Colloidal Suspensions: A Fascinating Dive into Tiny Particles and Big Discoveries
Have you ever wondered what's inside that milk you pour over your cereal, the toothpaste you brush with, or the ink you write with? These everyday items might seem simple, but they hide a complex world of particles in fluid: colloidal suspensions. Minuscule particles in such suspensions, each a hundred times smaller than the thickness of a human hair, float and move freely within the fluid. The particles can be made from diverse materials, from metals and polymers, as well as substances derived from living organisms. Interestingly, these so-called colloids can serve as an excellent model system for researchers. Because of their size and visibility under optical microscopes, scientists can directly study their behaviour, making them a useful tool for understanding systems and processes that are more challenging to observe directly. This unique aspect of colloidal study has led us to insights about many phenomena, for example, the difference between glasses and crystals.
The behaviour of these particle suspensions in confined spaces holds significant interest. When particles are confined, they “live” in conditions found in biological media, such as blood cells in narrow vessels or proteins in membranes. Since these particles float in the fluid, they are coupled by interactions mediated by their host solvent, and they are not free to move everywhere. Why does this matter? Imagine a crowded street; in the street, each person's movement is influenced by both their intention but also by the flow of the crowd around them. Similarly, the particles in the fluid experience solvent-mediated interactions. Called hydrodynamics, these interactions play a pivotal role affecting particle motion. Understanding these interactions is a basic problem in physics that dates back to Einstein’s work on Brownian motion and holds influence in applications ranging from the delivery of drugs within our body to advanced materials processing.
Probing Colloidal Fluids in Confined Geometries
Our experiments were performed using a microscope and a video camera: optical video microscopy. A monolayer of colloidal particles in suspension was confined between two glass coverslips, forming a nearly two-dimensional (2D) sample cell. Videos were captured of particles in the experimental field-of-view and were analysed. The position-versus-time trajectories of each particle were thus collected and correlated, and the correlations between individual trajectories revealed hydrodynamic forces between two particles, that is, they revealed the interactions between particles mediated through the fluid.
Deciphering Fluid Mediated Interactions from Movement of the Particles
As we analysed the trajectory data, we made a discovery: the hydrodynamic interactions between particles in the confined space were reminiscent of the dipolar field lines (Fig. 1, left) found for simple magnets. In layman's terms, each particle creates a kind of "hydrodynamic magnet" around itself, setting off a cascade of interconnected movements. Rather than meandering aimlessly, every particle's path is shaped by the movement of particles near it.
Specifically, zooming in on the dynamics of two particles, if one particle moves along the imaginary line connecting them, it essentially drags its partner along (Fig. 1, right). Conversely, if the particle moves perpendicular to the connecting line, an opposing force kicks in, causing the particles to drift apart. These effects diminish as the distance between the particles increases but are very important at close range. Further examination leads to even more interesting insights. There is a well-known rule in physics called the Stokes-Einstein relation (SER). This relation connects a fluid's viscosity to the movements of tiny particles suspended in it. We found that this relation is different for particles moving in confined spaces, due to hydrodynamics. Now, you might wonder, why is this important? The SER makes the prediction of the motion of minuscule particles possible, based only on the viscosity of the host liquid. To underscore its importance, imagine implications in medical science, specifically in drug delivery. Visualize a drug-carrier designed to manoeuvre through the bloodstream and deliver its therapeutic agent at a specific site, say a tumour. While the drug's passage through larger vessels might be predicted using the SER, when it enters the tighter spaces of capillaries and the interstitial fluid around cells, its behaviour deviates from that of standard SER. If the carrier particle is in close proximity to other particles or the vessel walls, the breakdown of the SER implies it might move slower (or faster) than expected, perhaps even stalling at certain points based on local constraints. Thus, full understanding of the SER in different regimes could help explain the efficiency for a drug to reach its target.
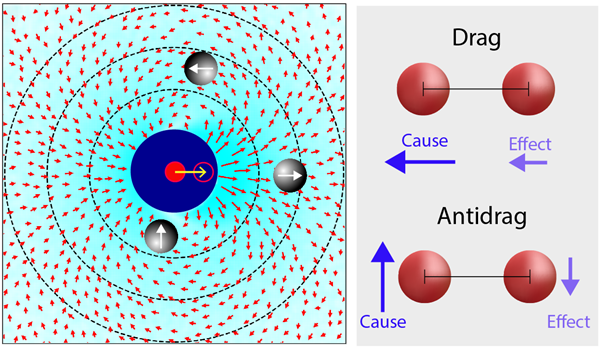
Figure 1: Hydrodynamics in confined 2D (left), experimentally determined map showing average response (red arrows) of particles at all due to the motion of a particle from red solid to hollow circle. Schematic of drag and antidrag motion of particles (right). In the drag scenario, both arrows signifying particle displacement point in the same direction, illustrating the synchrony in particle movement. In contrast, the antidrag demonstrates opposing movements, where the particles diverge from each other.
From Microscope to Real World: Beyond the Lab
Our study offers a lens through which we can understand and predict the movement and interactions of minuscule particles suspended in solvents within confined environments. We pinpointed the causes for the breakdown of SER and outlined simple conditions wherein SER is recovered. The research is useful for the development of analytic and simulation models of complex fluids and sets the stage for diving deeper to elucidate the hydrodynamics of anisotropic particles, be they passive or active particles. The ideas could also be useful for devising methods of assembling particles or even sorting them when space is tight.
Follow the Topic
-
Nature Communications

An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Mar 24, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in