Convalescent plasma for lowering mortality in COVID-19 patients – a frenemy?
Published in Biomedical Research
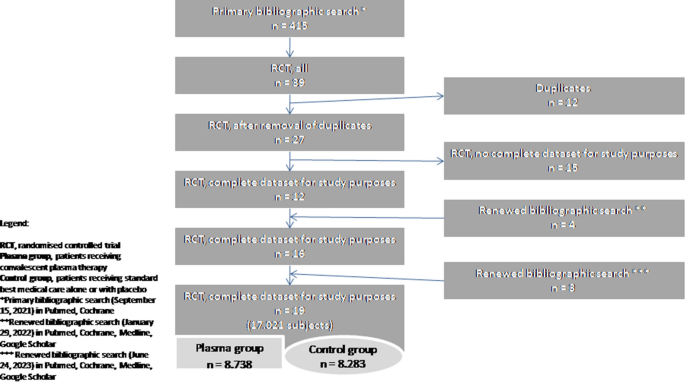
The ambiance of COVID intensive care units in previous years is still present to most clinicians. Healthcare professionals from around the world experienced horrific scenarios of hopelessness and helplessness when treating the most urgent cases without adequate resources. The lack of appropriate medications and rescue treatment modalities led to serious difficulties in curing severely-ill patients, and the lack of treatment success traumatized thousands of professionals. Therefore, in January 2021, our research group decided to launch a study to analyze the effectiveness of treatment methods in reducing the mortality rate of COVID-19 patients. We found controversial results on the efficacy of convalescent plasma therapy for severely-ill COVID-19 patients and therefore began to further analyze the published clinical trials on this topic. We designed a meta-analysis in spring 2021 with the aim to include all relevant high-quality randomized controlled trials focusing on convalescent plasma therapy in COVID-19 patients by that time. The initial literature search was completed in September 2021, but due to the rapidly growing number of publications in this area, we conducted renewed bibliographic searches in January 2022 and June 2023.
We included 19 randomized controlled trials with the overall number of 17,021 patients. As a main result, we did not find a statistically significant difference in all-cause mortality rate between the group of patients who received convalescent plasma transfusion in addition to the best standard medical treatment and the group of patients who received the best standard care alone. However, in patients with coexisting immunodeficiency and long-lasting seronegativity, convalescent plasma therapy appeared to be a useful alternative treatment modality. Moreover, passive immunization could prevent disease progression when administered early in the clinical course.
Our meta-analysis does not support the routine use of convalescent plasma therapy in all COVID-19 patients, but suggests that particularly vulnerable patient populations (e.g., cancer patients, patients with immune disorders, or advanced age) may benefit from passive immunization, especially as postexposure prophylaxis.
Convalescent plasma is an excellent source of polyclonal antibodies and is therefore highly adaptable to rapidly changing viral variants. Potential plasma donors are exposed to a wide range of viral variants, and antibody production depends on the underlying viral infection. This feature of plasma therapy is of great importance not only in SARS-CoV-2 infections but also in other infectious diseases. We consider it highly important to better understand in which patient populations and at what time point in the clinical course passive immunization may be a useful rescue measure. Thus, the message of our study is that administration of convalescent plasma therapy can be a friend in some carefully selected patient populations, but in most cases plasma therapy seems to be a frenemy that does not improve clinical outcome.
Finally, the personal background of our study is of great significance. This work is a result of a two-year collaboration between the authors and represents the first joint publication. Although the authors faced ups and downs several times during the study period, the overall experience of participating in this project, sharing and discussing ideas in this field is an incredible feeling that provides great satisfaction. Being part of a team striving toward a common goal is highly motivating, and we hope that this study was just the first step in the scientific collaboration that opens the door to a long-term partnership between the authors.
Follow the Topic
-
Scientific Reports
An open access journal publishing original research from across all areas of the natural sciences, psychology, medicine and engineering.
Related Collections
With Collections, you can get published faster and increase your visibility.
Computational biology and mathematical modelling of biological systems
Publishing Model: Open Access
Deadline: Jul 18, 2026
Water pollution and advanced treatment processes
Publishing Model: Hybrid
Deadline: May 31, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in