Bacterial Bubbles blow away the bugs
Published in Microbiology
Explore the Research
 sciencedirect.com
sciencedirect.com
ScienceDirect
About ScienceDirect Shopping cart Contact and supportTerms and conditionsPrivacy policy
There’s no shame in not knowing the names of every bacteria, there are billions if not trillions of different bacterial species. The number that are pathogenic in humans is significantly lower – probably in the high hundreds. One review estimates that there are 1,400 known species of human pathogens (including viruses and parasites). So, it is still ok to not have heard of all of them.
One that probably passes under the radar of many people is Acinetobacter baumannii, not least because it is an absolute nightmare to spell. It is a gram-negative opportunistic pathogen. Most cases of A. baumannii infection are hospital acquired, often catheter or ventilator associated. There are approximately 1 million A. baumannii infections annually and it is associated with a nearly 35% mortality rate. The most pressing problem associated with A. baumannii is antibiotic resistance; it is extremely drug resistant – with nearly half of the infections resistant to the last line antibiotic carbapenem. In fact Acinetobacter is the A in ESKAPE (the priority list of the most important antibiotic resistant bacteria).
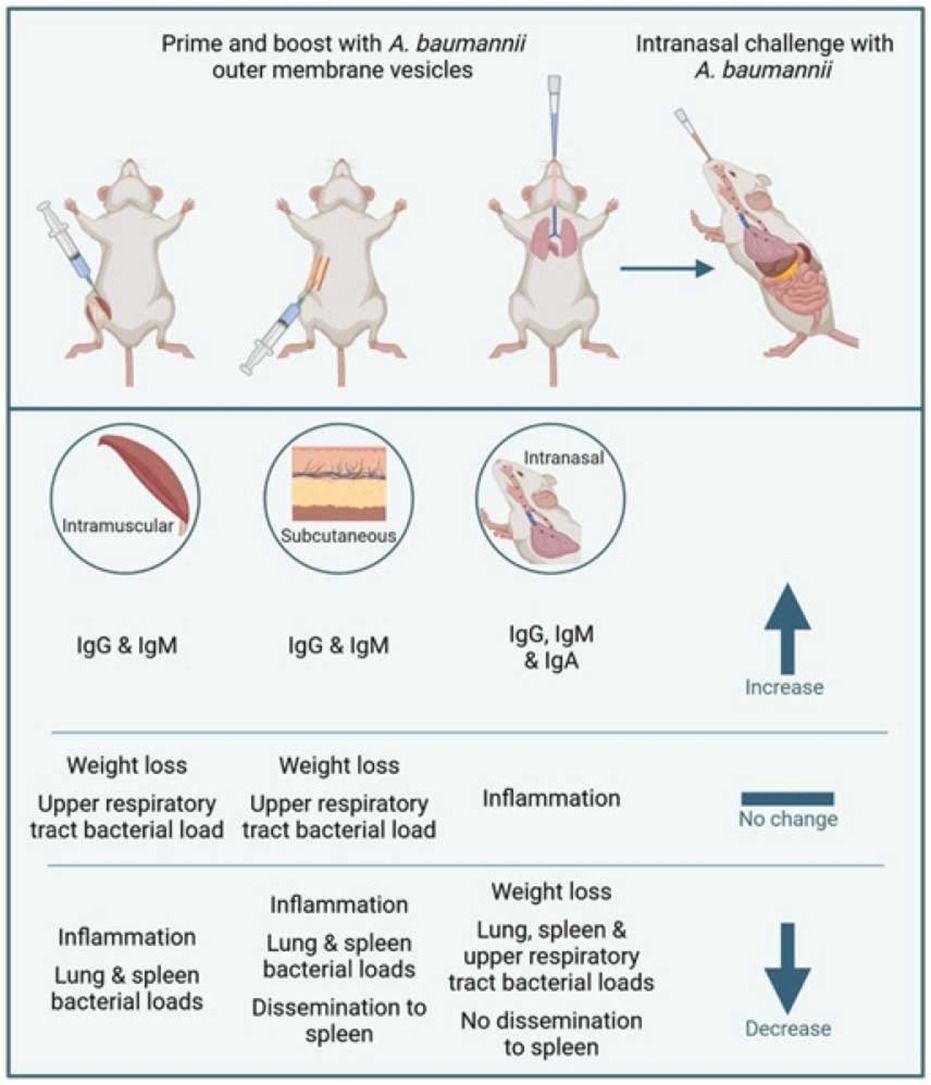
Since it is so drug resistant, other strategies are needed to control it. One of which might be vaccines. In our recent study - Intranasal immunization with outer membrane vesicles (OMV) protects against airway colonization and systemic infection with Acinetobacter baumannii we developed a novel vaccine against this important pathogen. The approach we used was to manipulate a product of the bacteria itself to generate protection. A. baumannii produces little bubbles of lipids and proteins called outer membrane vesicles (OMVs). Bacteria use these OMV to communicate with each other and potentially help them better infect us. However, they also contain lots of different bacterial antigens, some of which are potentially protective. The highly effective vaccine against Meningitis B (Bexsero made by GSK) contains OMV.
In our study, we isolated OMV from clinical isolates of A. baumannii. Using clinically derived strains was important – as these are probably closer to the ones in circulation, than the ones used in many studies from older varieties. To test whether the OMV worked, we developed new models of infection using the same strains. We showed the recent clinical isolates were more pathogenic than the standard lab strain. The clinical isolates were able to escape from the lungs into the blood and appeared to stably colonise the upper airways for at least 7 days after the initial infection.
Our first studies tested injecting the vaccine into the muscle – as this is the most common route of vaccination used. OMV injected this way did give a good immune response – leading to the induction of antibodies. However, the immunity raised following this route of immunisation was not very protective against subsequent infection. When area of interest in the vaccine field has been mucosal vaccination – delivering vaccines to the site of infection, in this case the nose and lungs. When Dr Sophie Higham (lead author on the paper) immunised via the nose, protection was significantly improved with a dramatic reduction in bacterial load following infection.
This work shows two things – OMV can be very effective vaccine candidates against bacterial infections and that immunising in the site of infection can be beneficial. The next step is to look how to scale up for human studies.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in