
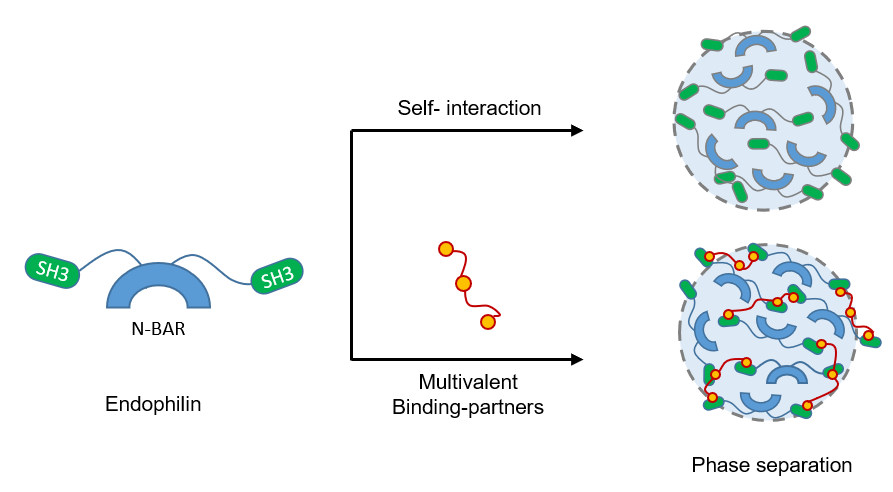
Protein phase separation is of central importance to an increasing number of biological processes. Bio-macromolecules (proteins, ribonucleotides) showing multivalent interactions can phase separate into liquid-like assemblies that can spatiotemporally regulate their cellular functions. Recently, we have realized that phase separation of Bin/Amphiphysin/Rvs (BAR)-domain proteins, the major drivers of membrane curvature in endocytosis, could play a crucial role in endocytic protein assembly on the membrane. We found that the BAR protein, endophilin, which is also essential for a clathrin-independent endocytic pathway, known as Fast Endophilin Mediated Endocytosis (FEME), can undergo phase separation via both self-association and binding to its multivalent partners (Fig. 1). Our study indicates that phase separation of endophilin facilitates protein network formation at the initiation stage as well as during the progression of FEME.
Earlier, it was shown that endophilin’s interaction with the C-terminal domain of lamellipodin (LPD) and the third intracellular loop (TIL) of G-protein coupled receptors are essential for the formation of FEME sites and for the progression of membrane trafficking. However, the underlying molecular mechanism was not clearly understood. To explore this complex multi-protein network, we began by developing a simplistic model system involving its three key components - endophilin, LPD, and TIL to closely understand their interactions in solution and on the membrane surface (Fig. 2).
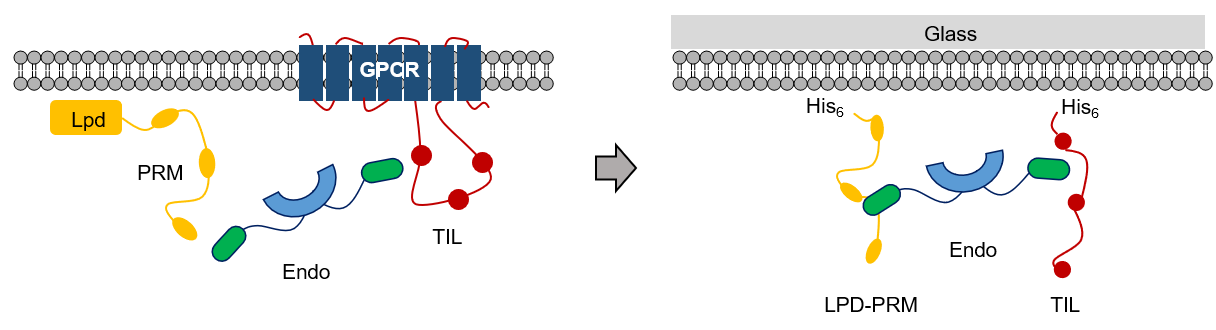
Fig. 2: Our simplistic membrane reconstitution model involving endophilin and its multivalent binding partners in FEME.
We started with an initial hypothesis that the multivalent nature of the interactions between endophilin’s Src Homology 3 (SH3) domain and proline-rich motifs (PRMs) present on both LPD and TIL would drive liquid-liquid phase separation. Surprisingly, we observed that endophilin phase separated not only with LPD and TIL but also by itself in a crowded environment. The BAR domains of endophilin and several other BAR-proteins (such as amphiphysin-1, and BIN1) were found to be a major driver of phase separation by ‘homotypic’ interactions. Moreover, various FEME-proteins were found to partition into endophilin droplets and modulate phase boundary and droplet size demonstrating their significance in the regulation of such multiprotein assemblies in cells.
Our results on protein-reconstituted model membranes suggest that the phase separation of endophilin and its multivalent binding partners (TIL and LPD) is a key mechanism behind the formation of protein assembly on the membrane. Further by reconstituting endophilin, TIL, and LPD – the three key components of FEME to membranes, we demonstrate that the evolution of FEME initiation sites to stable membrane-transport carriers could be facilitated by synergistic actions of these proteins in the formation of membrane clusters.
Our knowledge of protein phase separation is expanding exponentially day by day. To this field, we introduced a new class of proteins, which also demonstrates interesting membrane remodeling functionality. Future research would be directed toward understanding how this newly identified phase separation behavior modulates the membrane-activity in the BAR proteins.
This story would remain incomplete without mentioning the challenges we came across while working on this project. Purifying proteins with large, disordered domains in high concentrations is already quite challenging. However, reconstituting multiple such proteins to model membranes while maintaining the membrane integrity brings in another next level of difficulties. It would not have been possible to cross this barrier without such highly coordinated teamwork between our very enthusiastic team members.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in