Bioinspired Self-adaptive Dual-metal-site Pairs for Photoreduction of CO2 to CH4
Published in Chemistry

Photochemical reduction of carbon dioxide (CO2) to alcohols and hydrocarbons as value-added fuels and platform chemicals provides one of most promising technologies for timely achieving “emission peak” and “carbon neutrality” goals, which were outlined in The Paris Agreement1. Thermodynamically, the captured CO2 can be facilely converted into carbon monoxide (CO), formic acid (HCOOH), while the selectivities to methane (CH4), ethylene (C2H4) and methanol (CH3OH) under ambient conditions are relatively low. Compared to the two-electrons transfer products, the photoreduction of CO2-to-CH4 is an eight proton-coupled electron transfer (PCET) process with various C1 intermediates generated over active sites. As indicated by Sabatier principle, the optimal/moderate bonding strengths between the active sites and multiple reaction intermediates are highly required for the desired catalytic performance2. To achieve this, both the electronic and geometric properties of the active sites need to be finely tuned.
The single-atom catalysts (SACs) with the maximum atom utilization have attracted tremendous interests in heterogeneous catalysis. However, obtaining the high activity and selectivity for complex reactions with multiple elementary reaction steps and reaction intermediates over the SACs is almost impossible. Thus, photocatalysts with two neighboring dual-metal atom sites have been proposed to potentially adjust the binding strength of key reaction intermediate. With this tuned electronic rearrangement, the CO binding strength is stronger, resulting in the selectivity of CO2 photoreduction shift from CO to CH4.
Now, reporting in Nature Catalysis, inspired by the natural enzyme system3 in which the catalytic active sites would adapt suitable conformations to interact with substrates and intermediates for the best catalytic performance with optimum bonding strengths, Li et al. design and successfully synthesize a novel photocatalyst with flexible dual-metal-site pairs (DMSPs) that exhibit dynamic and self-adaptive behavior to fit mutative C1 intermediates in the photocatalytic conversion of CO2. As illustrated in Fig. 1, DMSPs consisting of Cu and Ni in their respective single-site forms (referred to as Cu/Ni DMSPs) are incorporated into a robust metal-organic framework (MOF), i.e., MOF-808. The dynamic and self-adaptive behavior/microenvironment of DMSPs is achieved by post-synthesizing flexible ethylenediaminetetraacetic acid (EDTA) ligands onto the rigid Zr-oxo clusters4, followed by the chelation of metal ions to yield MOF-808-CuNi. With this design, both electronic and geometric properties of catalytic active sites are tuned as desired. This is complemented and confirmed by ab initio molecular dynamics (AIMD) simulations and density functional theory calculations. Two characteristic parameters, i.e., the distance between Cu and Ni single sites (dCu-Ni) and the dihedral angle formed by the four N atoms in two EDTA ligands (θ2N-2N) are introduced to describe the dynamic and flexible configuration of the DMSPs. It is observed that the values of dCu-Ni and θ2N-2N dynamically fluctuate in the ranges of 4.250 to 5.036 Å and 12.419° to 90.114°, respectively, when the DMSPs interact with different intermediates along the reaction pathway leading to the CH4 formation. More importantly, the self-adaptive feature of DMSPs can also stabilize the transition states by adjusting the flexible configurations. As a result, a high production rate of CH4 (158.7 μmol•g-1•h-1) with an electron-basis (Selelectron) 99.4% (product-basis (Selproduct) 97.5%) for CH4 over the MOF-808-CuNi catalyst are observed.
This protocol presented here features a novel synthetic approach, inspired by enzymes, for the fabrication of heterogeneous catalysts with self-adaptive bimetallic or even multi-metal sites for stabilizing reactive intermediates. This strategy, for the first time, provides a unique way that adaptively and simultaneously tunes the electronic and geometric properties of the active sites for enhanced catalytic performance. You can read more about this work in Nature Catalysis. “Self-adaptive dual-metal-site pairs in metal-organic frameworks for selective CO2 photoreduction to CH4" DOI: 10.1038/s41929-021-00665-3
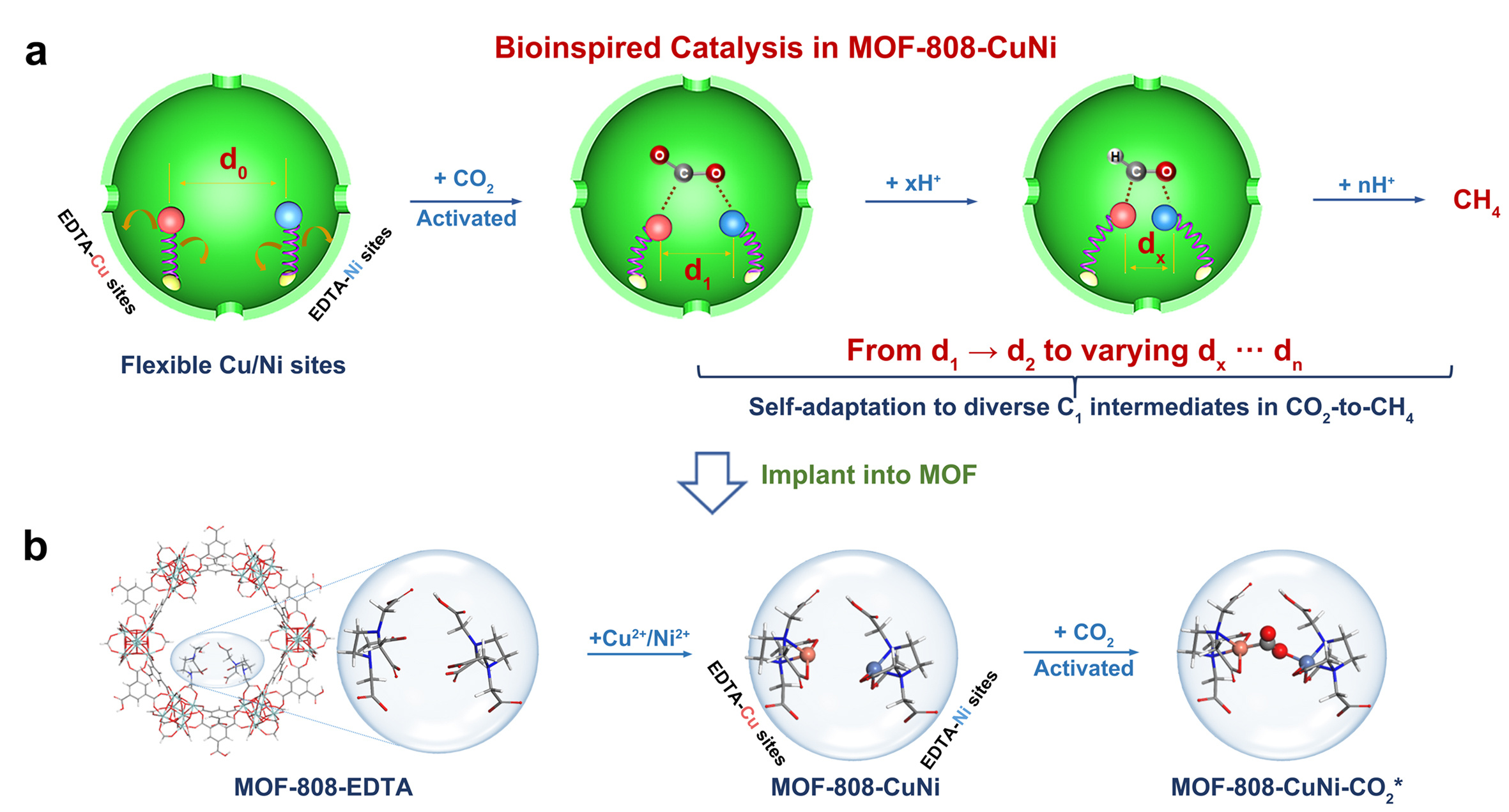
Fig. 1 Encapsulated flexible and self-adaptive Cu/Ni DMSPs into MOF-808 for highly selective CO2 photoreduction to CH4.
References.
- McDonald, T. M. et al. Cooperative insertion of CO2 in diamine-appended metal-organic frameworks. Nature 519, 303–308 (2015).
- Medford, A. J. et al. From the Sabatier principle to a predictive theory of transition-metal heterogeneous catalysis. J. Catal. 328, 36-42 (2015).
- Benkovic, S. J. & Hammes-Schiffer, S. A perspective on enzyme catalysis, Science 301, 1196-1202 (2003).
- Peng, Y. et al. A versatile MOF-based trap for heavy metal ion capture and dispersion, Nat. Commun. 9, 187 (2018).
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in