Carbon quantum dot doped NH₂-MIL-53 for sustainable atmospheric water harvesting
Published in Materials
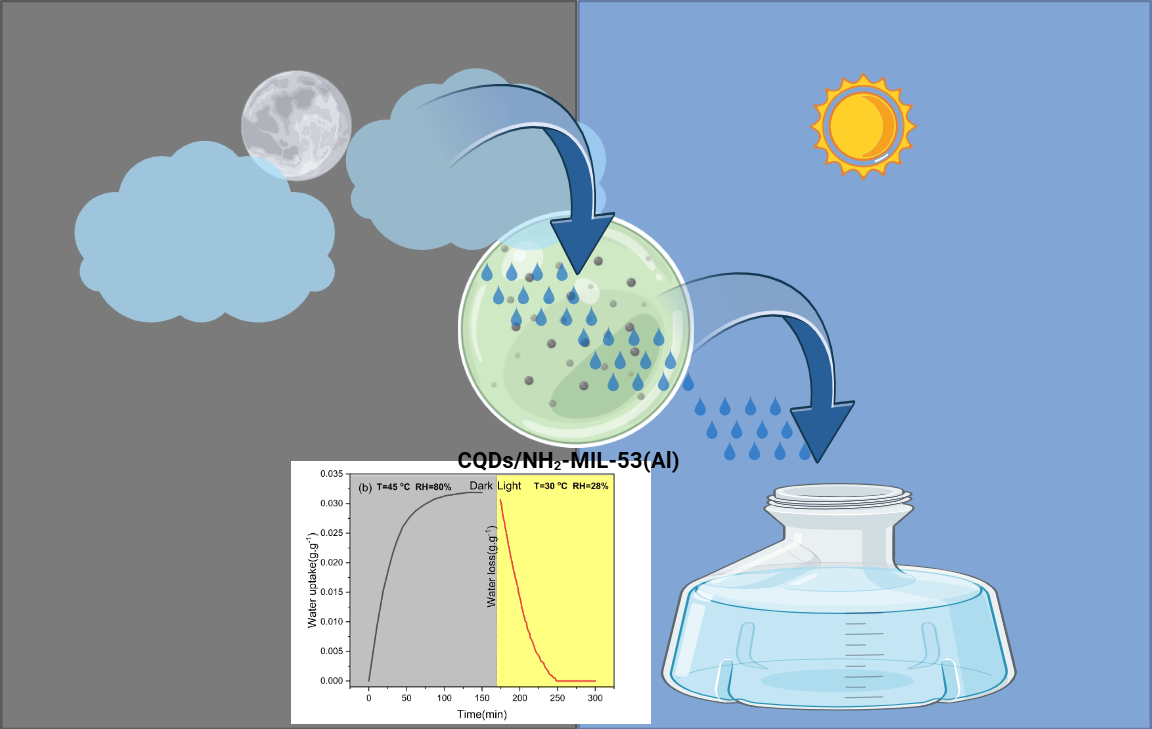
Sorption-based atmospheric water harvesting (SAWH) using metal–organic frameworks (MOFs) presents a promising solution to freshwater scarcity in arid regions. However, despite the intrinsic presence of abundant binding sites and polar functional groups, the practical application of MOFs remains limited due to their low adsorption/desorption efficiency and challenges associated with industrial-scale deployment in bulk form.
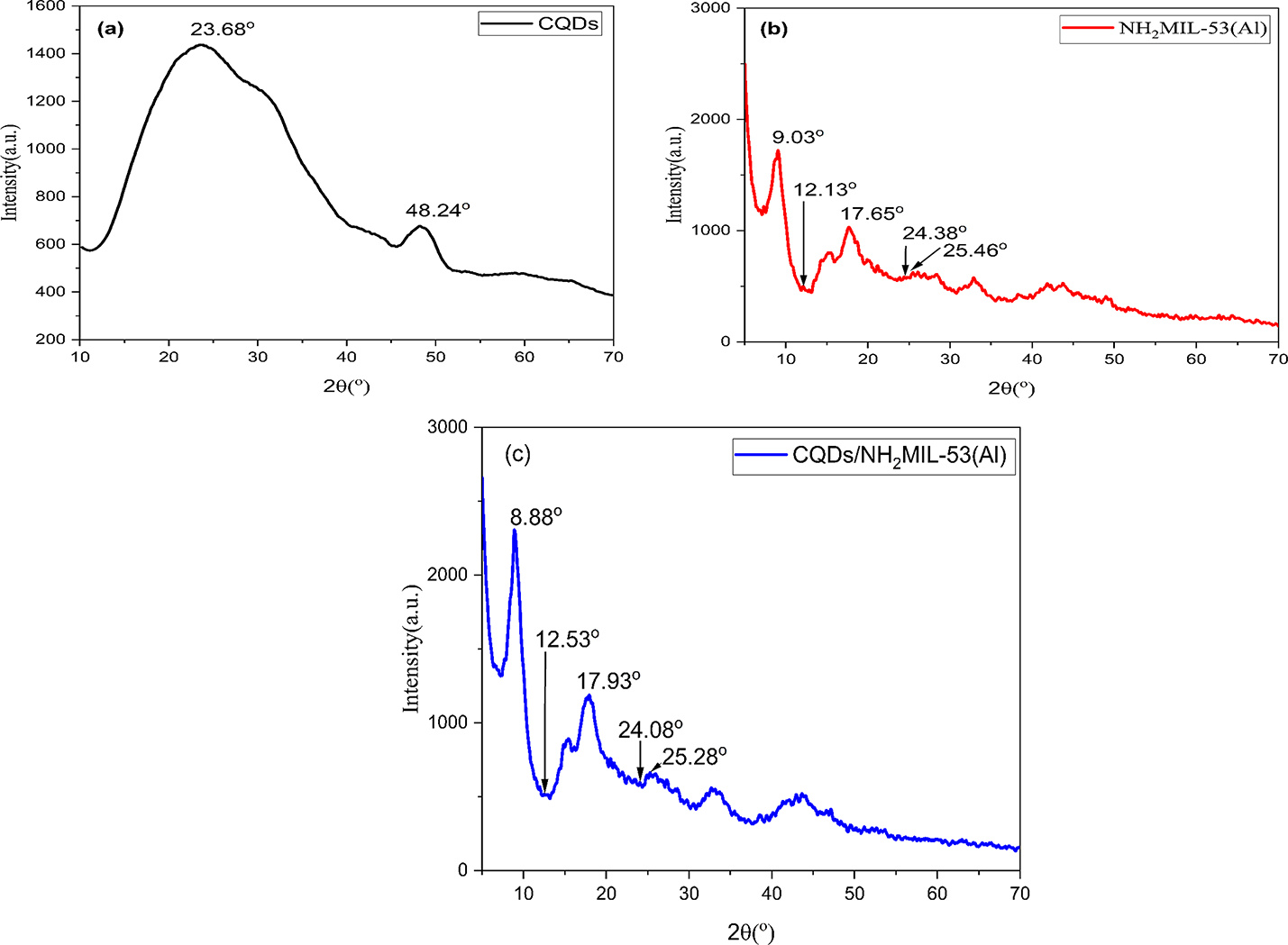
In this study, we developed a nanocomposite sorbent by doping carbon quantum dots (CQDs) onto the surface of NH₂-MIL-53(Al) via a hydrothermal method, ensuring the preservation of its original crystalline structure while enhancing its functionality. The hierarchical pore structure introduced by CQDs doping not only provides ample adsorption sites but also facilitates rapid moisture uptake and high solar-thermal conversion efficiency.
As a result, the material exhibits a significantly improved adsorption/desorption cycle, achieving complete water uptake and release within 300 min (150 min for adsorption and 150 min for desorption under simulated solar irradiation). Moreover, the nanocomposite demonstrates a water uptake capacity of 0.01317 g·g⁻¹ at 20% relative humidity (RH) and 0.092 g·g⁻¹ at 80% RH, surpassing the performance of most state-of-the-art MOF-based SAWH materials.
This work presents a simple yet effective strategy for enhancing the adsorption/desorption kinetics of NH₂-MIL-53(Al), offering valuable insights for the design of high-performance solar-driven water harvesting materials
Follow the Topic
-
Discover Chemistry
A fully open access, peer-reviewed journal supporting multidisciplinary research and policy developments across all fields of chemistry.
Related Collections
With Collections, you can get published faster and increase your visibility.
Advances in Material Chemistry for Life Science Research
Material chemistry has a long-standing history and has been extensively exploited in a wide range of life science research such as biological, chemical, and environmental engineering. The invention of novel materials for biological applications has led to the creation of biomaterials for tissue engineering, nanomaterials for drug delivery, and smart materials that interact dynamically with biological systems. These advances have significantly revolutionized the fields of healthcare including medical treatments, diagnostics, and biological studies. In recent years, the synergy between material chemistry and life sciences has been particularly evident in the development of materials that enable precision medicine, improve the accuracy of diagnosis, and facilitate regeneration of defected tissues. In particular, nanoparticles engineered for targeted drug delivery have significantly enhanced the efficacy of treatments while minimizing side effects, and responsive biomaterials have opened new avenues for developing adaptive therapeutic devices. The application of these advanced materials in life science not only enhances our understanding of biological processes but also paves the way for innovative solutions to pressing healthcare challenges. This topical collection aims to cover papers that explore the intersection of material chemistry and life sciences, focusing on the design, synthesis, and application of advanced materials in biological contexts. Translational advances in material chemistry for healthcare, biotechnology, and environmental sustainability will be highlighted in this topical collection, reflecting the multidisciplinary nature of this rapidly evolving field.
Keywords: Material chemistry, life science, biomaterials, smart materials, drug delivery, nanomaterials, medicinal chemistry.
Publishing Model: Open Access
Deadline: Jun 30, 2026
Material Chemistry in Biomedical Applications
Chemistry-driven processes are essential inspirations for researchers to design biomaterials applied as promising diagnostic tools, therapeutic solutions or tissue substitutes, etc. To increase the understanding of material chemistry in biomedical applications, this topical collection focuses on documenting the chemistry covering nanoscale to macroscale biomaterials, including surface science and interactions with the body at the molecular level. We seek articles that highlight the principles of material chemistry or approaches to investigate and improve the material properties in biomedical research. The material properties here mainly include biocompatibility, physical, chemical, mechanical, and other particular properties required for medical and dental materials. Reviews, original research, and commentaries are all welcomed.
Keywords: Material chemistry, surface science, nanotechnology, biocompatibility, physical and chemical property, mechanical property, biomedical application.
Publishing Model: Open Access
Deadline: Sep 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in