Asymmetric transformations of an achiral gold nanocluster
Published in Chemistry
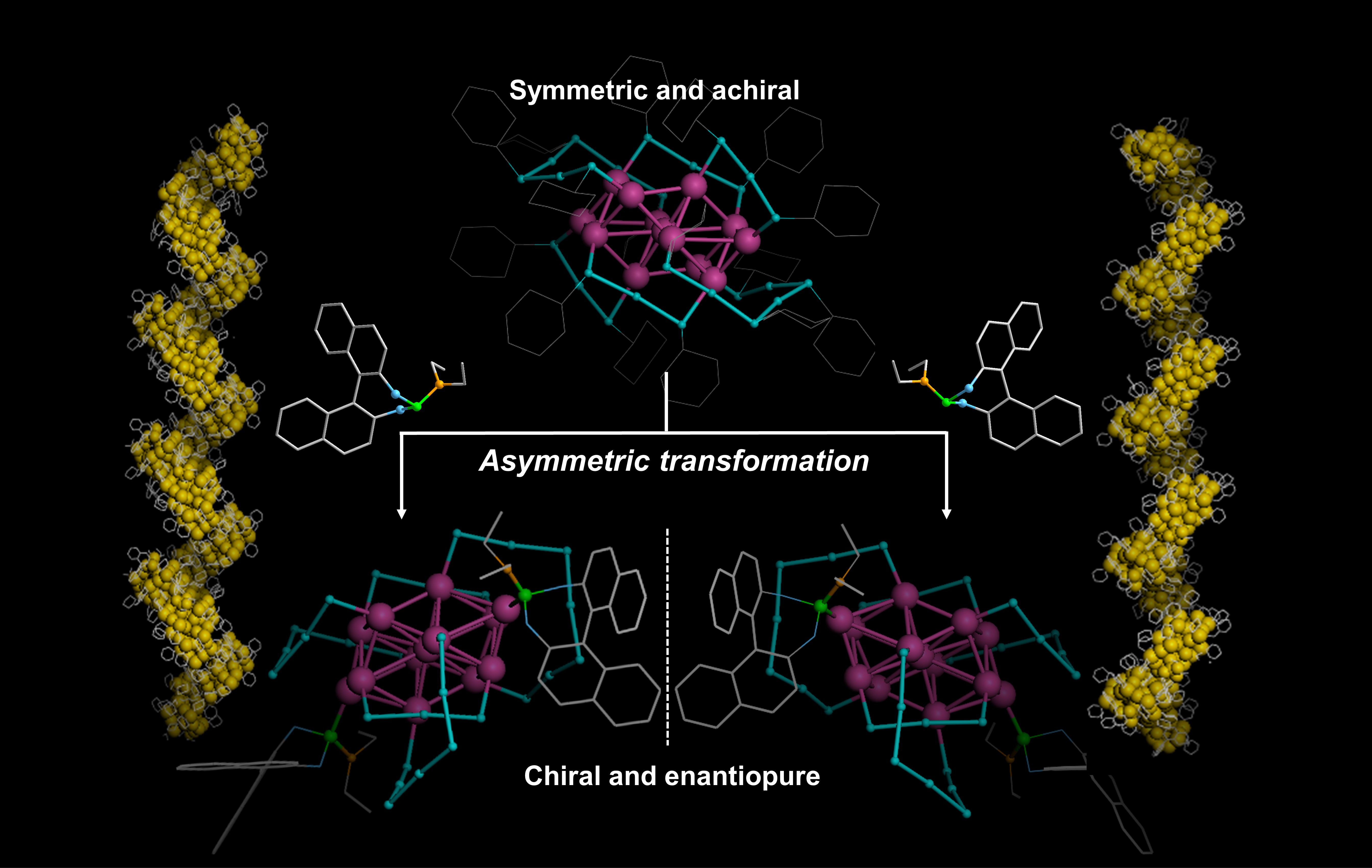
The research group of Prof. Man-Bo Li at Anhui University focuses on the atomically precise construction of metal nanoclusters and their applications in catalyzing organic transformations in recent years (ACIE 2023, e202215741; Nano Lett. 2023, 235; JACS 2023, 12164). Based on our combined background in metal nanocluster catalysis (Acc. Chem. Res. 2021, 2275; ACIE 2021, 670; CCS Chem. 2021, 1127; ACIE 2020, 10391; ACIE 2020, 1992; JACS 2018, 14604) and organic methodology (JACS 2023, doi: 10.1021/jacs.3c04864; Chem Catal. 2023, 100582; CCS Chem. 2022, 1938; ACIE 2012, 2968), we developed a ligand-induced asymmetric transformation method for the synthesis of enantiopure nanoclusters, which would be applied for asymmetric catalysis of atomically precise metal nanoclusters in the future. This work has been published on Nature Communications very recently.
The investigation of chirality at nanoscale is important for bridging the gap between molecular chirality and macroscopic helix.[1,2] Atomically precise metal nanoclusters constitute an ideal platform for the research of nanomaterials’ chirality at atomic level,[3] while the achievement of enantiopure metal nanoclusters remains a great challenge. Three methods have been developed toward acquiring metal nanoclusters with enantiomeric excess, including: (i) enantioseparation of racemic mixtures of metal nanoclusters by chiral high performance liquid chromatography (HPLC)[4,5] or resolving agents;[6,7] (ii) enantioselective phase transfer of chiral nanoclusters;[8] (iii) direct synthesis of enantiopure nanoclusters by using chiral organic ligands as the precursors.[9-13] However, the method for transforming an achiral metal nanocluster into a chiral one is limited. Considering that hundreds of achiral metal nanoclusters being reported, we conceive the possibility of using a chiral inducer to realize the asymmetric transformation of achiral metal nanoclusters, which would be developed into a general protocol for the construction of enantiopure metal nanoclusters.
Based on systematic studies, phosphoramidite (L) was found to be a good candidate to induce the asymmetric transformations of thiolate-protected gold nanocluster Au23(SC6H11)16. Two nanocluster enantiomers (R)-Au24(L)2(SC6H11)16 and (S)-Au24(L)2(SC6H11)16 were obtained separately in the reactions between Au23(SC6H11)16 and (R)-L or (S)-L. Crystal structures revealed that the asymmetric transformation not only brings the intrinsic chirality of phosphoramidite, but also triggers the asymmetric rearrangement of the staple motifs and nanocluster molecules that was originally symmetric in Au23, constituting the hierarchical chirality of Au24(L)2(SC6H11)16. A negative nonlinear CD-ee dependence was also discovered on the relationship between the chiroptical activity of Au24(L)2(SC6H11)16 and the enantiomeric excess of L.
More details of this work could be found here: “Asymmetric transformation of achiral gold nanoclusters with negative nonlinear dependence between chiroptical activity and enantiomeric excess” in Nature Communications.
References
- Yashima, Ousaka, N., Taura, D., Shimomura, K., Ikai, T. & Maeda, K. Supramolecular helical systems: helical assemblies of small molecules, foldamers, and polymers with chiral amplification and their functions. Chem. Rev. 116, 13752-13990 (2016).
- Magnin, , Amara, H., Ducastelle, F., Loiseau, A. & Bichara, C. Entropy-driven stability of chiral single-walled carbon nanotubes. Science 362, 212-215 (2018).
- Li, , Higaki, T., Du, X. & Jin, R. Chirality and surface bonding correlation in atomically precise metal nanoclusters. Adv. Mater. 32, 1905488 (2020).
- Knoppe, , Dolamic, I., Dass, A. & Bürgi, T. Separation of enantiomers and CD spectra of Au40(SCH2CH2Ph)24: spectroscopic evidence for intrinsic chirality. Angew. Chem. Int. Ed. 51, 7589-7591 (2012).
- Liu, , Xu, W. W., Huang, X., Wang, E., Cai, X., Zhao, Y., Li, J., Xiao, M., Zhang, C., Gao, Y., Ding, W. & Zhu, Y. De novo design of Au36(SR)24 nanoclusters. Nat. Commun. 11, 3349-3356 (2020).
- Deng, , Teo, B. K. & Zheng, N. Assembly of chiral cluster-based metal-organic frameworks and the chirality memory effect during their disassembly. J. Am. Chem. Soc. 143, 10214-10220 (2021).
- Zhu, , Wang, H., Wan, K., Guo, J., He, C., Yu, Y., Zhao, L., Zhang, Y., Lv, J., Shi, L., Jin, R., Zhang, X., Shi, X. & Z. Tang, Enantioseparation of Au20(PP3)4Cl4 clusters with intrinsically chiral cores. Angew. Chem. Int. Ed. 57, 9059–9063 (2018).
- Knoppe, , Wong, O. A., Malola, S., Häkkinen, H., Bürgi, T., Verbiest, T. & Ackerson, C. J. Chiral phase transfer and enantioenrichment of thiolate-protected Au102 clusters. J. Am. Chem. Soc. 136, 4129–4132 (2014).
- Man, W. Y., Yi, H., Malola, S., Takano, S., Tsukuda, T., Häkkinen, H., Nambo, M. & Crudden, C. M. Synthesis and characterization of enantiopure chiral bis NHC-stabilized edge-shared Au10 nanocluster with unique prolate shape. J. Am. Chem. Soc. 144, 2056-2061 (2022).
- Sugiuchi, , Shichibu, Y. & Konishi, K. An inherently chiral Au24 framework with double-helical hexagold strands. Angew. Chem. Int. Ed. 57, 7855-7859 (2018).
- Wang, -Q., Guan, Z.-J., Liu, W.-D., Yang, Y. & Wang, Q.-M. Chiroptical activity enhancement via structural control: the chiral synthesis and reversible interconversion of two intrinsically chiral gold nanoclusters. J. Am. Chem. Soc. 141, 2384-2390 (2019).
- Kong, -J., Yan, Z.-P., Li, S., Su, H.-F., Li, K., Zheng, Y.-X. & Zang, S.-Q. Photoresponsive propeller-like chiral AIE copper(I) clusters. Angew. Chem. Int. Ed. 59, 5336-5340 (2020).
- Yao, -Y., Lee, T. K.-M. & Yam V. W.-W. Thermodynamic-driven self-assembly: heterochiral self-sorting and structural reconfiguration in gold(I)-sulfido cluster system. J. Am. Chem. Soc. 138, 7260-7263 (2016).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in