Constructing Molecular Bridge for High-Efficiency and Stable Perovskite Solar Cells based on P3HT
Published in Materials
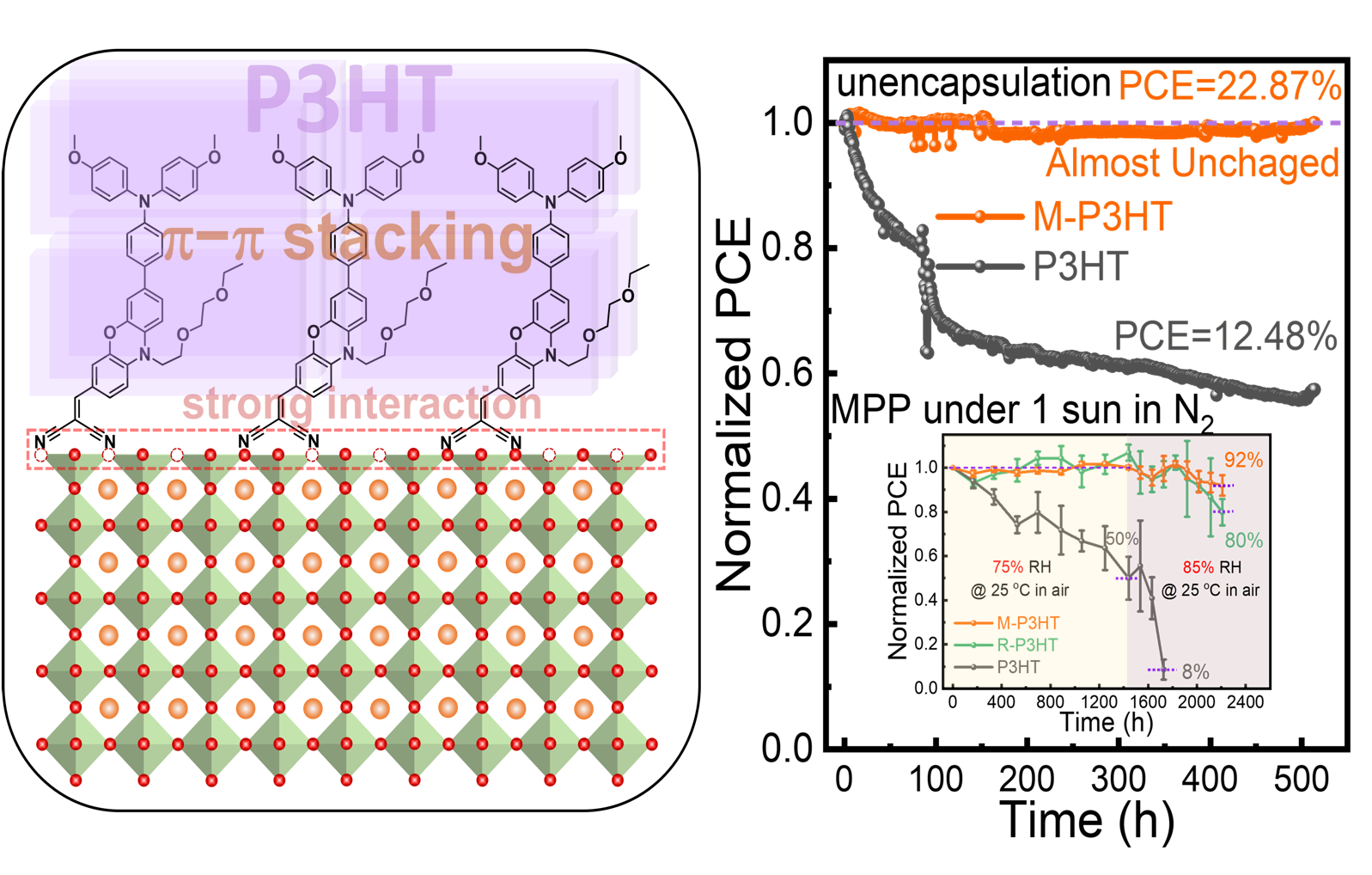
Organic−inorganic hybrid perovskite solar cells (PSCs) with power conversion efficiency (PCE) over 25% is widely considered one of the most promising photovoltaic technologies for its low-cost manufacturing process and appealing photovoltaic performances. In a typical n−i−p type PSC, hole transport material (HTM) is necessary to extract and transport hole to enhance efficiency, and simultaneously block the moisture ingestion to be the last barrier of perovskite degradation. The doped 2,2’,7,7’-tetrakis[N,N-di(4methoxylphenyl)amino]-9,9’-spirobifluorene (Spiro-OMeTAD) with appropriate hole mobility and energetic level alignment is the most commonly used HTM. However, the achievement of high efficiency has been always accompanied by substantial instability arising from the hygroscopicity and fluidity of dopants that can lead to perovskite corrosion and dopant migration, which is detrimental to the long-term stability and commercialization of PSCs.
Thus, utilizing the existing p-type semiconductors with high hole mobility and mature fabrication process, for example, poly (3-hexylthiophene) (P3HT), as HTMs is another significant attempt. Nevertheless, P3HT adopts the "edge-on" stacking arrangement, that is the alkyl side chains directly contact perovskite film, which normally presents an electronically poor contact at perovskite/P3HT interface, which aggravates the non-radiative recombination loss of PSCs. Thus, the pristine P3HT as a HTM can only achieve a PCE as low as 16% in the state-of-the-art PSCs.
Herein, we have introduced a molecular bridge 2-((7-(4-(bis(4-methoxyphenyl)amino)phenyl)-10-(2-(2-ethoxyethoxy)ethyl)-10H-phenoxazin-3-yl)methylene)malononitrile (MDN) to electronically link perovskite films with P3HT. In such bridge, the malononitrile group shows a strong interaction with the Iodine especially iodine vacancies of the perovskite surface to anchor the perovskite and passivates its surface defects,, while the triphenylamine (TPA) group can form stacking with the segments of P3HT. Finally, a PCE of PSCs of 22.87% has been achieved with an enhanced open-circuit voltage (VOC), from 0.88 ± 0.03 to 1.15 ± 0.02, and fill factor (FF), from 52.91% ± 5.14% to 75.02% ± 3.09%. Furthermore, MDN gives the un-encapsulated device enhanced long-term stability that 92% of its initial efficiency maintain even after two months of aging at 75% relative humidity (RH) follow by one month of aging at 85% RH in the atmosphere, and the PCE does not change after operating at the maximum power point (MPP) under 1 sun illumination (~45 oC in N2) over 500 hours. These distinguished results clearly show a bright future for the industrialization of low-cost, stable, and efficient PSCs by utilizing commercial P3HT-based HTMs.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in