Exploring SHANK3 in Neurodevelopment Using Stem Cells and Chemical Biology
Published in Neuroscience, Cell & Molecular Biology, and General & Internal Medicine
SHANK3 in neurodevelopment
At Ksilink, a French biotech, we have a long-standing interest in understanding and contributing to drug discovery and development for neurodevelopmental diseases together with partners from industry and academia. We are always on the lookout for diseases with an unmet medical need and unclear pathological mechanism and started to work on Phelan-McDermid syndrome (PMDS) in 2019. PMDS is a rare genetic disorder characterized by developmental delay, hypotonia, intellectual disability, speech impairments, and is considered part of the autism spectrum disorders (ASDs). In most PMDS affected individuals, the terminal region of chromosome 22q13 is impacted by point mutations or larger deletions, resulting in a haploinsufficiency of the SHANK3 protein, meaning that about half of the normal SHANK3 protein content is missing in cells.
SHANK3 is a key protein in the brain. It is located in synapses and acts as a scaffold for many binding partners, ensuring synaptic stability and functionality. In addition to its role in synapses, SHANK3 is also involved in synapse formation and the maturation of dendritic spines. During the development of neurons, SHANK3 helps organize excitatory synapses. It is also involved in various stages of neuronal development, influencing the area of growth cones, the length of neurites, and the number of neurite branches 1. SHANK3 is therefore a crucial player in both the development of neurons and the function of synapses. Despite their genetic heterogeneity, in fact many ASDs involve synaptic proteins with roles in development or circuit formation such as Neuroligin-1 (NLGN1), Neuroligin-4 (NLGN4X), or Neurexin-1 (NRXN1) 2. Synaptic dysfunction during development might therefore represent an underlying mechanism for a larger subset of ASDs 3 (Figure 1).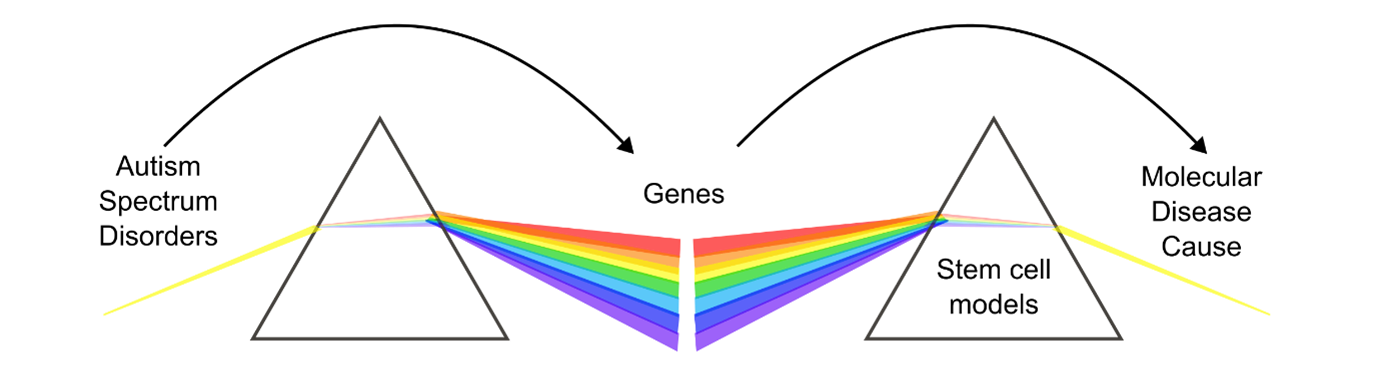
Figure 1: Autism Spectrum Disorders (ASDs) are genetically heterogenous, but stem cell models might help to identify commonalities between genes and their molecular disease cause, for example the role of synaptic proteins during neuronal development.
In the case of PMDS and SHANK3, two treatment strategies can be imagined: 1) Increasing the expression of SHANK3 protein to improve neural function thereby alleviating autistic-like traits and 2) restoring biochemical pathways linked to SHANK3 to rescue related phenotypes. With these approaches in mind, we began the project and sought experienced partners in PMDS modelling.
From Inspiration to Challenges
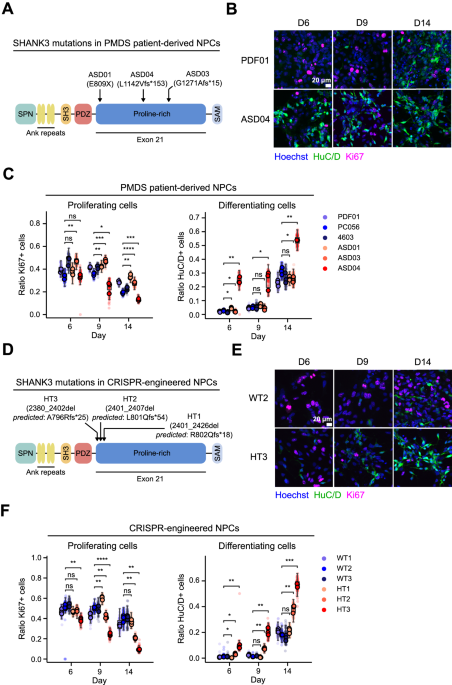
Soon after we met Alexandra Benchoua at ISTEM, Paris, a passionate neuroscience researcher with expertise in the cellular modelling of autism. In 2016 she and her team had screened a library of compounds for their potential to increase SHANK3 mRNA content in neurons differentiated from control human embryonic stem cells. The active compounds were then tested for their efficacy in correcting dysfunctional networks of neurons differentiated from individuals with deleterious point mutations in SHANK3. Lithium and valproic acid showed the best efficacy. One patient was treated with lithium, and after one year, a significant decrease in autism severity was observed 4. This suggested that stem cell-derived neurons could be a valuable tool in the search for specific disease-modifying treatments for ASD. We therefore decided to bring together Ksilink’s expertise in assay automation, drug screening, and cellular imaging with Alexandra’s expertise in modelling neurodevelopmental diseases. Although we faced important technical obstacles on the way, this was the beginning of a fruitful collaboration. One of the major challenges was making the link between heterogenous patient-derived cell lines and the CRISPR-edited cell lines with engineered SHANK3 mutations that we used for screening. Finding the right assay time point was another hurdle - striking a balance between technical feasibility and physiological relevance. Prioritizing molecules and then investigating a possible mode of action was another tough choice to make. We decided to focus on the actin remodeler ARPC2 due to previous reports linking SHANK3 to actin-dependent neuronal development.
Stem Cell-derived Neurons for Drug Discovery
After having worked on this and similar projects, we are convinced that human stem cells have the potential to accelerate the drug discovery and development process. They allow for the creation of valid, human in vitro models for dissecting disease mechanisms, discovering novel drug targets, and screening drug candidates 5. The complexity of stem cell culture models in neuroscience continues to grow. Astrocyte/neuron or microglia/neuron co-culture models are becoming the norm, complex biological structures such as the blood-brain-barrier can be assembled in vitro, and organoid models are becoming amendable to drug screening. Could the application of these human preclinical models enhance the success rates and diminish the incidence of unforeseen toxicities in human trials? While we do not know the answer at this point, new regulations such as the FDA Modernization Act 2.0 now allow the use of certain alternatives to animal testing. These alternatives include cell-based assays like iPSC models, organoids, and organs-on-chips. The law also permits the use of advanced artificial intelligence (AI) methods, such as generative AI or large language models. We and our partners certainly aim to contribute to this new and exciting era of drug discovery.
- Durand, C. M. et al. SHANK3 mutations identified in autism lead to modification of dendritic spine morphology via an actin-dependent mechanism. Mol. Psychiatry 17, 71–84 (2012).
- Jiang, C.-C. et al. Signalling pathways in autism spectrum disorder: mechanisms and therapeutic implications. Signal Transduct. Target. Ther. 7, 229 (2022).
- Heavner, W. E. & Smith, S. E. P. Resolving the Synaptic versus Developmental Dichotomy of Autism Risk Genes. Trends Neurosci. 43, 227–241 (2020).
- Darville, H. et al. Human Pluripotent Stem Cell-derived Cortical Neurons for High Throughput Medication Screening in Autism: A Proof of Concept Study in SHANK3 Haploinsufficiency Syndrome. EBioMedicine 9, 293–305 (2016).
- Donowitz, M., Turner, J. R., Verkman, A. S. & Zachos, N. C. Current and potential future applications of human stem cell models in drug development. J. Clin. Invest. 130, 3342–3344 (2020).
Follow the Topic
-
Translational Psychiatry

This journal focuses on papers that directly study psychiatric disorders and bring new discovery into clinical practice.
Related Collections
With Collections, you can get published faster and increase your visibility.
From mechanism to intervention: translational psychiatry of childhood maltreatment
Publishing Model: Open Access
Deadline: Jun 30, 2026
Moving towards mechanism, causality and novel therapeutic interventions in translational psychiatry: focus on the microbiome-gut-brain axis
Publishing Model: Open Access
Deadline: May 19, 2026
Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in