Discovery of a natural IgA human monoclonal antibody against Mtb protein LpqH and its significance in TB prophylaxis
Published in Microbiology
Tuberculosis (TB) is a life-threatening airborne infectious disease caused by the intracellular pathogen Mycobacterium tuberculosis (Mtb). TB has become a global health concern, with a quarter of the global population being latently infected and 10 million active infections annually. The advent and global spread of multidrug-resistant Mtb represents a significant challenge for current antibiotic treatment regimes. Under these circumstances, improved vaccination and/or alternative therapeutic approaches are vital to combat the global disease burden. A better understanding of natural immune correlates of protection is vital to enable this. Several cohort and vaccine studies provide a compelling argument for the role of antibody response, in the prevention and control of Mtb infection via mucosal immunity. Thus, a detailed analysis of antibody repertoires against Mtb, particularly from asymptomatic individuals in high Mtb exposure settings, can provide important information on antibody-mediated protective immunity. This would help us gain insights into immune determinants and immune response mechanisms mediating protection against this pathogen. The identification of epitopes and immune determinants that stimulate protective antibodies against Mtb from a natural infection would have significant implications for vaccine development strategies. The isolation and characterization of these natural human monoclonal antibodies may also open avenues for new TB therapeutic/prophylactic candidates.
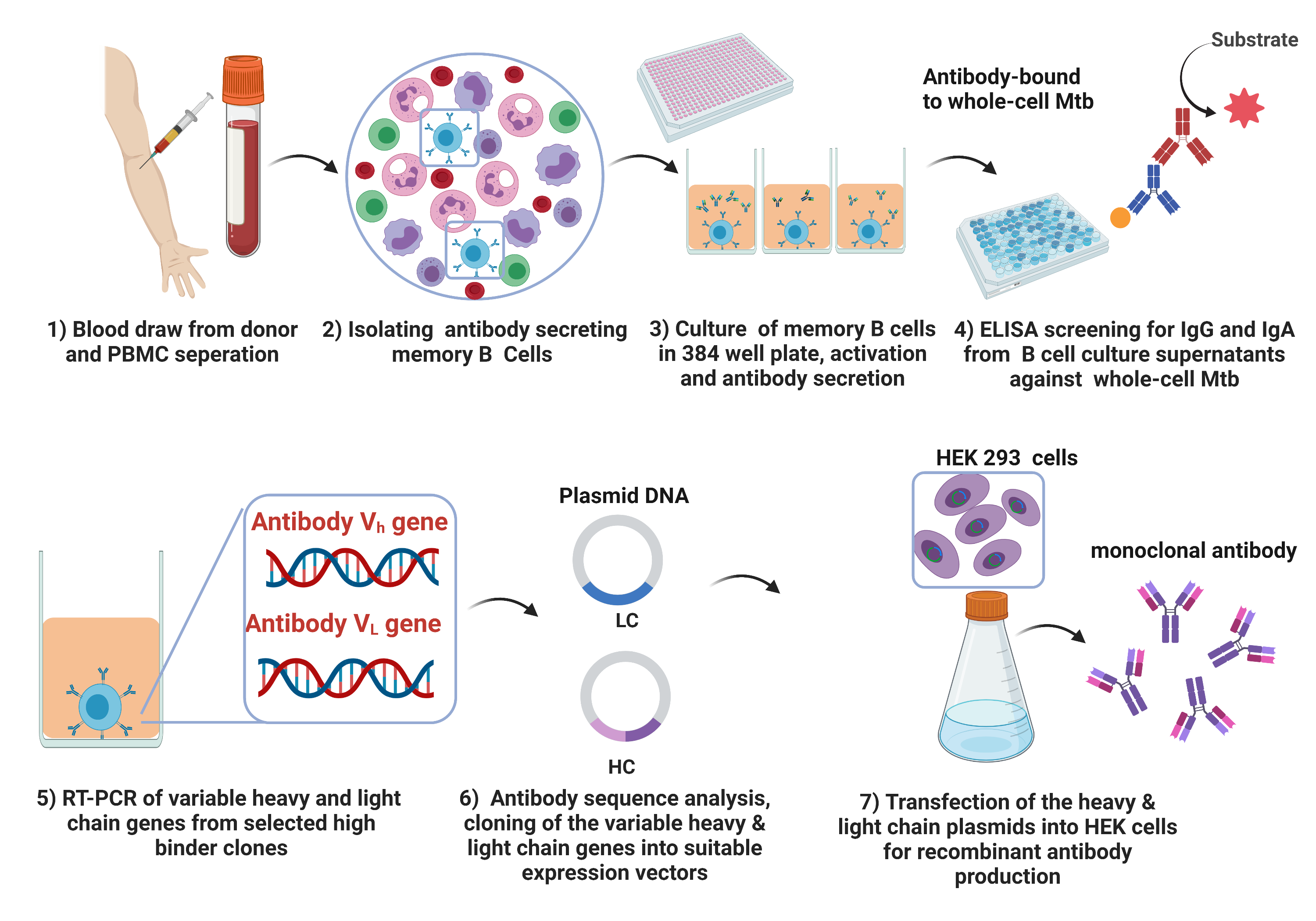
The aim of our study was to develop fully human monoclonal antibodies against whole-cell Mtb from a cohort of asymptomatic TB healthcare workers in a high Mtb exposure setting in China and conduct a thorough characterization of the isolated antibodies. A high-throughput IgG and IgA screening from the donor memory B cell repertoire was performed, using gamma-irradiated whole-cell Mtb as the target antigen (Fig 1). From the IgA/IgG repertoire, four unique human monoclonal antibodies targeting Mtb surface antigens were isolated from our occupationally exposed immune volunteers.
Our recent paper describes the identification of a human monoclonal antibody HuMab-28-009 targeting a major virulence factor LpqH. This antibody was isolated from the IgA repertoire of an immune donor, demonstrating a moderate protective response against Mtb. The human IgG2 subclass of HuMab-28-009, showed significant protection against Mtb in vivo and in vitro. This further supports the proposed anti-bacterial role of the IgG2 subclass in mediating pro-inflammatory cytokine responses. The study further demonstrates a protective role for murine IgA isotype of HuMab-28-009. These findings could have significant implications in designing future prophylactic or therapeutic antibody cocktails in TB disease management. The identification of a natural protective antibody targeting LpqH, a major virulence factor, from an unbiased screening using whole-cell Mtb, implicates LpqH as an important immune correlate of protection. This interesting discovery could pave the way for future vaccine development strategies. These findings were also suggestive of the role of such mucosal antibodies in early host defense and have important implications in our understanding of natural mucosal immunity against Mtb infection.
Follow the Topic
-
npj Vaccines
A multidisciplinary journal that is dedicated to publishing the finest and high-quality research and development on human and veterinary vaccines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Therapeutic HPV vaccines
Publishing Model: Open Access
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in