Behind the Paper: Stroma AReactive Invasion Front Areas (SARIFA) improves prognostic risk stratification of perioperative chemotherapy treated oesophagogastric cancer patients from the MAGIC and the ST03 trial
Published in Cancer, General & Internal Medicine, and Anatomy & Physiology
Introduction:
The field of oesophagogastric cancer treatment has seen significant advancements with the introduction of perioperative combination chemotherapy. However, the challenge of locally recurrent disease or distant metastasis remains, necessitating the identification of a reliable biomarker to predict patient outcomes. In this paper, we present our Stroma AReactive Invasion Front Areas (SARIFA) study, which validated a novel biomarker with the potential to change post-operative treatment decisions and improve patient quality of life.
Background:
Despite a large number of proposed biomarkers, none have been widely adopted in clinical practice, leaving TNM disease stage as the most important prognostic parameter for treatment decisions. This study aimed to address this urgent clinical need by validating a recently identified easy to implement biomarker that can predict the risk of recurrent disease and overall survival after neoadjuvant chemotherapy, independent of well-established prognostic factors.
Research Objective:
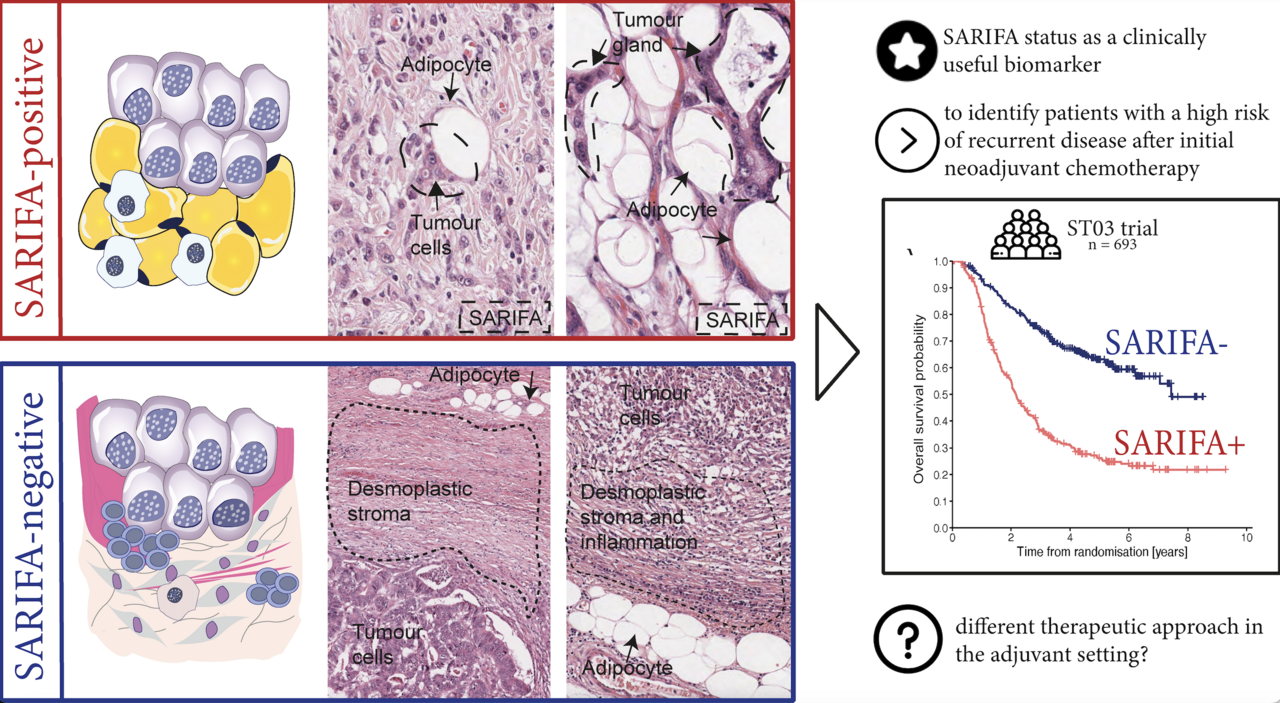
Building directly on our previous research, the study investigated the presence of SARIFA in patients with oesophageal cancer recruited into 2 large randomised UK clinical trials (MAGIC trial and ST03 trial). SARIFA status depends on the presence or absence of tumour-associated fat cells at the invasive edge and was previously identified as a prognostic biomarker in gastric cancer patients treated with surgery alone. The current study hypothesized that SARIFA status can predict the prognosis in all oesophagogastric cancer patients with resectable disease, regardless of whether they were treated by surgery alone or perioperative combination chemotherapy.
Key Findings:
The study validated the prognostic value of SARIFA status in an independent patient series from the MAGIC trial. Additionally, it was the first to demonstrate that SARIFA status can identify patients at high risk of death from relapse after gold standard perioperative chemotherapy, independent of known prognostic factors (patients from the ST03 trial). Importantly, SARIFA status can be determined from routine Haematoxylin & Eosin-stained tissue sections, requiring no additional materials or staining procedures. This allows for quick turnaround time and easy implementation into routine practice worldwide.
Combining SARIFA status with lymph node status enabled the identification of subgroups of patients with different survival outcomes. Notably, patients with SARIFA-positive tumours without lymph node metastasis had the same poor survival as patients with SARIFA-negative and lymph node metastases. This suggests that the assessment of the SARIFA status could be particularly useful in identifying patients without lymph node metastasis after neoadjuvant therapy who may require changes to their adjuvant treatment regimen to improve their otherwise poor survival.
Significance:
The results of this study provide clinicians and patients with a decision-making tool independent of known prognostic factors such as disease stage. This breakthrough will provide clinicians and patients with the opportunity for better informed for personalized treatment decisions after initial standard chemotherapy and surgery ultimately improving outcomes and enhancing patient care. Furthermore, SARIFA status is an easy to understand biomarker which can be incorporated into routine histopathology reports with minimal effort for the pathologist.
Conclusion:
The SARIFA study introduces a novel biomarker that has the potential to transform the management of patients with oesophagogastric cancer. The improved prediction of individual patient outcomes should improve precision and effectiveness in post-operative care in th near future. With its ease of implementation in any histopathology laboratory, SARIFA status determination represents a significant advancement in the field.
Follow the Topic
-
British Journal of Cancer
This journal is devoted to publishing cutting edge discovery, translational and clinical cancer research across the broad spectrum of oncology.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in