Exosomes release: An additional back-up system in pathological cardiac remodeling?
Published in Healthcare & Nursing
Stress conditions have proven to have a negative impact on cardiomyocytes in aspects such as oxygen demand, vasculature growth, and cellular communication. These processes lead to changes in transcriptional profiles with the aim of maintaining homeostasis. Despite these efforts, homeostasis is not guaranteed due to limited cellular adaptation and lack of regenerative capacity of adult cardiomyocytes resulting in cellular loss. Thus, this process is characterized by a combination of beneficial and detrimental mechanisms. In an attempt to gain a deep insight into this process, we compared changes in gene expression level at single-cell resolution in mouse models of pathological remodeling including a transgenic mouse with Wnt signaling activation, a hallmark of pathological remodeling, and an experimental induced hypertrophic model.
After confirming the stress signature within the cardiomyocytes' transgenic model, our analysis focused on the enriched biological processes, which revealed that ubiquitination and secretion of protein involving vesicles, among others, were enriched in the disease model. That gave us the kickstart for our search. In order to investigate the role of extracellular vesicles (EVs) in cardiac remodeling, we adapted a published protocol for EVs isolation of heart tissue. After obtaining and validating the nature of these nanoparticles as exosomal vesicles, mass spectrometry analysis of the protein content revealed 391 enriched proteins in our transgenic model. Diseased hearts' EVs were significantly enriched for all seven α and seven β chains of the 20S proteasome components. We also observed the enrichment of molecular chaperones, co-chaperones, and protein-quality-control-associated proteins and confirmed increased ubiquitinated proteins contained in these vesicles. Of all these enriched proteins, one called our attention: CRYAB. This is a small heat shock protein that triages misfolded proteins for proteasomal degradation or repair in cardiomyopathy. Since CRYAB is associated with the Z disk and shows close interactions among the 20S core proteins, other chaperones, and cardiac proteins; data suggests shared biological processes.
These findings encouraged us to further investigate whether pathological stress caused by aging and cardiac hypertrophy in non-genetic modified cardiomyocytes were characterized by similar mechanisms. Following that line, we subjected aged mice hearts to pressure overload by trans-aortic constriction and analyzed the transcriptional landscape at single-cell resolution. As previously demonstrated (Iyer et al. NAR, 2018), many similarities between the transgenic Wnt activated and the non-transgenic disease model were observed when compared with their respective controls. One striking finding was that exosomal markers and CRYAB were significantly upregulated in all cardiomyocyte clusters at an early stage of remodeling with a reduced differential expression at late remodeling.
Naturally, we asked ourselves if the identified mechanism could be reproduced in human cells. Therefore, we derived cardiomyocytes from human induced pluripotent stem cells (iPSCs) and triggered stress by activating Wnt signaling to mimic our in vivo model and isolated secreted EVs. Similar to the in vivo findings, we observed increased levels of ubiquitinated proteins in the disease iPSCs-derived cardiomyocytes model. Moreover, a clear perinuclear accumulation of CRYAB in cardiac Troponin 2 (TNNT2)-positive cells was observed, which was in line with our in vivo findings.
The above-mentioned findings tempt us to speculate that cellular stress induces protein misfolding, leading to an excess of ubiquitination and activation of chaperone-mediated processes, which are redirected as exosome release. This may represent an additional “backup” system, by which cells alleviate the accumulation of intracellular aggregate-prone or misfolded proteins and deserve further investigation in different disease contexts.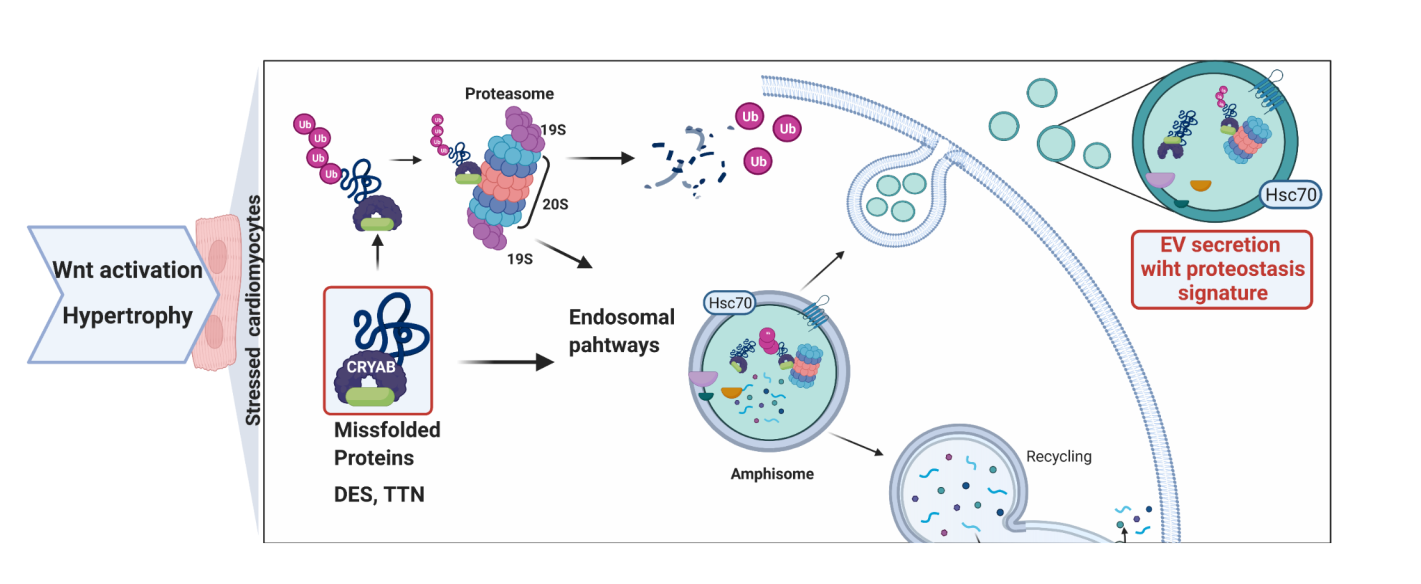
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
DNA repair and human disease
Publishing Model: Hybrid
Deadline: Oct 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in