both mites and nematodes, individually, reduced culturable bacterial loads, but it was their combined presence that produced the most dramatic shift in bacterial community composition. The carrion environment was being restructured by the interplay of all three partners simultaneously, suggesting that the microbial dimension of this system is both important and largely unexplored.
What this means — and where we go next
The broader implication of our work is that phoretic associations — long assumed to be essentially commensal, a free ride with no strings attached — may routinely act as hidden drivers of host fitness through indirect, density-dependent pathways. The mites are not just passengers; they are active participants in a three-way negotiation whose outcome depends on who else is at the table.
For future research, we are eager to understand the microbiome side more deeply. Which bacterial taxa are being suppressed by mites and nematodes, and does this translate into measurable differences in larval nutrition and immune function?
Science, like symbiosis, is rarely a simple story of friend or foe. The most interesting answers tend to live in the space between: contingent, context-dependent, and always shaped by who else is in the room.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
DNA repair and human disease
Publishing Model: Hybrid
Deadline: Oct 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in