A positive feedback loop regulation between NOTCH1 and USP11 in T-cell leukemia
Published in Cancer and Cell & Molecular Biology
Explore the Research

A positive feedback loop regulation between NOTCH1 and USP11 in T-cell leukemia - Leukemia
Leukemia - A positive feedback loop regulation between NOTCH1 and USP11 in T-cell leukemia
Activating mutations of NOTCH1 represent the most common genetic cause of T-cell lymphoblastic leukemia (T-ALL) and are detected in more than 50% of patients suffering from the disease1. The affected NOTCH pathway regulates cell fate determination and has been directly implicated in oncogenic transformation. In addition to genetic mutations, transcriptional dysregulation and posttranslational activation have been shown to lead to aberrant NOTCH pathway activity. In an example of the latter, ubiquitin-specific peptidases (USP) inhibit NOTCH1 proteasomal degradation2. USP7 has been implicated in stabilizing NOTCH1 protein complex members and its pharmacological inhibition allowed the blockade of T-ALL cell growth in the absence of high on-target toxicity normally associated with direct NOTCH1 inhibition2. In our current work we further investigated the cross play between the deubiquitination pathway and NOTCH signaling.
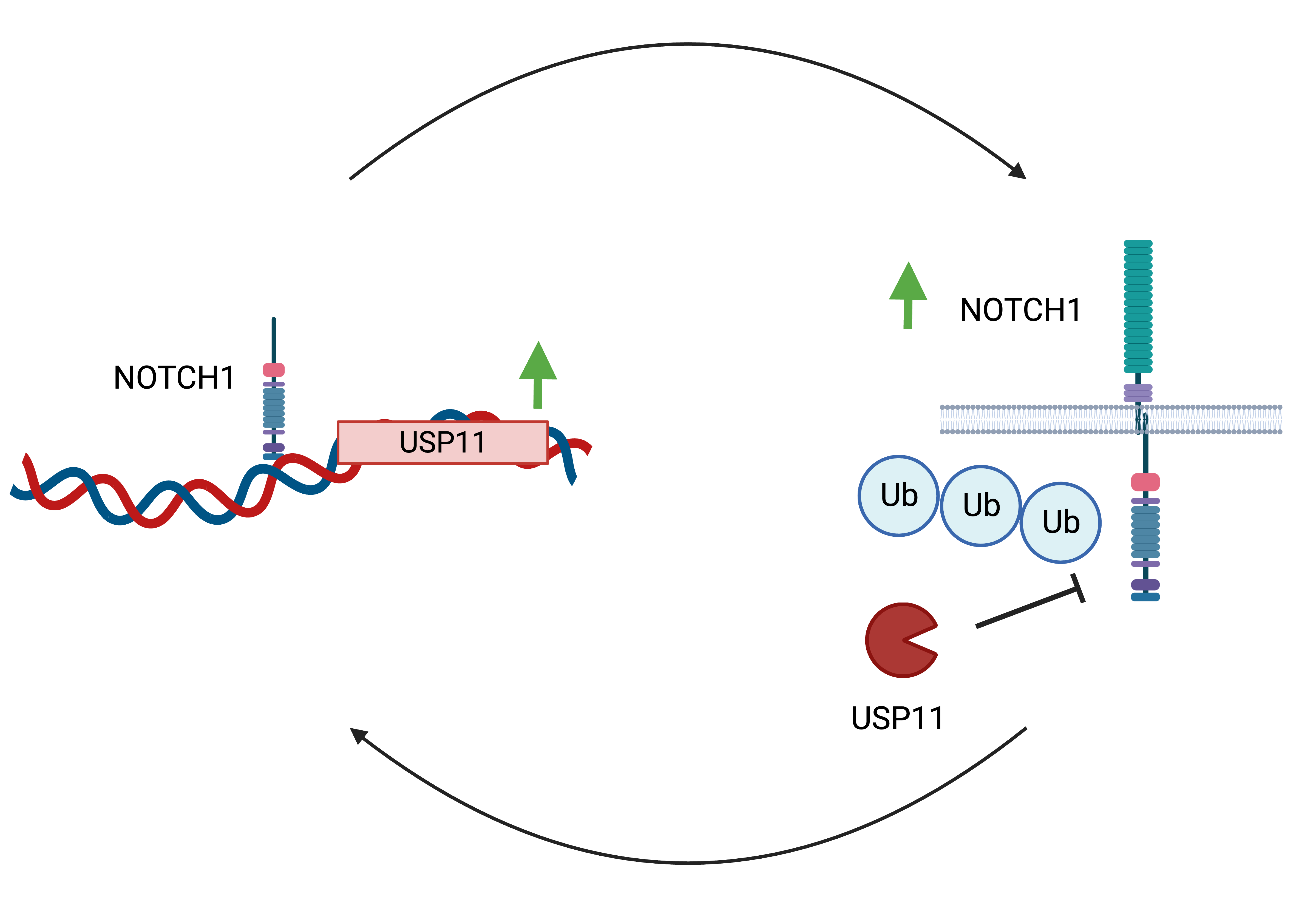
We found that NOTCH1 and USP11 expression positively correlate in T-ALL patients of the pediatric cancer genome project and that USP11 expression has prognostic value3. This correlation was confirmed through reverse-phase protein array in an independent patient cohort. We show that NOTCH1 binds the USP11 locus and transcriptionally regulates USP11 expression based on chromatin immunoprecipitation assays (ChIP-seq). Both USP11 and USP7 expression is also suppressed by NOTCH1-targeting γ-secretase inhibition. We also demonstrate that USP11 interacts with and actively deubiquitinates NOTCH1 stabilizing the protein and protecting it from proteasomal degradation. Short hairpin RNAs-mediated silencing of USP11 led to decreased NOTCH1 expression and inhibited proliferation in T-ALL cell lines. This effect could be rescued by ectopic expression of USP11 but not of catalytically dead USP11 mutant.
Taken together, our results demonstrate a positive feedback loop between NOTCH1 and USP11 in T-ALL. NOTCH1 drives the expression of USP11, while the peptidase actively deubiquitinates the NOTCH1 protein stabilizing it via a posttranslational mechanism. USP11 has been previously implicated in several oncogenic mechanisms both in solid and hematological tumors including protein translation, DNA damage response and apoptosis4-6. Despite this, aberrant deubiquitination remains a relatively understudied mechanism of oncogenesis. The precise role of USP11 and USP7 in NOTCH1 complexes and other sources of transcriptional regulation of these proteins warrant further research. Given other emerging roles of USP11 in therapy resistance, where it controls lymphocyte-specific kinase (LCK) blocking the glucocorticoid response in T-ALL, our findings illustrate yet another manifestation of USP11-mediated oncogenic deubiquitination in cancer3. We present a strong rationale supporting further exploration of the therapeutic potential of USP11 inhibition in T-ALL.
- Ntziachristos P, Lim JS, Sage J, Aifantis I. From fly wings to targeted cancer therapies: a centennial for notch signaling. Cancer Cell 2014 Mar 17; 25(3): 318-334.
- Jin Q, Martinez CA, Arcipowski KM, Zhu Y, Gutierrez-Diaz BT, Wang KK, et al. USP7 cooperates with NOTCH1 to drive the oncogenic transcriptional program in T-cell leukemia. Clinical Cancer Research 2019; 25(1): 222-239.
- Jin Q, Gutierrez Diaz B, Pieters T, Zhou Y, Narang S, Fijalkwoski I, et al. Oncogenic deubiquitination controls tyrosine kinase signaling and therapy response in acute lymphoblastic leukemia. Sci Adv 2022 Dec 9; 8(49): eabq8437.
- Ke JY, Dai CJ, Wu WL, Gao JH, Xia AJ, Liu GP, et al. USP11 regulates p53 stability by deubiquitinating p53. J Zhejiang Univ Sci B 2014 Dec; 15(12): 1032-1038.
- Kapadia B, Nanaji NM, Bhalla K, Bhandary B, Lapidus R, Beheshti A, et al. Fatty Acid Synthase induced S6Kinase facilitates USP11-eIF4B complex formation for sustained oncogenic translation in DLBCL. Nat Commun 2018 Feb 26; 9(1): 829.
- Maertens GN, El Messaoudi-Aubert S, Elderkin S, Hiom K, Peters G. Ubiquitin-specific proteases 7 and 11 modulate Polycomb regulation of the INK4a tumour suppressor. EMBO J 2010 Aug 4; 29(15): 2553-2565.
Follow the Topic
-
Leukemia
This journal publishes high quality, peer reviewed research that covers all aspects of the research and treatment of leukemia and allied diseases. Topics of interest include oncogenes, growth factors, stem cells, leukemia genomics, cell cycle, signal transduction and molecular targets for therapy.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in