A Pre-Pandemic vaccine.
Published in Microbiology
Explore the Research
 sciencedirect.com
sciencedirect.com
ScienceDirect
About ScienceDirect Shopping cart Contact and supportTerms and conditionsPrivacy policy
We first started writing the grant application that supported this work in 2017. The work was funded by the Coalition for Epidemic Preparedness Innovations (or CEPI). CEPI is a global partnership that was set up to accelerate the development and delivery of vaccines. The original plan was to look at three viruses – the one I was leading was influenza. We had a remit to develop a vaccine that could provide protection against infection within 6 weeks of the first immunisation.
The approach we used was an RNA vaccine, which unless you have been living under a rock for the last 3 years I am assuming you have heard of. But just in case, what these vaccines do is to take the genetic material that encodes a tiny bit of the virus and inject it into the muscle. Once injected your muscle cells make the viral protein training your immune system to recognise them. This means that when you are exposed to the real virus, you can fight it off better. Specifically we were using a self-amplifying RNA vaccine, this is subtly different; you can in theory get bigger responses for smaller amounts of material. I have described them before here.
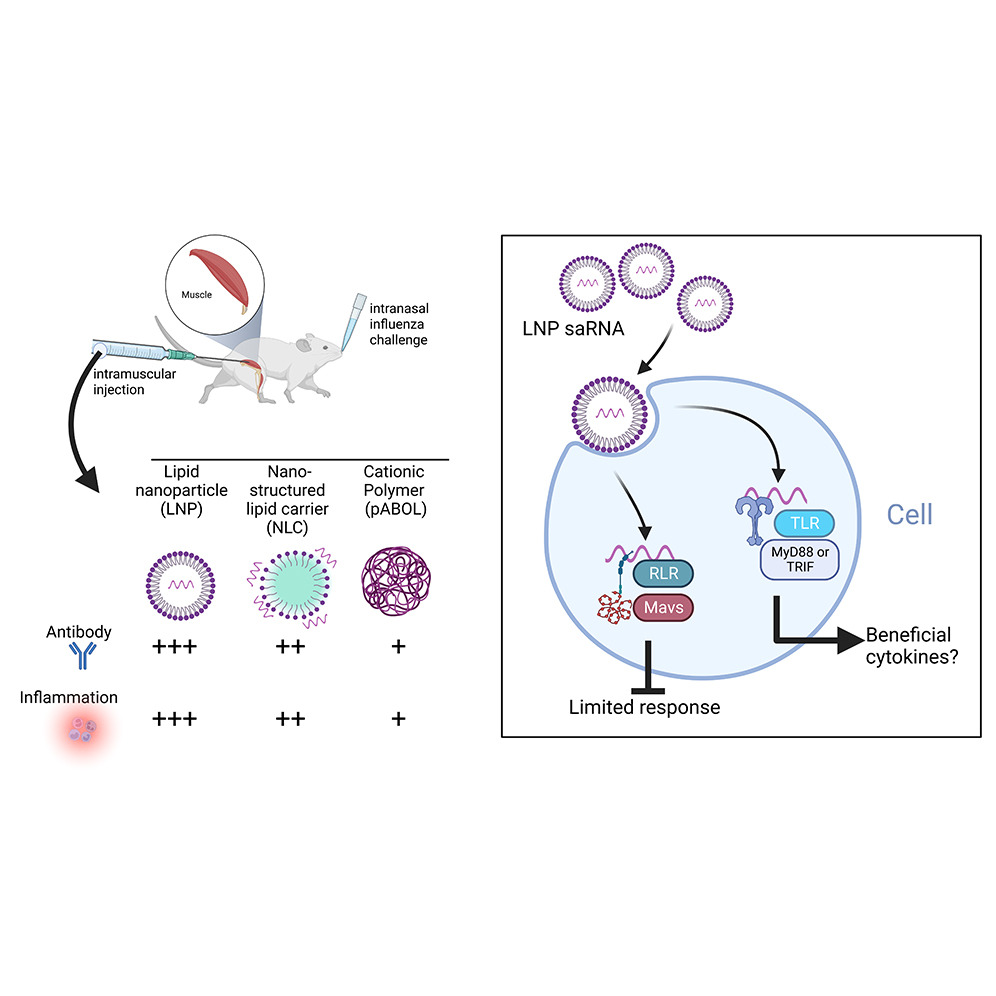
One of the first questions we looked at was how best to formulate the vaccine; the work is described in our recently published paper Formulation, inflammation and RNA sensing impact the immunogenicity of self-amplifying RNA vaccines. RNA is quite unstable and needs to be mixed with other compounds in order to get it into cells. We tried three approaches a cationic polymer (pABOL), a lipid emulsion (nano-structured lipid carrier, NLC) and three lipid nanoparticles (LNP). In simple terms to get RNA into a cell, you either need to add positive molecules (the cationic polymer) or some fat bubbles (the LNP and the NLC). We noticed that responses to the LNP were very much better than the other approaches and wanted to understand why.
One of the questions we asked was about the role of inflammation. Inflammation is a cascade of signalling by which the immune system recruits cells to a site of infection or danger. In conventional vaccines it is important because it alerts the cells that there is something foreign to be recognised and trains up the vaccine response. However, there was a question about its role in RNA vaccines, because RNA vaccines need to be made into proteins in the body to work some aspects of the immune system might inhibit this. Surprisingly (which is up there in the go to words of academic writing, alongside interestingly), we saw that vaccine induced inflammation was associated with better, not worse responses. The next step is to further dissect how this inflammation is beneficial.
Coming back to the timing of this project. We started the labwork in 2019, when the idea of a world changing pandemic virus was somewhere in the future. Then of course the events of 2020 caught up with us. The work definitely slowed down – though aspects of it were incorporated into the Imperial College vaccine trial. The formulation that we had showed to work best in the saRNA system was the one used in the clinical trial. As part of the ongoing vaccine research, I was able to come into the lab occasionally, providing a sanity lifeline and also the very odd experience of commuting through an empty London. Where this exciting vaccine technology goes next is still an important research question and one I look forward to continuing to investigate.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in