Dicarboxylation of alkenes, allenes, and (hetero)arenes with CO2 via visible-light photoredox catalysis
Published in Chemistry
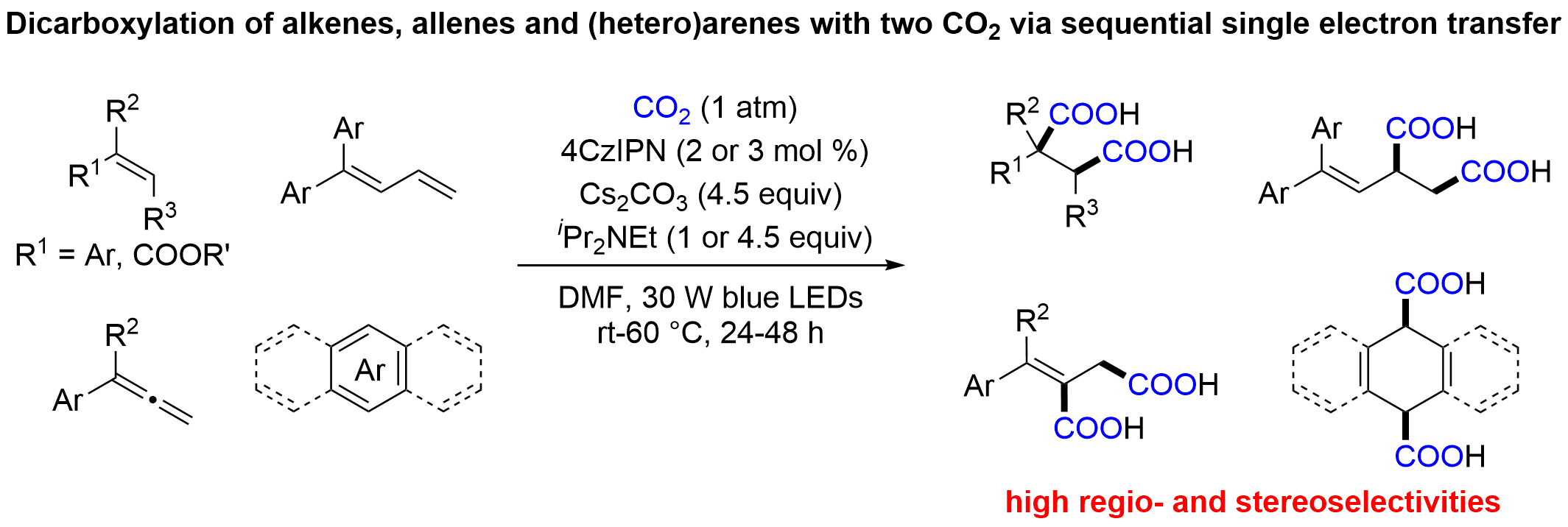
Catalytic difunctionalization of alkenes with CO2 via visible light photoredox catalysis is mainly limited to the addition of free radicals1 or CO2 radical cation2 to the alkenes to generate new carbon radicals intermediate. However, the visible light-driven difunctionalization of alkenes with CO2 via radical anions of alkenes still remains unknown. Herein, we report a successive single electron transfer (SSET) strategy for the dicarboxylation of alkenes, allenes with two molecules of CO2 via radical anion intermediate, providing a simple and sustainable approach to valuable succinic acids. Moreover, diverse (hetero)arenes also are suitable substrates to undergo the dearomative dicarboxylation with high regio- and diastereoselectivities.
The research group of Prof. Da-Gang Yu at Sichuan University has been focusing on developing new strategies for CO2 utilization and visible-light photoredox catalysis in the past few years. In the area of carboxylation with CO2 via SSET process, we have developed the selective and catalytic hydrocarboxylation of enamides and imines with CO2 to generate α,α-disubstituted ɑ-amino acids (ACIE 2018, 57, 13897), cross-electrophile couplings of tetraalkyl ammonium salts with CO2 without external reductant (JACS 2018, 140, 17338), ring-opening carboxylation of cyclic oxime esters with CO2 (ChemSusChem 2020, 13, 6312), as well as reductive dearomative arylcarboxylation of indoles with CO2 (Nat. Commun. 2020, 11, 3263). Very recently, we have developed a novel strategy for dicarboxylation of alkenes, allenes, and (hetero)arenes with CO2 via radical anions of such unsaturated substrates with SSET process, as shown below (Nat. Catal. 2021, DOI : 10.1038/s41929-021-00594-1).
Electrochemical dicarboxylation of alkenes have been reported, but unfortunately, the high electrochemical potentials (-2 to -2.5 V vs SHE) for practical current densities leading to narrow substrate socpe and low selectivity3. In the area of visible-light photoredox-catalyzed carboxylations with CO2, our group and others4 have reported many examples to the synthesis of industrially relevant carboxylic acids, however, these carboxylation protocols remain currently confined to single CO2 insertion. This situation might arise from the high reduction potential of alkenes and (hetero)arenes (for styrene, Ered = -2.91 V vs Ag/Ag+)5 and competitive side reactions, including reduction6, reductive homocoupling7, hydrocarboxylation8. As we developed highly reductive system for reductive carboxylation of imines and indoles with CO2, we considered that whether alkenes also could be reduced and then react with two CO2 through SSET process. With these challenges and the strategy in mind, we further realized dicarboxylation of alkenes, allenes, and (hetero)arenes with CO2 via radical anion intermediate with SSET process. Notably, this method realized multiple C-C bond formations with high chemo- and diastereoselectivity under mild conditions. Moreover, this transition-metal-free protocol exhibits low catalyst loading, good functional group tolerance, broad substrate scope, facile scalability and easy product derivatizations to drug and material molecules. The Stern-Volmer quenching studies, UV−Vis monitoring studies and control experiments all suggested alkene radical anion was the key intermediate. Deuterium-labeling control experiments also suggested the generation of a benzylic carbanion. These studies indicated a pathway by SSET via radical anions of such unsaturated substrates, broadening the repertoire of strategies for CO2 fixation.
More details of this work could be found here: “Dicarboxylation of alkenes, allenes, and (hetero)arenes with CO2 via visible-light photoredox catalysis” in Nature Catalysis (https://www.nature.com/articles/s41929-021-00594-1).
References
- Yatham, V. R., Shen, Y. & Martin, R. Catalytic intermolecular dicarbofunctionalization of styrenes with CO2 and radical precursors. Chem., Int. Ed. 56, 10915-10919 (2017).
- Ye, J.-H. et al. Visible-light-driven iron-promoted thiocarboxylation of styrenes and acrylates with CO2. Chem. Int. Ed. 56, 15416-15420 (2017).
- Yuan, G.-Q., Jiang, H.-F., Lin, C. & Liao, S.-J., Efficient electrochemical synthesis of 2-arylsuccinic acids from CO2 and aryl-substituted alkenes with nickel as the cathode. Electrochimica Acta 53, 2170-2176 (2008).
- Zhang, Z. et al. Visible-light-driven catalytic reductive carboxylation with CO2. ACS Catal. 10, 10871-10885 (2020).
- Senboku, H., Komatsu, H., Fujimura, Y. & Tokuda, M. Efficient electrochemical dicarboxylation of phenyl-substituted alkenes: synthesis of 1-phenylalkane-1,2-dicarboxylic acids. Synlett 3, 418-420 (2001).
- Pac, C., Ihama, M., Yasuda, M., Miyauchi, Y. & Sakurai, H. Tris(2,2'-bipyridine)ruthenium(2+)-mediated photoreduction of olefins with 1-benzyl-1,4-dihydronicotinamide: a mechanistic probe for electron-transfer reactions of NAD(P)H-model compounds. Am. Chem. Soc. 103, 6495-6497 (1981).
- Fruianu, M., Marchetti, M., Melloni, G., Sanna, G. & Seeber, R. Electrochemical reduction of 1,1-diaryl-substituted ethenes in dimethylformamide. Chem. Soc., Perkin Trans. 2, 2039-2044 (1994).
- Huang, H. et al. Visible light-driven anti-markovnikov hydrocarboxylation of acrylates and styrenes with CO2. CCS Chem. 2, 1746-1756 (2020).
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in