Proteins acting as Electrical Plugs to Charge Microbial Nanowires
Published in Chemistry, Microbiology, and Protocols & Methods
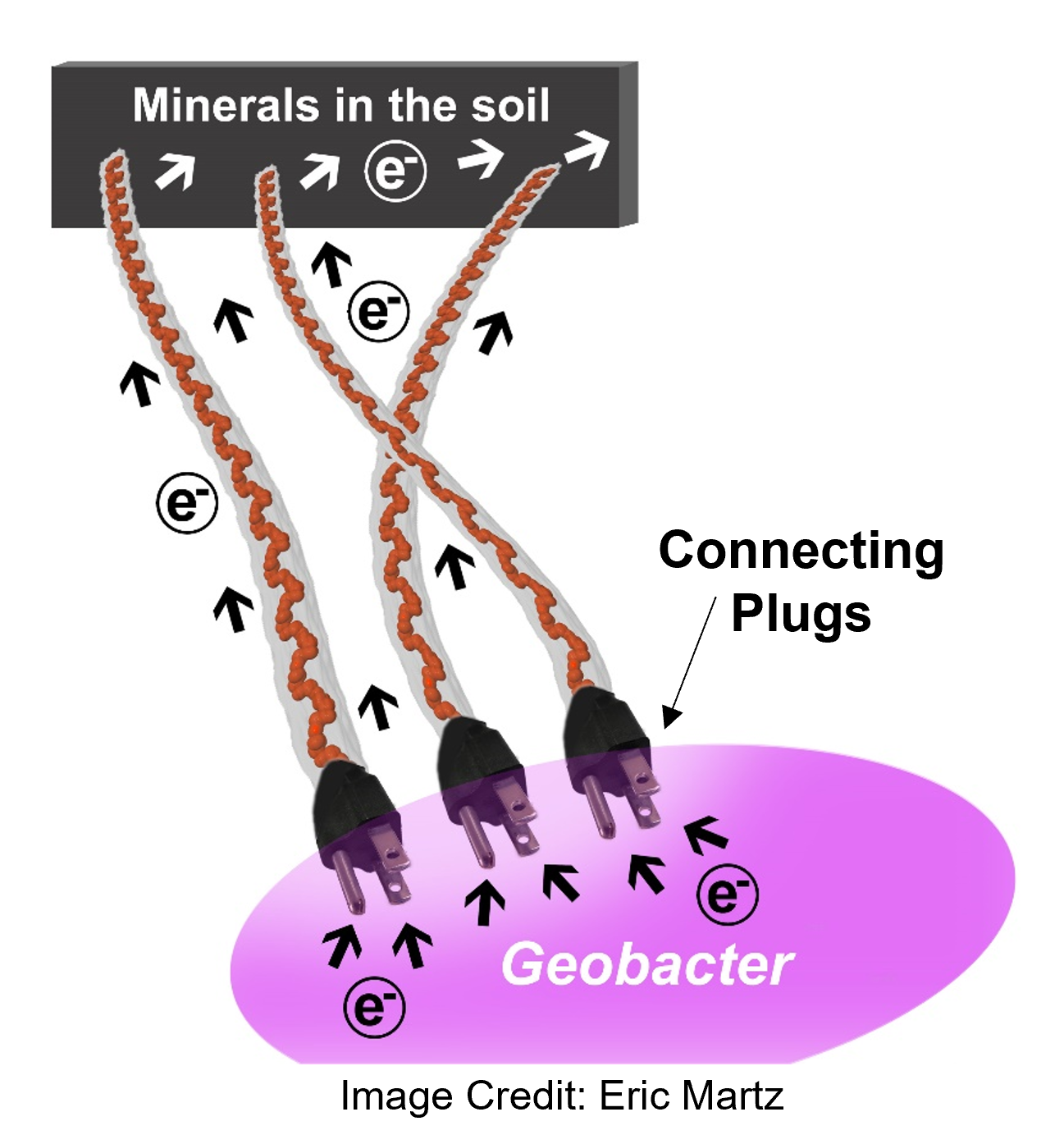
What we found, and why is it important? We found that microbial nanowires are charged by a single protein family acting as electrically connecting "plugs" to inject electrons into them, as shown in the picture below. This five-protein family remains inside the bacterial periplasm, hence the name periplasmic cytochrome (Ppc)ABCDE. The family injects electrons into filaments on bacterial surfaces that act as "nanowires." This electric connection allows metal-breathing Geobacter to dispose of excess electrons produced during metabolism to minerals in the soils without needing oxygen-like soluble, membrane-ingestible electron acceptors.
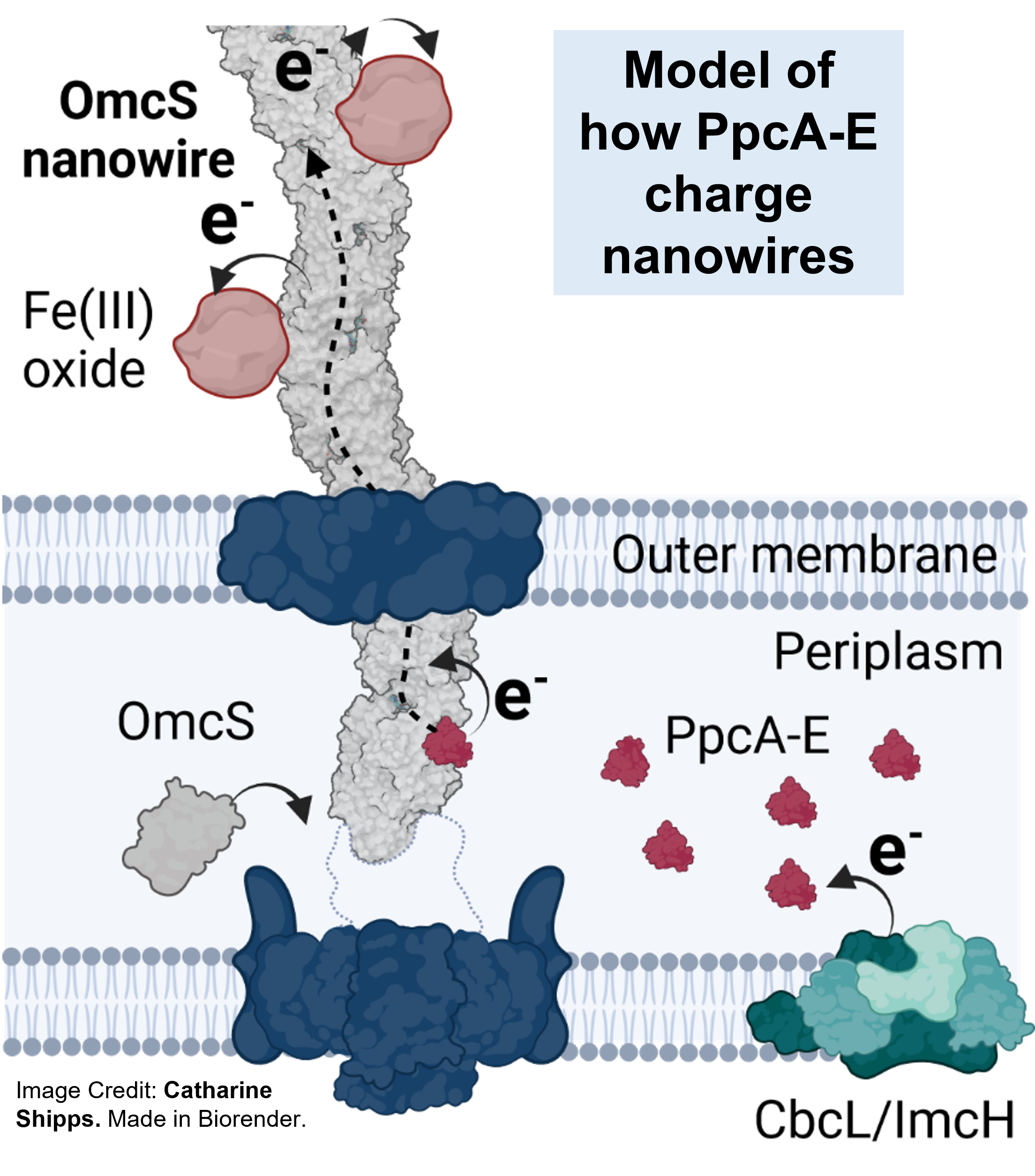
In 2002, filaments were first observed on the bacteria during electron transfer to minerals. However, until recently, they were thought to be made up of pili proteins (pili means hairs in Latin) because many bacteria do have pili on their surface, and all the genetic data suggested a similar role in Geobacter. In 2021, my lab solved the atomic structure of pili and showed that pili instead act as pistons to push filaments made up of cytochromes. Atomic structures of these cytochromes OmcS and OmcZ showed a chain of metal-containing heme molecules that carry electrons (red in the schematic).
Although these atomic structures explain how electrons move through nanowires, how bacteria move electrons to the nanowire remains a mystery, as most cell surfaces are electronically non-conducting. It was thought that another family of proteins embedded in the membrane called "porin cytochromes" connected to nanowires despite bacteria being able to transmit electricity even in their absence. Periplasmic proteins themselves transferring electrons to nanowires eliminates the need for any intermediate electron carriers and explains how cells transmit electrons at a remarkably fast rate (1 million electrons per second) despite electrons in proteins being able to move at rates at least 10 times slower.
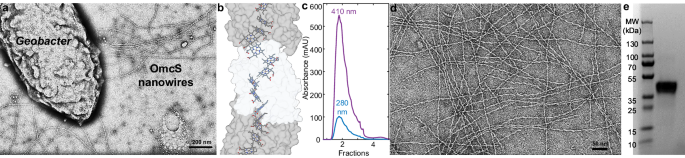
Why did we do what we did? When PhD student Catharine Shipps measured the redox potential in OmcS nanowires, she found it like PpcA-E. This was very surprising because, since 2011, OmcS's redox potential was thought to be 0.1 V different. However, at that time, it was not known that OmcS formed nanowires, and the measurements were made by treating them as non-filamentous, which could explain this large discrepancy. Although Salgueiro's group had long been interested in testing PpcA-E's electron transfer to OmcS, it was not feasible to test this possibility due to the limitation in obtaining purified OmcS nanowires at high yields.
Catharine's finding, complemented by studies of another graduate student, Vishok Srikanth, who developed the OmcS purification method and noticed that both OmcS and PpcA-E stay together when extracted from the bacteria, opened a route to test the hypothesis. Furthermore, a study by the Salgueiro group showed that all PpcA-E can couple electrons and protons, a necessary condition for energy generation. This data made us reinforce in a review that PpcA-E could pass electrons to nanowires. To test this hypothesis, we collaborated with the Salgueiro group. A PhD student, Pilar Portela, purified nanowires with Catharine and Vishok, and unambiguously demonstrated the binding and electron injection of PpcA-E to OmcS using painstaking Nuclear Magnetic Resonance spectroscopy.
Implications and Applications. The discovery that just two proteins, OmcS nanowires and one of PpcA-E, are sufficient to wire the inside of the bacteria to the outside greatly simplifies the model of how these bacteria export electrons by overcoming the slow electron flow among individual proteins. Then, Ph.D. student Cong Shen discovered that this protein family is evolutionarily conserved across many species, not just Geobacter, and OmcS and PpcA-E co-occur in all these species. Thus, this minimal wiring machinery is ubiquitous!
Next Steps. We are now engineering this newly discovered machinery into other bacteria important to our climate or capable of making biofuels to make them grow faster. We are also working on how another nanowire of cytochrome OmcZ is charged and identifying the role of porin-cytochromes in these processes.

Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in