Germanium-enriched d4r units inducing zeolite-confined subnanometric Pt clusters for efficient propane dehydrogenation
Published in Chemistry
Propylene is an important bulk chemical that is annually produced at a scale of approximately 130 million tons1. As an industrial technology for direct propylene production, propane dehydrogenation (PDH) has received extensive attention in recent years. Currently, non-oxidative direct dehydrogenation is the only commercialized process, and the most active catalysts are typically Pt or Cr based. Platinum is preferred for its ability to cleave C-H bonds and low activity in breaking C-C bonds. However, the C-C bond cleavage tends to occur on atomic steps and kink sites of large Pt particles, leading to low selectivity, coke formation and catalyst deactivation.
To suppress metal sintering and undesirable side reactions, a second metal (Sn2,3, Zn4,5, Ga6, Mn7) can be incorporated to improve the selectivity and/or stability of Pt. To date, the second metals of Sn and Zn, confined in the zeolite channels with Pt, are the two most promising options for preparing high-performance bimetallic PDH catalysts2-5,8. Increasing studies indicate that for bimetallic PtM alloy catalysts, the propylene selectivity and catalytic stability are improved at the expense of PDH activity, which even causes irreversible catalyst deactivation4-6,9. Therefore, developing encapsulated Pt clusters with non-alloy structure is of great interest for the purpose of overcoming the activity-stability trade-off in PDH.
For zeolite-confined Pt catalysts, the as-prepared Ptx-O-My (M = Sn, Zn, etc) species tend to evolve into the less active PtM alloy during the high-temperature prereduction or the PDH reaction operated at above 550 °C. For example, we have shown that the active Ptx clusters stabilized in Pt@Sn-Beta via Pt-O-Sn bonds could be partially transformed into PtxSny alloy under the harsh pretreatment (calcination-reduction or direct reduction) or long-term reaction processes due to the instable skeleton-Sn, which leads to the eventual catalyst deactivation10. Furthermore, for the Sn-Beta confined Pt catalysts, the location of Pt species in zeolite framework are hard to control due to the random distribution of skeleton-Sn. Thus, alternative strategies are required to control the placement of guest Pt while inhibiting the reduction of framework hetero-metal atoms, and the zeolites with larger pores are also needed to provide improved diffusion and accommodation capability for guest metal source in wet-impregnation process.
As elements in the same group, framework Ge4+ ion is relatively more difficult to be reduced compared to the Sn4+ ion. Thus, the germanosilicates with abundant framework Ge4+ ions and extra-large pores are considered better candidates as the carriers11,12. However, they exhibit poor hydrothermal stability even at room temperature due to the easy hydrolysis of Ge-O bonds, which greatly limits their use. We have once proposed a versatile stabilization strategy for the germanosilicates to achieve high structural stability without losing large-pore structures.13
Herein, we innovatively use a thermally/hydrothermally stable UTL-type germanosilicate (Ge-UTL) obtained by an acid treatment as an alternative support to confine the Pt species, taking advantage of its extra-large 14-membered ring pores, high content of stable skeleton-Ge, and the specific double four-membered ring (d4r) subunits. The Pt@Ge-UTL catalyst was readily prepared from Ge-UTL carrier and platinum source H2PtCl6 by simple wet-impregnation and subsequent H2 reduction.
iDPC-STEM, in situ XAFS, 19F MAS NMR, full-range synchrotron pair distribution function G(r) analysis and DFT calculations verify that Pt clusters with average four atoms are firmly restricted in the 14-membered ring channels, which is realized by selectively and directionally anchoring Pt via Pt-O-Ge bonding to UTL’s unique secondary building units with d4r configuration and Ge-enriched composition. The schematic diagram is as follows:
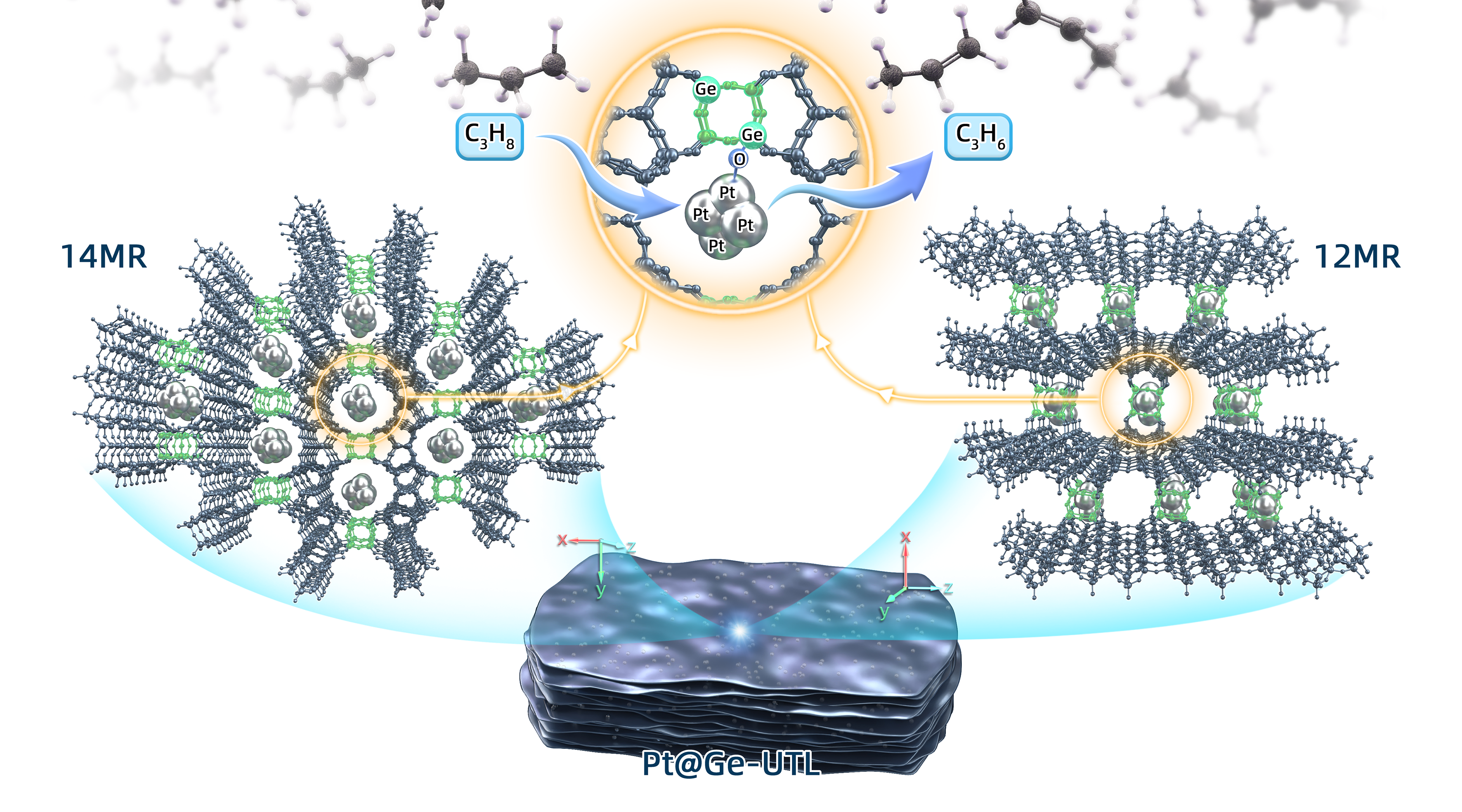
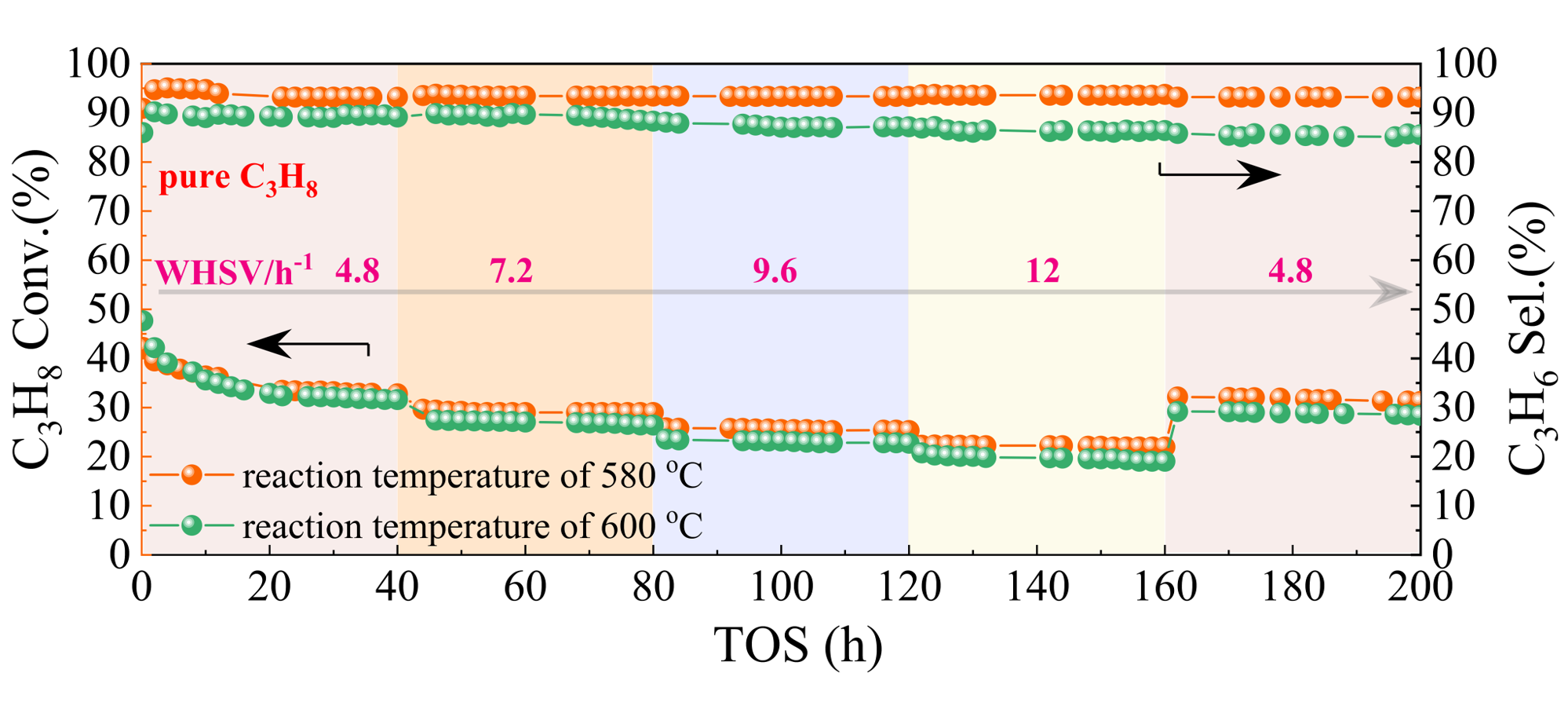
The host-guest bimetallic structure (Pt4-Ge2-d4r@UTL) achieves high activity, propylene selectivity and long-term stability in propane dehydrogenation (Figure 1). This research pioneers the utilization of germanosilicates in the designed synthesis of high-performance PDH catalysts.
Reference
- Carter JH, et al. Direct and oxidative dehydrogenation of propane: from catalyst design to industrial application. Green Chem 23, 9747-9799 (2021).
- Liu L, et al. Regioselective generation and reactivity control of subnanometric platinum clusters in zeolites for high-temperature catalysis. Nat Mater 18, 866-873 (2019).
- Zhu J, et al. Ultrafast Encapsulation of Metal Nanoclusters into MFI Zeolite in the Course of Its Crystallization: Catalytic Application for Propane Dehydrogenation. Angew Chem Int Ed 59, 19669-19674 (2020).
- Sun Q, et al. Subnanometer Bimetallic Platinum-Zinc Clusters in Zeolites for Propane Dehydrogenation. Angew Chem Int Ed 59, 19450-19459 (2020).
- Wang Y, et al. Ultrasmall PtZn bimetallic nanoclusters encapsulated in silicalite-1 zeolite with superior performance for propane dehydrogenation. J Catal 385, 61-69 (2020).
- Searles K, et al. Highly Productive Propane Dehydrogenation Catalyst Using Silica-Supported Ga-Pt Nanoparticles Generated from Single-Sites. J Am Chem Soc 140, 11674-11679 (2018).
- Wu Z, et al. Changes in Catalytic and Adsorptive Properties of 2 nm Pt3Mn Nanoparticles by Subsurface Atoms. J Am Chem Soc 140, 14870-14877 (2018).
- Liu L, et al. Structural modulation and direct measurement of subnanometric bimetallic PtSn clusters confined in zeolites. Nat Catal 3, 628-638 (2020).
- Deng L, et al. Elucidating strong metal-support interactions in Pt-Sn/SiO2 catalyst and its consequences for dehydrogenation of lower alkanes. J Catal 365, 277-291 (2018).
- Ma Y, et al. Skeleton-Sn anchoring isolated Pt site to confine subnanometric clusters within *BEA topology. J Catal 397, 44-57 (2021).
- Paillaud J, et al. Extra-Large-Pore Zeolites with Two-Dimensional Channels Formed by 14 and 12 Rings. Science 304, 990-992 (2004).
- Corma A, et al. ITQ-15: The first ultralarge pore zeolite with a bi-directional pore system formed by intersecting 14- and 12-ring channels, and its catalytic implications. Chem Commun 12 1356-1357 (2004).
- Xu H, et al. Post-Synthesis Treatment gives Highly Stable Siliceous Zeolites through the Isomorphous Substitution of Silicon for Germanium in Germanosilicates. Angew Chem Int Ed 53, 1355-1359 (2014).
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in