Enhancing Diabetes Treatment with a Biomimetic Co-formulation of Insulin and Amylin Analogues
Published in Bioengineering & Biotechnology
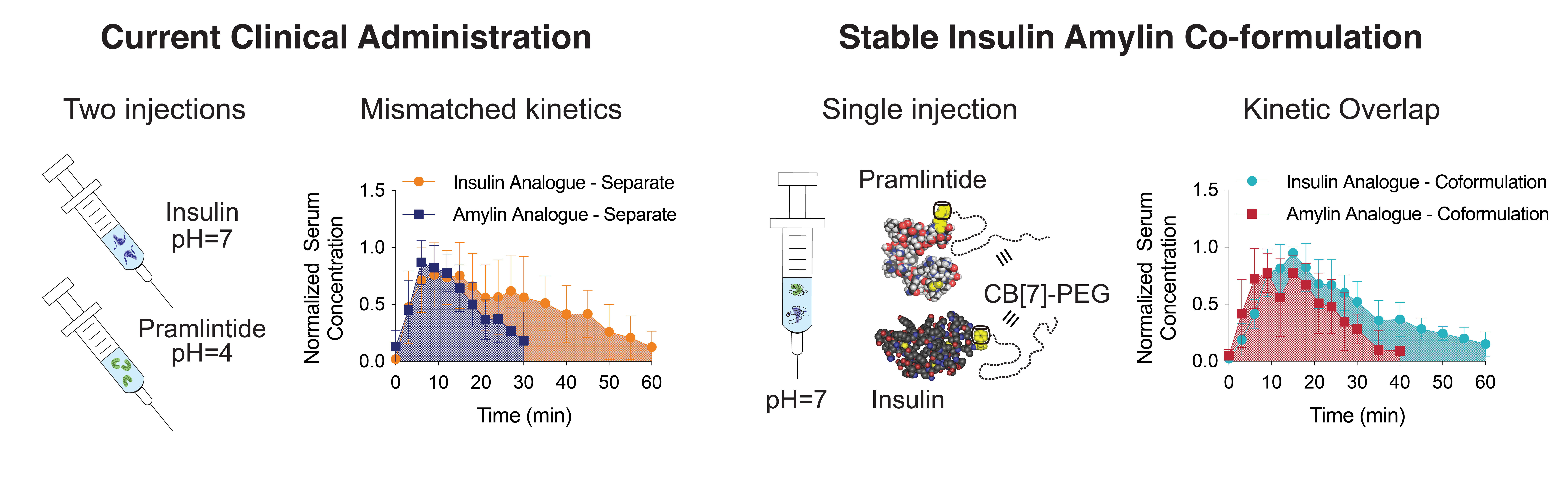
Insulin has been the focus of diabetes treatment for almost 100 years. In contrast, a hormone called amylin, which is co-secreted alongside insulin from healthy beta cells in the pancreas, has been largely overlooked. Amylin acts synergistically with insulin to slow gastric emptying, suppress glucagon and reduce appetite at mealtimes. While treatment of diabetes with insulin and amylin analogues (pramlintide) has been shown to be more effective than insulin alone, only 1.5% of rapid-acting insulin users have adopted pramlintide because of the burdensome requirement for a separate administration. An insulin pramlintide co-formulation would reduce patient burden and enable more widespread adoption of this treatment for improved glycemic control.
This project was born in 2016 at a new faculty lunch at Stanford University through a casual conversation about past work with Professor of Pediatrics and Division Chief of Pediatric Endocrinology, Dr. David Maahs. This chat turned into a fruitful collaboration with Dr. David Maahs and Professor of Pediatrics Dr. Bruce Buckingham. A primary focus of our lab is the design and evaluation of systems that can more efficiently and effectively deliver active drug compounds to where they need to be in the body, when they need to be there. Through discussion with Dr. Maahs and Dr. Buckingham, we identified that pramlintide is rapidly destabilized and loses activity when formulated at pH~7, the pH of most insulin formulations used around the world. This pH mismatch has necessitated the administration of pramlintide as a separate injection from insulin and renders it incompatible with insulin infusion pumps. Moreover, unlike co-secretion from healthy pancreatic beta cells, the absorption kinetics of these two hormones is substantially different when delivered subcutaneously in separate injections, which limits their synergistic effects. We set out to design a stable co-formulation of insulin and pramlintide that also increases the overlap in absorption kinetics following subcutaneous delivery to more closely mimic the endogenous co-secretion of insulin and amylin from a healthy pancreas.
Here we showcase the use of non-covalent interactions between cucurbit[7]-uril and the terminal aromatic amino acids occurring naturally on insulin and pramlintide to dynamically attach a protective polymer, poly(ethylene glycol), to the proteins to simultaneously stabilize them both in co-formulation. When the concentration of this “designer excipient” and insulin is high in formulation, over 98% of the protein-excipient complexes will be bound and stabilized. Yet, these complexes will rapidly dissociate upon dilution when injected into the body, allowing the unmodified proteins to be free to act. Further, these dynamic polymer interactions temporarily alter the molecular weight of insulin and pramlintide so that they diffuse, and therefore are absorbed, at more similar rates than when they are administered separately. We saw these that insulin and pramlintide exhibit increased overlap in their time-frame of action in both diabetic rat and pig models when administered in our co-formulation than in separate injections according to current clinical practice. This enhanced overlap in activity also resulted in increased synergy between the two hormones, improving glucagon suppression at mealtimes in diabetic pigs. This approach more closely mimics endogenous co-secretion of insulin and amylin by the pancreas and shows promise as a dual-hormone replacement therapy.
Follow the Topic
-
Nature Biomedical Engineering
This journal aspires to become the most prominent publishing venue in biomedical engineering by bringing together the most important advances in the discipline, enhancing their visibility, and providing overviews of the state of the art in each field.
Related Collections
With Collections, you can get published faster and increase your visibility.
Implantable wireless communication technologies
Publishing Model: Hybrid
Deadline: Nov 28, 2026
Medical Ultrasound: Emerging Techniques and Applications
Publishing Model: Hybrid
Deadline: Jan 29, 2027




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in