Cones, sticks and croissants: Early cephalopod evolution
Published in Ecology & Evolution

Most people know cephalopods such as squid, octopus and cuttlefish from various books, documentaries, movies, aquaria, snorkelling or perhaps even as food. They are particularly famous for their cognitive abilities compared to other invertebrates – recently, a cephalopod passed the so-called Stanford marshmallow experiment, an intelligence test originally designed for children. Almost equally famous are the fossils of cephalopods, where ammonites and belemnites represent probably some of the most iconic fossils of all time: everybody who went fossil collecting a few times has probably seen at least couple of them. It might thus be surprising that the earliest fossil cephalopods are still poorly understood – and despite the great amount of available material, research on them has been somewhat neglected. In our new study published in BMC Biology, we aim to change that a bit by providing a fresh perspective on the early evolution of cephalopods using quantitative methods. Hopefully this will inspire more research in the future!
Maybe the neglectance of research on early cephalopods was caused by the fossils looking relatively boring on first glance, especially when compared to the sometimes astonishingly beautiful ammonites. Rousseau H. Flower, one of the most prolific researchers on early cephalopods in the past century who single-handedly named more than 400 species, once wrote about endocerids (a group with very long slender straight shells) that “a collection of them seems about as fascinating as a collection of telegraph poles”. As with many things, the true beauty lies hidden beneath the surface. Cutting and polishing fossils of early cephalopods reveals an astonishing diversity of forms in their internal shell structures that essentially served as a buoyancy device. This is also what makes them so fascinating to me personally, because they show so many different morphologies that have no modern analogues.

One important feature to distinguish between different fossil species of externally shelled cephalopods is the siphuncle. This is a tube that runs through their chambered shell and pumps out water to replace it with gas, essentially allowing the animal to become a little submarine, as seen in the only living cephalopod with an external shell, Nautilus. In Nautilus, the siphuncle has a relatively simple shape, as it basically consists of a thin straight tube. However, early cephalopods exhibit a great variety of siphuncle shapes, from thin to very broad and everything between siphuncles with very strongly expanded segments to concave segments and with various strange calcified structures that grew inside the siphuncle or even within the chambers (endosiphuncular and cameral deposits). Add to this the variety in external shell shape that basically ranges from long sticks to cones, croissants, drops, spirals and some others, and you get a large spectrum of different combinations of morphological structures (Fig. 1). And of course, these morphological characteristics had a very direct impact on the lives of these animals, as they directly controlled their swimming capabilities, e.g., the efficiency of buoyancy adjustments, manoeuvrability, swimming speed, etc. This means that there were probably a lot of different lifestyles and ecological roles among early cephalopods. It is hard to imagine how they might have looked like, but some of them may seem really strange to us (Fig. 2).

The big question is here: how did these diverse forms evolve and how are all these groups related to each other? In the past, there have been multiple attempts to solve this, and different evolutionary scenarios have been proposed. The problem was that different researchers focussed on only few, but different characters to reconstruct their evolutionary trees and thus reached different, often contradicting results. In our study, we took a different approach by collecting a large amount of morphological data and analysing them quantitatively with state-of-the-art methods, i.e., Bayesian inference and the so-called Fossilized Birth-Death model. According to our results, early cephalopods diverged quite early into three major groups (Fig. 3): The Orthoceratoidea (mostly slender straight shells with calcified deposits within their chambers and siphuncles), the Multiceratoidea (very disparate shell shapes and thus hard to define, but generally ventrally enlarged muscle attachments and empty siphuncles) and the Endoceratoidea (sometimes very large straight to slightly curved shells, with characteristic conical deposits called endocones within their siphuncles).
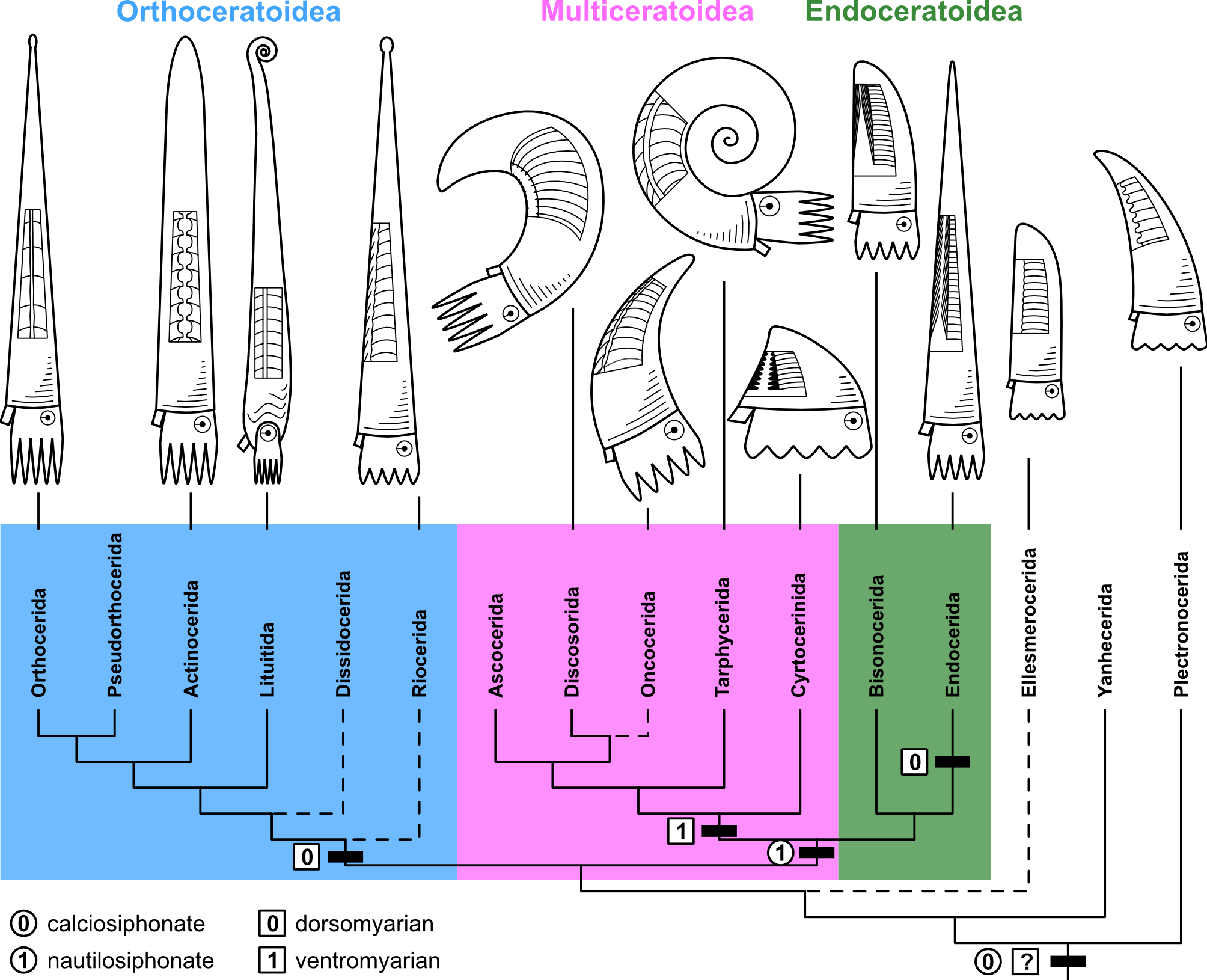
In our paper, we go into detail about each of these groups and the methods. Here, I want to focus a bit more on the real heart of the paper: the morphological character matrix. Building a character matrix from the ground up was a challenging and long process that I started in 2017. Very roughly, my approach was as follows:
- Intensive literature research, reading through countless papers, some of them in Russian or Chinese (luckily, modern translation tools are available and work relatively well for this purpose).
- Defining characters and character states.
- Looking at specimens in Museum collections and the published literature to score characters and measure proportions.
- Realising that I need additional characters or change existing characters to capture some of the variation, thus revisiting all the previously scored species.
- Repeat 3. and 4. many (!) times.
- Check all species again, asking co-authors for their opinions, repeat 3. and 4. again where necessary, final revisions of characters.
As you can imagine, this was a very long process, but also very instructive at the same time and I was repeatedly perplexed by the strangeness of some of these extinct animals. During this entire process, I stumbled upon multiple cases where definitions of characters were problematic (perhaps no wonder, as they were not defined with phylogenetic analysis in mind) or where different names were applied for the same thing. Our paper is thus accompanied by an extensive supplementary material, which discusses every single character in detail and will hopefully prove helpful in future research.
I was lucky to be supported by an awesome, international team of co-authors (Fig. 4) from seven countries (Switzerland, Finland, Germany, United Kingdom, Czech Republic, Argentina and China), which helped a lot in above step 6, but also whenever I had questions or in preparation or revision of the manuscript, so a huge thanks goes to Björn, Rachel, Andy, Dave, Martina, Marcela, Xiang and Christian!

Interested in the full article? Be sure to check it out here:
Early cephalopod evolution clarified through Bayesian phylogenetic inference
Follow the Topic
-
BMC Biology
This is an open access journal publishing outstanding research in all areas of biology, with a publication policy that combines selection for broad interest and importance with a commitment to serving authors well.
Related Collections
With Collections, you can get published faster and increase your visibility.
Cancer metabolism
BMC Biology is calling for submissions to our Collection on Cancer metabolism. Cancer metabolism is a developing field that explores the biochemical and physiological changes that occur in cancer cells, which often exhibit distinct metabolic pathways compared to normal cells. Alterations such as the Warburg effect, where cancer cells preferentially utilize glycolysis for energy production even in the presence of oxygen, play a pivotal role in tumor progression and survival. Understanding the intricacies of cancer metabolism provides insights into how tumors adapt to their microenvironments and highlights potential therapeutic targets for intervention.
Future research in cancer metabolism promises to yield transformative insights that could reshape therapeutic approaches and improve precision medicine. The continued focus on metabolic pathways may lead to the identification of new biomarkers for cancer diagnosis and prognosis, as well as novel strategies to enhance the sensitivity of cancer cells to treatment. As we deepen our understanding of the metabolic landscape of tumors, we may uncover innovative strategies that exploit these vulnerabilities, ultimately leading to novel and more effective cancer treatments, as well as improved patient outcomes.
Recent advancements in the field, including the identification of metabolic reprogramming strategies and the influence of diet on tumor growth, have opened new avenues for research. Investigations into the roles of lipids, fatty acids, and dietary interventions, such as ketogenic diets, are revealing potential methods for manipulating tumor metabolism and enhancing the efficacy of existing treatments. We invite researchers to submit their work to this Collection, which aims to showcase groundbreaking research and technologies addressing cancer metabolism and support the advancement of this field, encompassing a wide array of topics related to metabolic pathways and their implications for cancer biology and therapy.
Potential topics include but are not limited to:
- Metabolic reprogramming in cancer cells
- The Warburg effect and its implications
- Role of mitochondria in cancer metabolism
- Impact of dietary interventions on tumor metabolism
- Glycolysis and lipid metabolism in cancer
- Animal models
- Imaging and method developments
- Metabolic engineering
This Collection supports and amplifies research related to SDG 3 (Good Health and Well-being).
All manuscripts submitted to this journal, including those submitted to collections and special issues, are assessed in line with our editorial policies and the journal’s peer review process. Reviewers and editors are required to declare competing interests and can be excluded from the peer review process if a competing interest exists.
Publishing Model: Open Access
Deadline: Oct 30, 2026
Organelles and molecular assemblies in intercellular signalling
BMC Biology is calling for submissions to our Collection on Organelles and molecular assemblies in intercellular signalling. This Collection aims to highlight the diverse roles of organelles and molecular assemblies in driving cellular and developmental processes. From intracellular organization to tissue-level coordination, these structures are essential for maintaining homeostasis and responding to physiological and pathological cues.
We welcome research that explores how organelles and molecular assemblies contribute to intercellular communication, development, tissue dynamics, and disease—across all biological systems and model organisms.
We welcome studies on exosomes, migrasomes, blebbisomes, mechanosignalling, and the role of organelles—such as mitochondria—in extracellular and intercellular communication. Studies describing the role of cell adhesion molecules, gap junctions, synapses and cell surface receptors in intercellular communication are also encouraged. Studies may also focus on the disruption of secretory pathways and other organelle-mediated processes during disease onset, and their potential as a therapeutic target. Modelling and simulations that investigate and provide significant insights in intercellular communications and emergent properties in physiological contexts are also welcome.
Submissions on novel approaches in imaging, omics-based analyses, and high-throughput analysis to study organelle function and dynamics are particularly welcome.
All manuscripts submitted to this journal, including those submitted to collections and special issues, are assessed in line with our editorial policies and the journal’s peer review process. Reviewers and editors are required to declare competing interests and can be excluded from the peer review process if a competing interest exists.
Publishing Model: Open Access
Deadline: Jul 18, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in