Acute myeloid leukemia (AML) is the most common acute leukemia in adults of European ancestry and is associated with a poor outcome for many patients. Germline variants is genes regulating hematopoietic cell differentiation and proliferation can cause familial AML, but these families are exceptionally rare and do not make a major contribution to population disease burden. Outside of familial disease, genetic susceptibility to AML remains largely unexplained for the majority of patients.
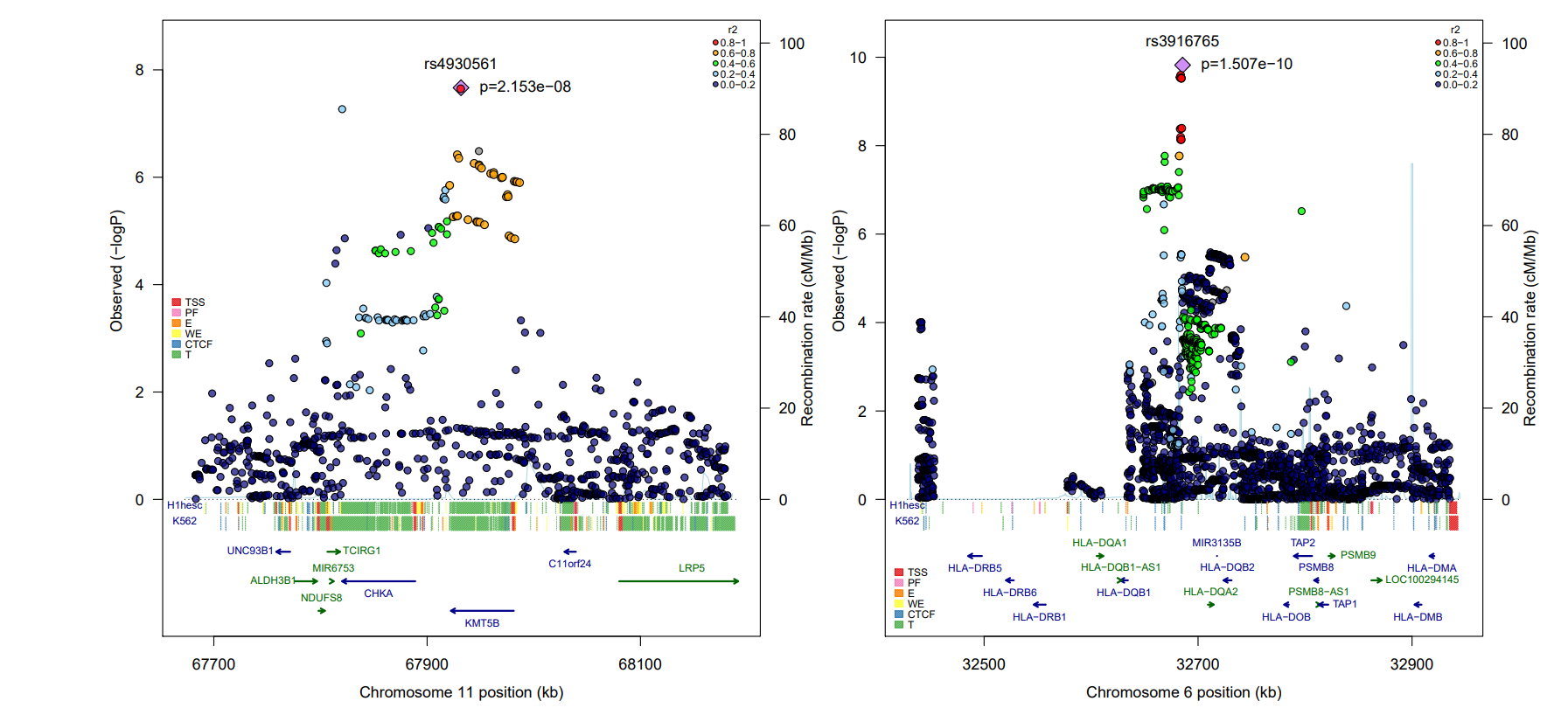
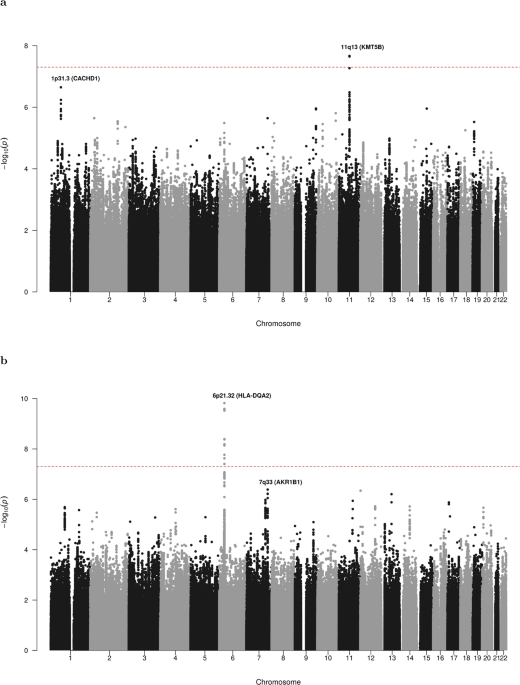
Thanks to a highly collaborative network of AML researchers and clinicians across the United Kingdom, continental Europe and North America we compared the DNA sequence from over 4000 AML patients with DNA from over 10000 healthy individuals and found two common genetic variants that are strongly associated with risk of developing AML. One of these variants is located on chromosome 11 adjacent to the KMT5B lysine methyltransferase gene, which has previously been implicated in the pathogenesis of AML. We also identified a major susceptibility locus for cytogenetically normal AML (the largest AML sub-type) on chromosome 6 at the human leukocyte antigen (HLA) gene. This region is important for immune function and carries susceptibility variants for numerous human cancers, where risk is mediated either via differential antigenic presentation/T-cell receptor recognition or altered risk of oncogenic infection.
Our study identifies common susceptibility alleles for AML risk, with evidence of sub-type specific risk loci reflecting the existence of multiple etiological pathways to disease development. Further work is required to decipher the functional basis of these risk loci although our data builds on existing evidence demonstrating a role for aberrant histone modification and altered immune function as etiologically important in AML.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in