Why animals (and predatory bacteria) outsource costly amino acids
Published in Ecology & Evolution, Microbiology, and Genetics & Genomics

The origin of this study was unexpectedly simple: preparing a lecture for first-year medical students.
While assembling material for a basic molecular biology and genetics course, we came to the familiar topic of essential versus non-essential amino acids. Students are taught that nine of the twenty amino acids are essential and must be obtained from the diet because the body cannot synthesize them. But when we tried to answer the inevitable question — why these particular nine? — we realized we did not have a satisfying explanation.
Textbooks typically present the list of essential amino acids as a fact to memorize. Scientific literature discusses the underlying biosynthetic pathways, but rarely addresses the evolutionary logic behind their loss in animals. The absence of a clear, general explanation was striking.
Moreover, the very terms “essential” and “non-essential,” coined in the context of animal nutrition, are somewhat misleading. All twenty proteinogenic amino acids are indispensable for cellular life, and even those classified as non-essential may require dietary supplementation to support optimal growth and physiology.
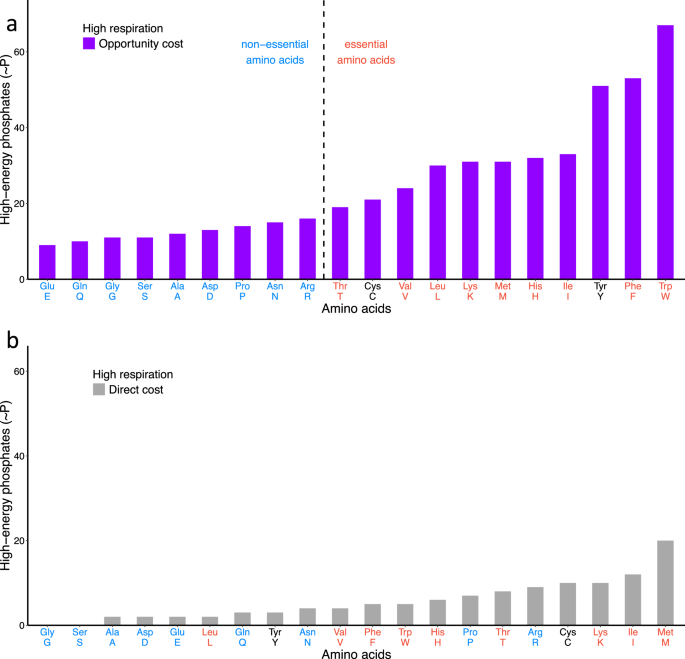
One obvious difference among amino acids is how expensive they are to synthesize. Some require only a few biochemical steps; others demand long pathways consuming large amounts of cellular energy.
We wondered what would happen if amino acids were simply ranked by biosynthetic cost.
The result was startling. They separated into two groups that almost perfectly matched the biological classification: expensive amino acids were overwhelmingly essential, while cheaper ones were typically non-essential. Could natural selection have driven this?
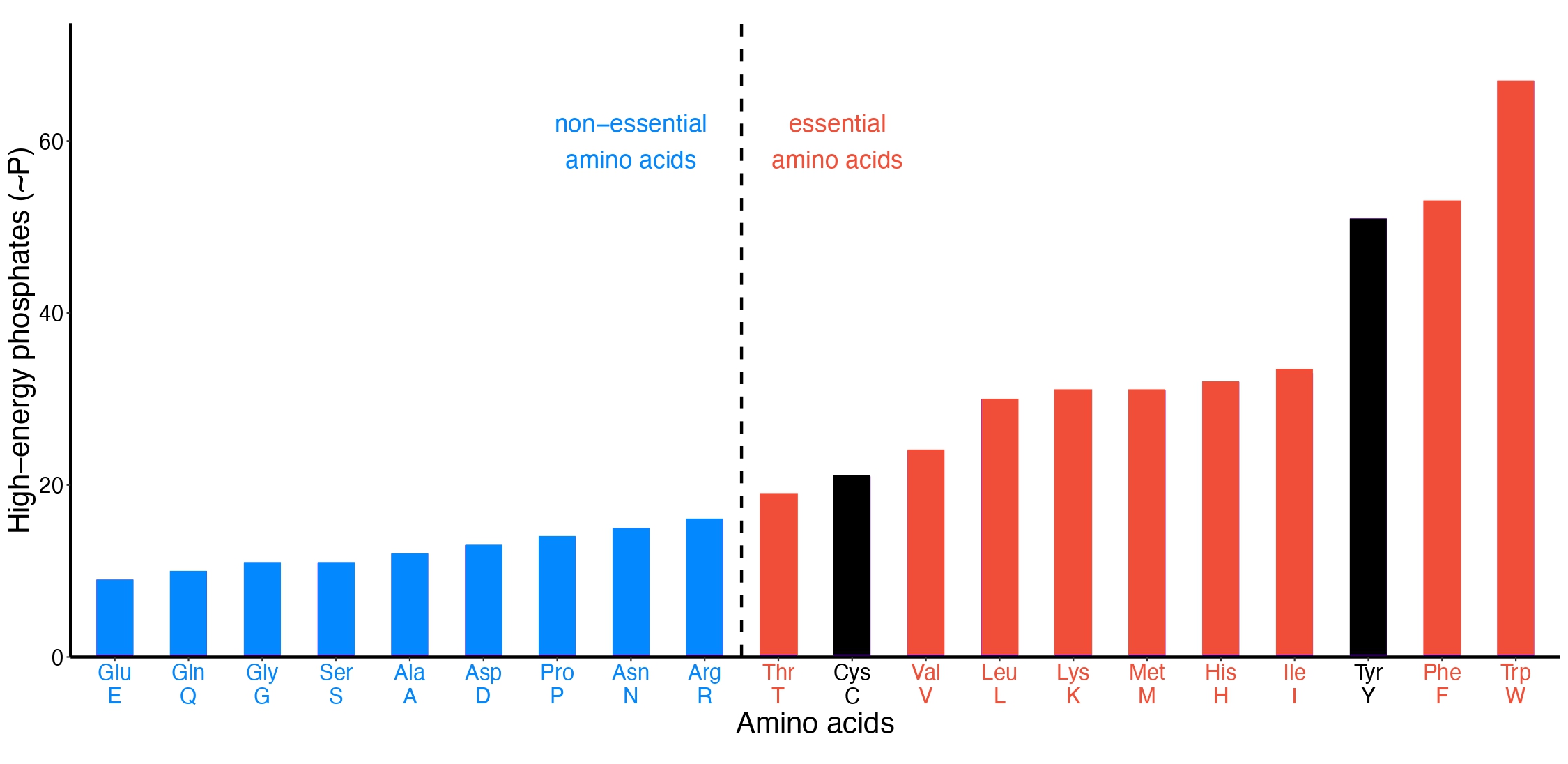
This observation suggested a simple but powerful idea: animals may have selectively lost the ability to synthesize the most energetically costly amino acids because it was advantageous to obtain them from the environment instead.
Conceptually, this builds on ideas we have explored previously by borrowing from economics. In earlier work, we framed the loss of biosynthetic pathways as functional outsourcing, where organisms rely on external sources instead of maintaining costly internal production.
In the present study, we extend this perspective using the notion of opportunity cost: resources invested in synthesizing expensive amino acids cannot be used for other processes, so losing these pathways can free energy for alternative evolutionary innovations.
From a simple idea to a full-scale project
What seemed like a straightforward question quickly grew into a collaborative effort. With Niko joining the group as a doctoral student and Mirjana contributing her computational expertise — and bringing valuable perspective through familiarity with analogous concepts in economics — the three of us set out to test whether this pattern could be explained by evolutionary principles.
Initially, we expected a relatively simple project: compile data on amino acid essentiality and energetic costs, perform a few statistical analyses, and produce a concise explanation. Instead, we encountered a series of challenges.
Existing estimates of biosynthetic costs were limited or outdated, so we expanded them — ultimately incorporating multiple complementary measures, including links between biosynthesis and respiration. Data on amino acid auxotrophies in animals were scattered, which led us to construct a comprehensive enzyme database and systematically infer auxotrophies across more than a hundred animal and related genomes.
As we progressed, new questions emerged. What happens to protein composition after organisms lose the ability to synthesize certain amino acids? Do proteomes adapt in predictable ways? Are there broader macroevolutionary patterns shaped by these constraints?
Answering these questions required increasingly sophisticated computational approaches. What began as a simple visualization evolved into large-scale analyses of proteomes, supported by newly developed algorithms and pipelines.
Developing a new test: CPS
The central challenge remained: how to demonstrate that energy-related selection actually drives the observed pattern.
Standard evolutionary methods were not designed for this. They typically focus on individual genes or sites, whereas our hypothesis concerned a global phenotype — the overall configuration of amino acid biosynthetic capabilities and their usage across proteomes.
To address this, Niko — inspired by our recent study on the macroevolution of the N-glycosylation pathway — developed a new framework: the combinatorial phenotype selection (CPS) test. Rather than analyzing traits in isolation, CPS evaluates whether a complete configuration is unusually well aligned with a particular selective pressure — in this case, minimizing biosynthetic energy costs.
Using this approach, we found strong evidence that energy optimization shaped the loss of amino acid biosynthesis in the lineage leading to animals.
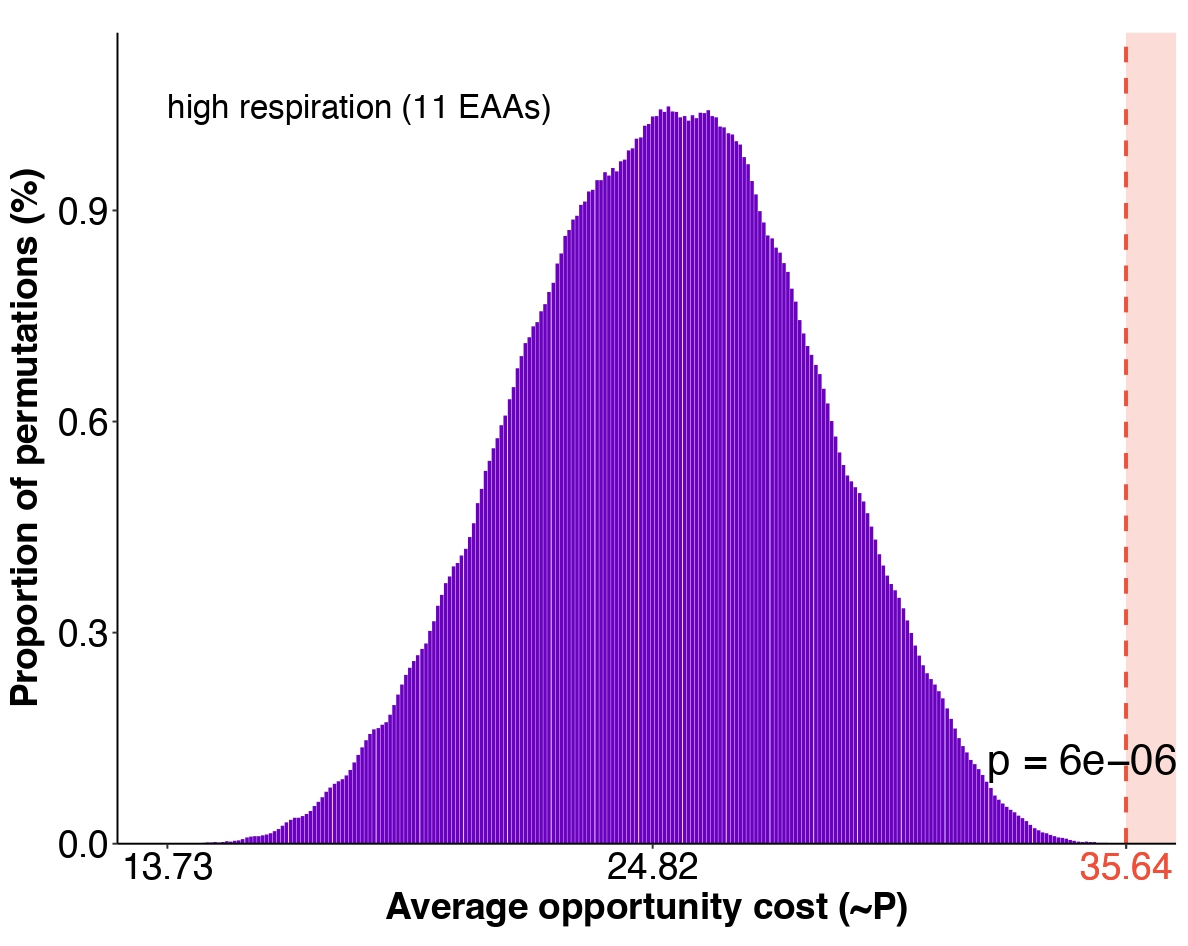
(CPS test, Kasalo et al. Nat Commun 2026)
A paradox: outsourcing enables costly proteins
An interesting consequence emerged from these analyses. By outsourcing the most expensive amino acids, organisms reduce their metabolic burden — but this also relaxes constraints on protein composition.
In animals, this appears to have enabled increased use of energetically costly amino acids in proteins. In our related work on bacteria, we observed a similar phenomenon: amino-acid auxotrophies can function as “costly outsourced traits,” allowing organisms to explore protein sequence space more freely and potentially evolve new functionalities.
In this sense, losing metabolic capabilities may actually facilitate molecular innovation.
Convergence in predatory bacteria
A natural next step was to ask whether animals are unique in this respect.
If the pattern is driven by selection, similar metabolic strategies should evolve independently under comparable ecological conditions.
We identified an ideal system in predatory bacteria of the phylum Bdellovibrionota. These organisms invade and consume other bacteria, effectively living in amino-acid-rich environments — a situation that parallels key aspects of animal ecology.
Applying the same analyses revealed a striking convergence: these bacteria have lost the ability to synthesize many of the same energetically costly amino acids as animals.
The ecological contrast further strengthened this conclusion. Obligate predators, which consistently rely on prey for nutrients, exhibit extensive amino acid auxotrophies. Closely related facultative predators, such as Myxococcota, retain far more biosynthetic pathways.
Together, these findings point to a general principle: metabolic outsourcing is shaped by ecological context and energy economics.
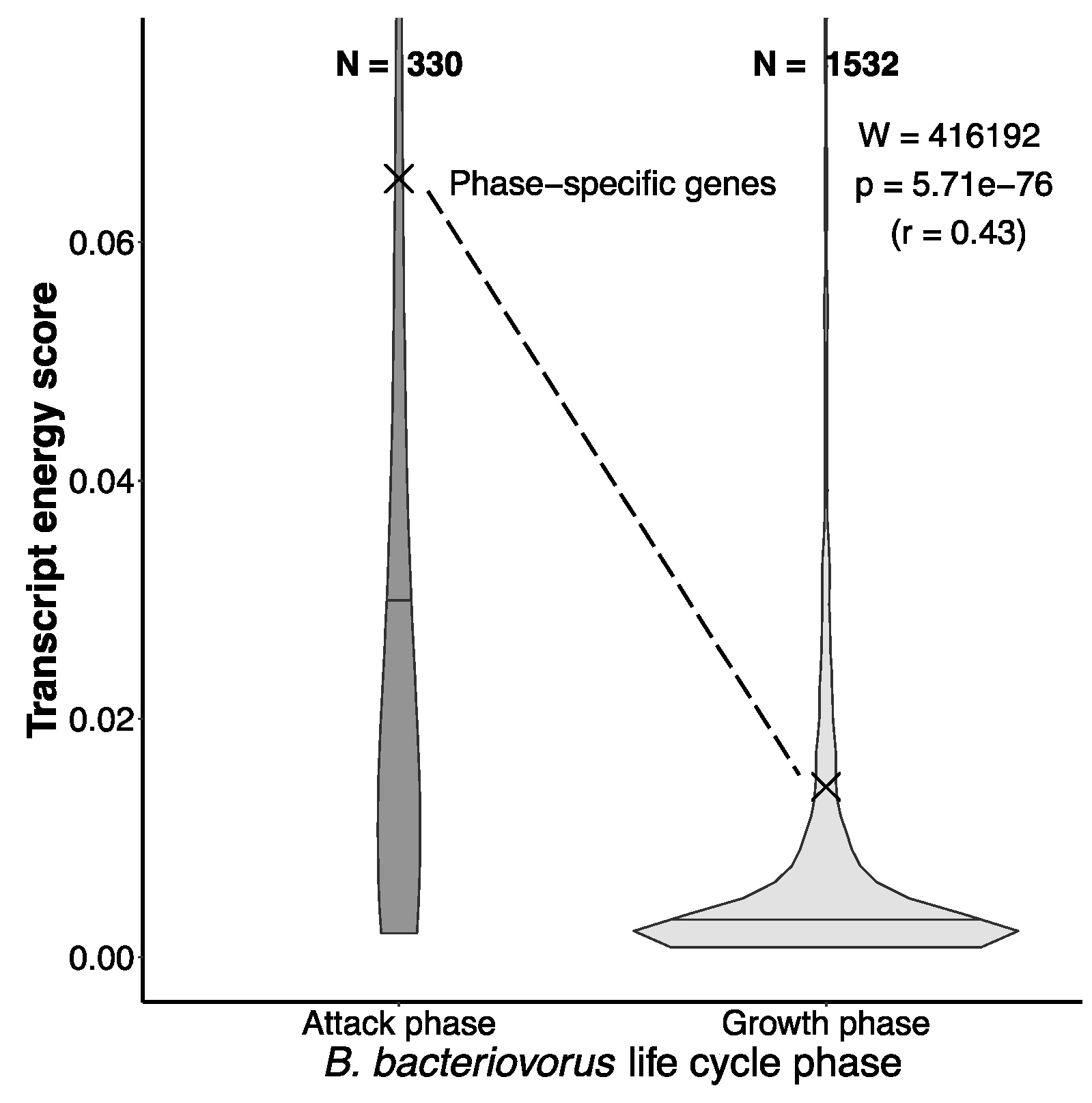
(Kasalo et al. IJMS 2025)
A model for animal-type auxotrophy
Bringing these results together, we developed a general model for the evolution of animal-like amino acid dependence.
Two conditions appear to be key. First, high respiration rates: aerobic metabolism provides abundant energy, increasing both the cost of maintaining unnecessary pathways and the benefits of reallocating resources. Second, a reliable external supply of amino acids: predation or nutrient-rich environments ensure that outsourced compounds remain consistently available.
In predatory bacteria, these conditions clearly distinguish obligate from facultative predators. We propose that similar conditions likely applied to early animal ancestors — metabolically active organisms embedded in microbe-rich ecosystems.
This perspective also highlights a broader conceptual shift. From a physiological standpoint, the important distinction is not whether amino acids are “essential” or “non-essential,” but whether they are synthesized internally or obtained externally.
For this reason, it may be more informative to think in terms of prototrophic (internally synthesized) and auxotrophic (externally supplied) amino acids — effectively, whether they are produced in-house or outsourced.
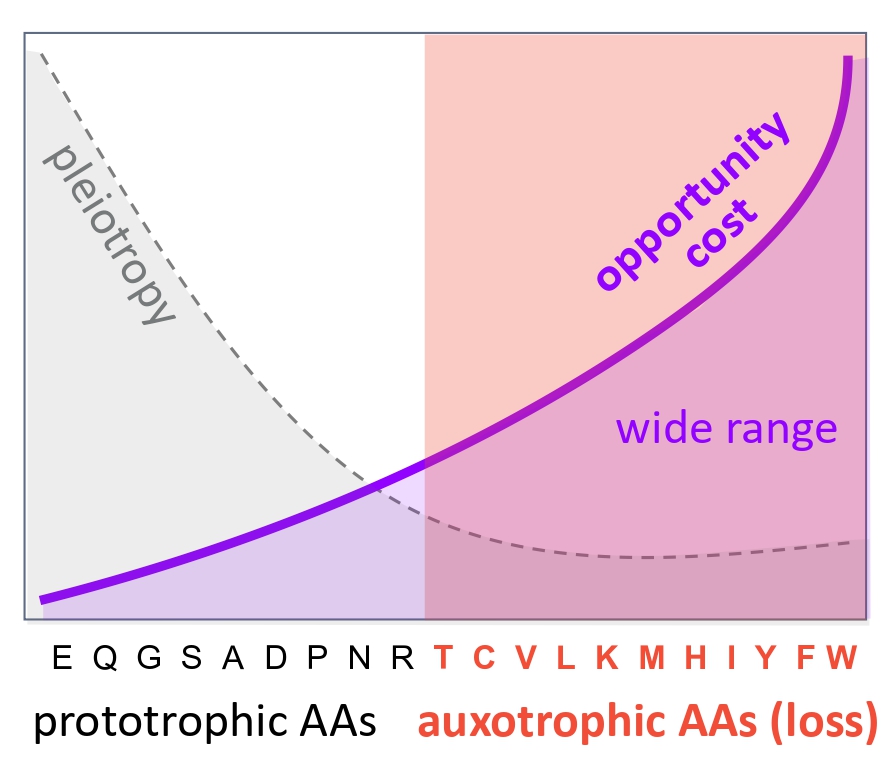
(Kasalo et al. Nat Commun 2026)
From project to research area
What began as a simple question gradually evolved into a broader research direction.
Along the way, it became the central focus of Niko’s doctoral work, while continued collaboration expanded the scope further. More recently, we have started incorporating phylogenetic approaches, reconstructing how the energetic properties of proteomes change through evolutionary time.
This has led us to define a broader framework we call phyloenergetics — integrating energetics, pleiotropy, and evolutionary history across levels ranging from individual amino acids to entire proteomes and lineages.
What started as a simple classroom question has grown into an active area of research, now involving multiple students and projects.
Back to the classroom
Perhaps the most rewarding aspect of this journey is returning to where it began.
An attempt to better explain a basic concept to students revealed a gap in understanding and opened a new line of research. It is a reminder that fundamental questions are often hiding in plain sight.
Now, when students ask why essential amino acids exist, we no longer rely on incomplete explanations. Instead, we can describe an evolutionary story shaped by energy, ecology, and interactions between organisms — and share how that very question led to discovery.
Sometimes, the simplest “why?” can take you the furthest.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
I found this really interesting.
Another commonality between predatory bacteria and animals compared with other bacterial lineages is a reduction in effective population size, where selection is less strong. Could that be part of the explanation?
David Liberles