Adaptation of Staphylococcus aureus to the airways correlates with increased exploitation of metabolites released by the lung microbiota
Published in Microbiology and General & Internal Medicine
Our lab is located on the campus of a children’s hospital that manages hundreds of children with CF. When I started my PhD, the team had already shown that the main quorum-sensing system, encoded by the agr genes, was frequently inactivated in lung-adapted isolates collected from the sputa of children with CF1,2. However, the explanation for the accumulation of agr mutants in CF lungs remained elusive, and we decided to investigate the metabolic drivers of this phenomenon. We hypothesized that the abnormally thick mucus composed of hypersialylated mucins found in the lung environment of CF patients might play a key role in the selection of agr mutants. We thus decided to focus on the as yet poorly characterized interaction of airway sialic acid with S. aureus.
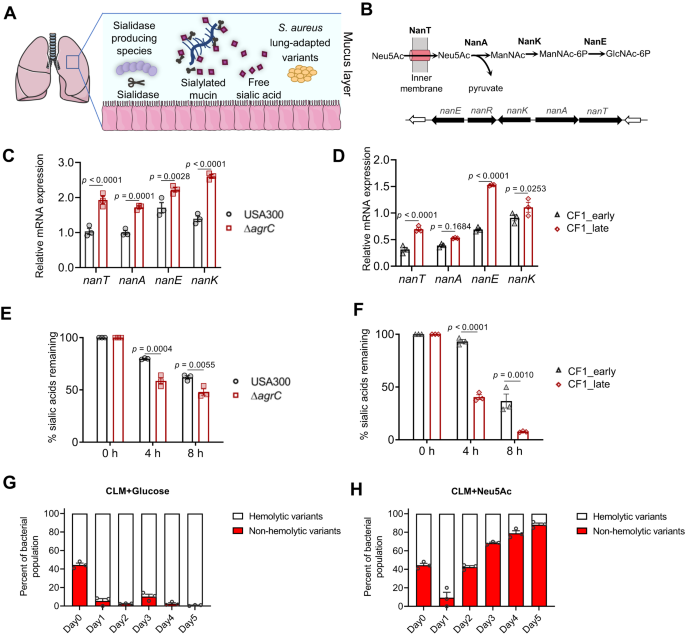
The starting point was the observation that the nan gene locus, involved in sialic acid catabolism in S. aureus, was overexpressed in agr mutants and lung-adapted clinical isolates with Agr dysfunction compared to their ancestral counterparts. The main consequence is a rapid consumption of sialic acid. In a mixed culture of WT and agr mutant, the agr mutant progressively became dominant over the WT strain in the presence of sialic acid. A metabolic driver of agr mutant accumulation has been discovered!
We reasoned that, although S. aureus lacks sialidase to release sialic acid from airway mucins, other bacterial species sharing the same ecological niche might very well do the job. We began by confirming the presence of free sialic acid in patients' sputum and analyzing the microbiota using a combination of 16S, shotgun metagenomics and sputum culture. We directly demonstrated the ability of sialidase-producing species in CF sputum to release free sialic acid from CF mucins. Cross-feeding between sialidase-producing species and S. aureus has thus been demonstrated in the CF lung environment.
To further simulate the lung environment, we used an air-liquid model of respiratory cells grown in vitro and demonstrated the growth advantage of the agr mutant in the presence of sialic acid. The agr mutant strain also utilized sialic acid more efficiently and exhibited faster proliferation in a mouse lung infection model.
To better understand the consequences of the presence of sialic acid in the lung environment, we performed an RNA-seq analysis. The “sialic acid regulon of S. aureus” encompasses the biosynthesis locus of staphyloferrin B, a siderophore, but also several virulence factors involved in acute infection. RNA-seq analysis revealed that sialic acid regulated the small RNA isrR, which is involved in iron utilization and displays predicted NanR binding motifs in its upstream region. NanR, the repressor of the nan locus, could regulate isrR, which in turn could affect siderophore production. To validate this hypothesis, we generated a nanR deletion mutant, and demonstrated an increase in isrR and sbnA expression compared with WT. The detailed regulatory circuits linking NanR and siderophore expression remain to be investigated.
Altogether, our findings suggest that sialic acid, released by sialidase-producing species in the airway, favors the accumulation of Agr dysfunctional variants by enhancing their sialic acid utilization and iron acquisition capabilities. As a result, lung-adapted S. aureus isolates are better able to exploit and respond to metabolites released by the surrounding microbiota.
The life of an apprentice researcher is not a smooth and straightforward path. I’ve just opened the door to unveil the incredible potential of interactions between bacterial species. I am now ready to continue unravelling these fascinating interactions, where each member of the community possesses its own metabolic skills and contributes to the dynamic structure of the ecosystem as a whole.
1. Tan, X. et al. Chronic Staphylococcus aureus Lung Infection Correlates With Proteogenomic and Metabolic Adaptations Leading to an Increased Intracellular Persistence. Clin Infect Dis 69, 1937-1945, doi:10.1093/cid/ciz106 (2019).
2. Ramond, E. et al. Lung-Adapted Staphylococcus aureus Isolates With Dysfunctional Agr System Trigger a Proinflammatory Response. J Infect Dis 226, 1276-1285, doi:10.1093/infdis/jiac191 (2022).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
Very nice correlation between staphylococcus persistence and CF lung metabolites. Thanks for sharing