An Intranasal Vaccine for Lyme Disease
Published in Microbiology, General & Internal Medicine, and Immunology

What is Lyme disease?
Humans acquire Borrelia burgdorferi after a bite of an infected deer tick (Ixodes scapularis) in areas where Lyme disease (LD) is endemic. In the US, LD is a major public healthcare problem throughout the Northeast, Mid-Atlantic and Midwest regions and its range continues to expand. It is a progressive, potentially debilitating disease with a wide array of largely non-specific clinical manifestations gradually developing from early to late stage. About 35% of people develop a typical rash in the tick bite site which facilitates diagnosis and treatment 1. However, ~35% develop atypical rashes and ~ 30% do not develop a rash at all. These individuals are at increased risk for developing late LD which is associated with permanent damage to the nervous and musculoskeletal systems. Although LD is the most prevalent vector-borne disease in the US and Europe, there’s no licensed vaccine available.
What is the current status of vaccine development for Lyme Disease?
Valneva/Pfizer have a Phase III clinical trial under way that may lead to FDA approval of a second-generation vaccine based in OspA for LD prevention. The vaccine is administered by 3 intramuscular shots over a period of 4-6 months. An additional shot may be needed at a later time within a year.
The case for using Outer surface protein A. OspA is the only safe immunogen proven to provide protection 2 against tick-transmitted B. burgdorferi in fully vaccinated human subjects. It is equally effective in other hosts such as mice and dogs. Because host generated anti-OspA antibodies neutralize B. burgdorferi within the midgut of the feeding tick, this is a vaccine that blocks transmission of the spirochete from the tick to the host. In contrast to other vaccines that prevent pathogen dissemination and mitigate disease severity, the transmission blocking function of OspA antibody prevents overall infection of the host.
As it is known, an OspA based vaccine was approved by the FDA in 1998 for commercialization by SmithKline Beecham (SKB, now GSK). However, the vaccine was removed from the market four years later due to concerns raised by a study published within months of vaccine approval. It was proposed that molecular mimicry between a self-peptide and an epitope present in OspA expressed by B. burgdorferi in the joint might lead to an autoimmune inflammatory event in susceptible treatment-resistant Lyme arthritis patients. The limitations of the molecular mimicry hypothesis were not assertively discussed at the time; thus, rebuttals and challenges to this hypothesis did not arise 3. Going forward, vaccine developers focused on creating second generation OspA vaccines that replaced the epitope of concern. This includes the sequences of the vaccine candidates developed by Baxter for intramuscular inoculation 4, Valneva (now licensed to Pfizer) also for intramuscular inoculation 5, and the sequence contained in our own vaccine developed for intranasal administration.
Of note, twenty years of additional clinical research on the safety profiles of second generation OspA vaccines reproduced the initial clinical trial data, that there are no significantly different safety issues among human subjects in the vaccine and in the placebo groups.
Our contribution
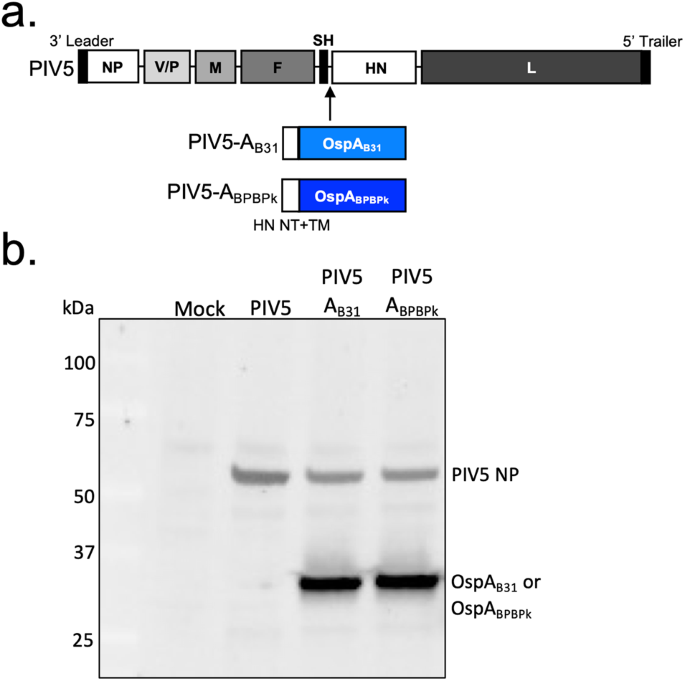
We focused on developing a second generation OspA vaccine using a live parainfluenza virus 5 (PIV5) viral-vector to deliver the antigen, free of the epitope of concern, intranasally. In addition to using a new, non-invasive route of administration (intranasal), other goals were to reduce the number of vaccine doses and extend the vaccine efficacy longevity.
PIV5 is a single-stranded, negative-sense, RNA virus belonging to the family Paramyxoviridae. Many PIV5-vectored vaccines for viral and bacterial pathogens have been evaluated in pre-clinical studies, and PIV5-based RSV (BLB201) and COVID-19 (CVXGA1) vaccines have undergone phase I 6 and II clinical trials in human subjects, with results showing the vaccines to be safe and immunogenic in healthy adults.
Mice were vaccinated by prime-boost intranasal inoculation with live PIV5 carrying the wild type ospA (PIV5-AB31) or live PIV5 carrying the mutated ospA devoid of the epitope of concern (PIV5-ABPBPk) in comparison with subcutaneous inoculation of purified recombinant OspA (s.c. rOspAB31) in alum adjuvant, and the respective controls. Immunogenicity and protective efficacy of the vaccine candidates were tested in three independent studies lasting 4- (Study 1), 9- (Study 2), and 18- (Study 3) months to evaluate the longevity of protective immune responses after tick challenge. Challenge with infected ticks carrying 10-19 strains of B. burgdorferi performed at 4-, 9- or 15-months post-immunization showed increased breakthrough infections in mice vaccinated with s.c. rOspAB31 compared to intranasal PIV5-AB31 or PIV5-ABPBPk at 9- and 15-months, as determined by quantification of serologic antibodies to B. burgdorferi proteins as well as flaB DNA in tissues, and by visualization of motile B. burgdorferi in culture of tissues under dark field microscopy. Neutralization activity of antibody was maintained up to 18-months post-immunization, with the response greater in live PIV5-delivered OspA vaccines, than that induced by s.c. rOspAB31.
We found that prime-boost immunization of mice with PIV5 delivered OspA generates immune responses that produce longer-lasting protection (> 1 year) against tick-transmitted B. burgdorferi than the parenteral recombinant OspA.
Our work advances the Lyme disease vaccine field in 3 ways: it reduces the number of immunizations needed from at least 3 to 2; it provides a non-invasive immunization route, an alternative to the classic intramuscular shot injection; it provides much longer protection against tick transmitted B. burgdorferi as the longevity of the vaccine was extended from 4-6 months to >1 year.
References
- Marques, A. R. Lyme disease: a review. Curr Allergy Asthma Rep 10, 13-20, doi:10.1007/s11882-009-0077-3 (2010).
- Steere, A. C. et al. Vaccination against Lyme disease with recombinant Borrelia burgdorferi outer-surface lipoprotein A with adjuvant. Lyme Disease Vaccine Study Group. N Engl J Med 339, 209-215, doi:10.1056/NEJM199807233390401 (1998).
- Dattwyler and Gomes-Solecki. The year that shaped the outcome of the OspA vaccine for human Lyme disease. NPJ Vaccines. 2022. Jan 27; 7(1):10. DOI: 10.1038/s41541-022-00429-5
- Wressnigg, N. et al. Safety and immunogenicity of a novel multivalent OspA vaccine against Lyme borreliosis in healthy adults: a double-blind, randomised, dose-escalation phase 1/2 trial. Lancet Infect Dis 13, 680-689 (2013). https://doi.org:10.1016/S1473-3099(13)70110-5
- Comstedt, P., Schuler, W., Meinke, A. & Lundberg, U. The novel Lyme borreliosis vaccine VLA15 shows broad protection against Borrelia species expressing six different OspA serotypes. PLoS One 12, e0184357 (2017). https://doi.org:10.1371/journal.pone.0184357
- Spearman, P. et al. Intranasal parainfluenza virus type 5 (PIV5)-vectored RSV vaccine is safe and immunogenic in healthy adults in a phase 1 clinical study. Sci. Adv. 2023 Oct 27;9 (43). DOI: 10.1126/sciadv.adj7611
Follow the Topic
-
npj Vaccines
A multidisciplinary journal that is dedicated to publishing the finest and high-quality research and development on human and veterinary vaccines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Therapeutic HPV vaccines
Publishing Model: Open Access
Deadline: Jun 30, 2026
Lipid nanoparticle (LNP)-adjuvanted vaccines
Publishing Model: Open Access
Deadline: May 19, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in