How can kinetics and thermodynamics be disentangled in phase separation?
Published in Materials

Nucleation and growth (N&G) is possibly the most common mechanism by which heterogeneous systems form in the universe. In solid-liquid and gas-liquid phase separation, the newly forming phase is relatively simple to study and understand even in multi-component systems: think for example of CO2 gas bubbles forming in a freshly opened bottle of champagne or the solid alloy developing from a cooling molten metal blend. The reason for this behavior is that the newly forming phase follows a nearly vertical binodal line, i.e. the composition-dependent line separating the one-phase from the two phase region in the corresponding phase diagram.
In liquid-liquid phase separation (LLPS) the situation is very different: binodal lines tend to have a moderate slope (1) which coupled with the components’ finite transport rate associated with N&G, impose a time-dependence fingerprint to the effective binodal lines, making the time parameter pervasive in most heterogeneous systems and intimately connecting thermodynamics and kinetics aspects. Indeed, binodal lines during phase separation are only followed for infinitely fast systems, which do not exist in nature. In real systems, changes in temperature, pressure or any other trigger of phase separation requires time to be reflected in phase separation and the system typically is always super-saturated before phase separation starts, i.e. departing from truly thermodynamic equilibrium conditions and binodal lines. For example, upon continuous change in temperature of a polymer solution, any droplet phase separating out from the continuous phase would change in composition in a way that depends both on the profile of the binodal lines (expressed as temperature-composition lines) but also on the kinetics parameters, (e.g. diffusion coefficient). Thus, the possibility of completely separating kinetics and thermodynamics effects in phase separation phenomena remains pretty much a dream of all soft matter physicists.
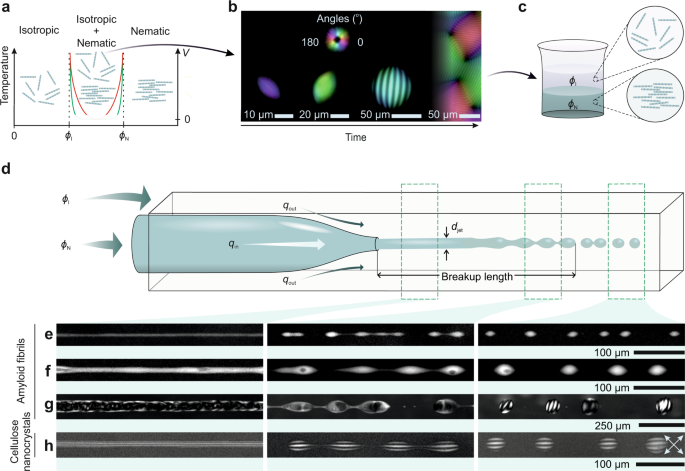
In our recent work (https://www.nature.com/articles/s41467-023-36292-8) we show that this dream has now come true! We select filamentous colloids based on both amyloid fibrils and cellulose to realize phase separation via a different, purely entropically driven process, more correctly referred to as liquid-liquid crystalline phase separation (LLCPS). The radically different shape of the binodal lines in LLCPS compared to LLPS allows us to more easily disentangle thermodynamic and kinetic effects on phase separation, providing the basis to understand them as two totally separated contributions.
Indeed, differently from LLPS, in LLCPS the thermodynamic binodal lines are two vertical asymptotes set at precise compositions depending only on the aspect ratio of the filamentous colloids (2); in other words, the system is not (less) sensitive to changes in temperature or other environmental parameters. Amyloid fibrils and cellulose offer two experimental examples in which the effective binodal lines, expressed by volume-compositions trajectories, approach these two vertical asymptotes (i.e. the thermodynamic binodal lines) with either gentle or steep slopes, giving a different weight to the role of kinetics on the final morphology. We study for these two systems the heterogeneous colloidal morphology developing by either classical N&G or by extruding one phase set at one thermodynamic asymptote into the other phase set at the other thermodynamic asymptote and inducing droplet formation no longer by N&G, but by Rayleigh-Plateau instability (See Figure 1). By doing so, we can show that departing from the N&G paradigm unveils new physical phenomena, such as orders of magnitude shorter timescales, a wider phase diagram, different internal structures of the colloidal droplets and unprecedented control on the composition of multi-component heterogeneous colloidal fluids: all features which cannot be achieved by conventional phase separation via N&G. We also show that the milder the slope of the effective binodal lines, the more evident non-equilibrium aspects become, and we demonstrate how, even for these systems, our approach allow establishing morphologies based purely on thermodynamics.
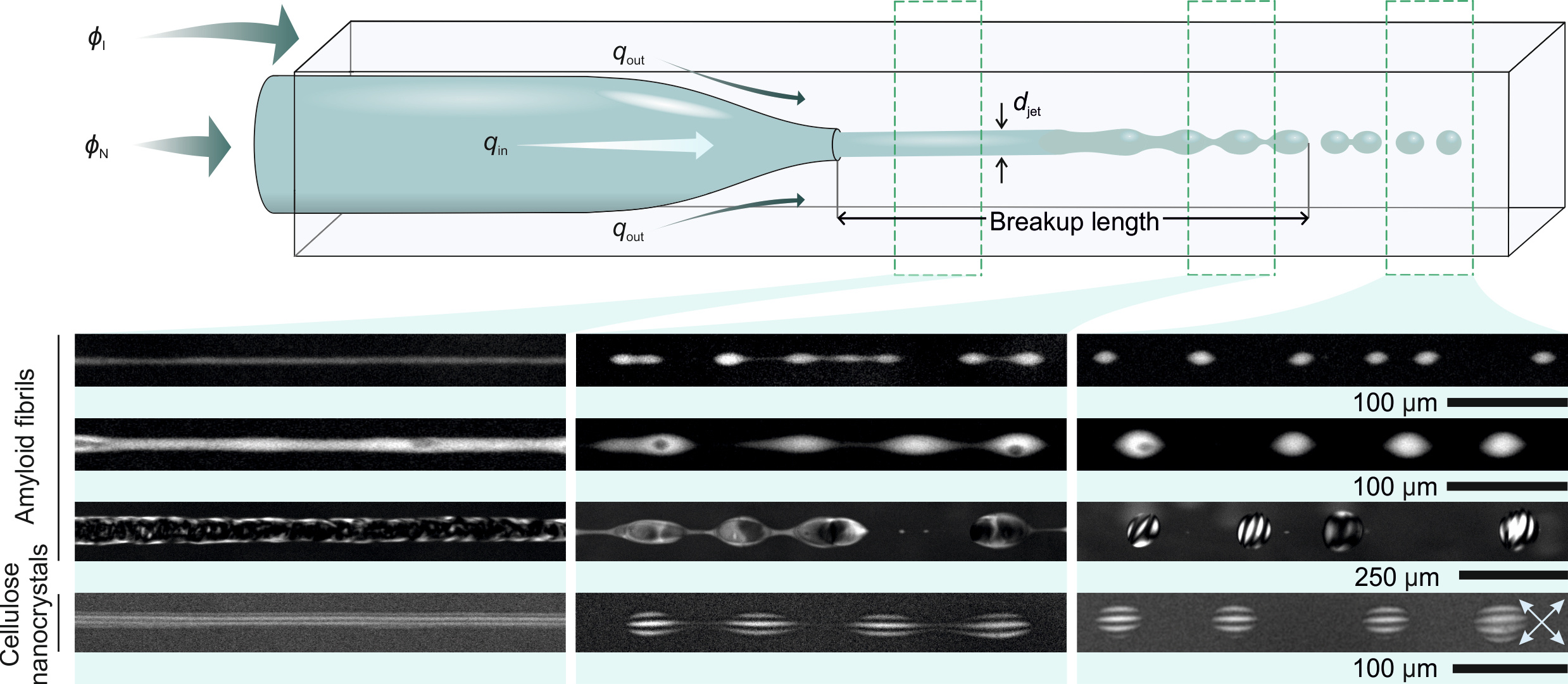
Figure 1. Microfludics setup used to create droplets at equilibrium by Rayleigh-Plateau instability on co-extruded macroscopically separated phases.
For example, a striking feature of LLCPS in amyloids is the production of droplets of cholesteric phases, called cholesteric tactoids, in which the characteristic fringe band reflecting the chiral twist organization of the filamentous colloids decreases its periodicity with the size of the droplet and change with time (3). These effects are understood to arise from non-equilibrium, kinetics effects, since amyloid-cholesteric phases are known to be following slow relaxation kinetics (4). However, when the same amyloid tactoids are formed by Rayleigh-Plateau instability, no changes in periodicity of the cholesteric bands are observed either with volume or time. Furthermore, the periodicity observed in droplets formed by Rayleigh-Plateau instability is up to 3 times larger than that obtained by N&G (46.1 vs 16.7 µm). Via this and several other observations, we conclude that droplets obtained by Rayleigh-Plateau instability are already formed at thermodynamic equilibrium and reveal structural and morphological features that kinetics effects in N&G does hide instead.
In sharp contrast, tactoids formed by sulphated nanocellulose, a system known to follow a quick relaxation kinetics (4) and for which tactoids formed by N&G do not feature time or size dependence of the cholesteric band periodicity (5), show no significant difference when obtained by the two methods, demonstrating that the, in this case, droplets formed by N&G approach the binodal asymptotes quickly enough to keep kinetics effects to a minimum.
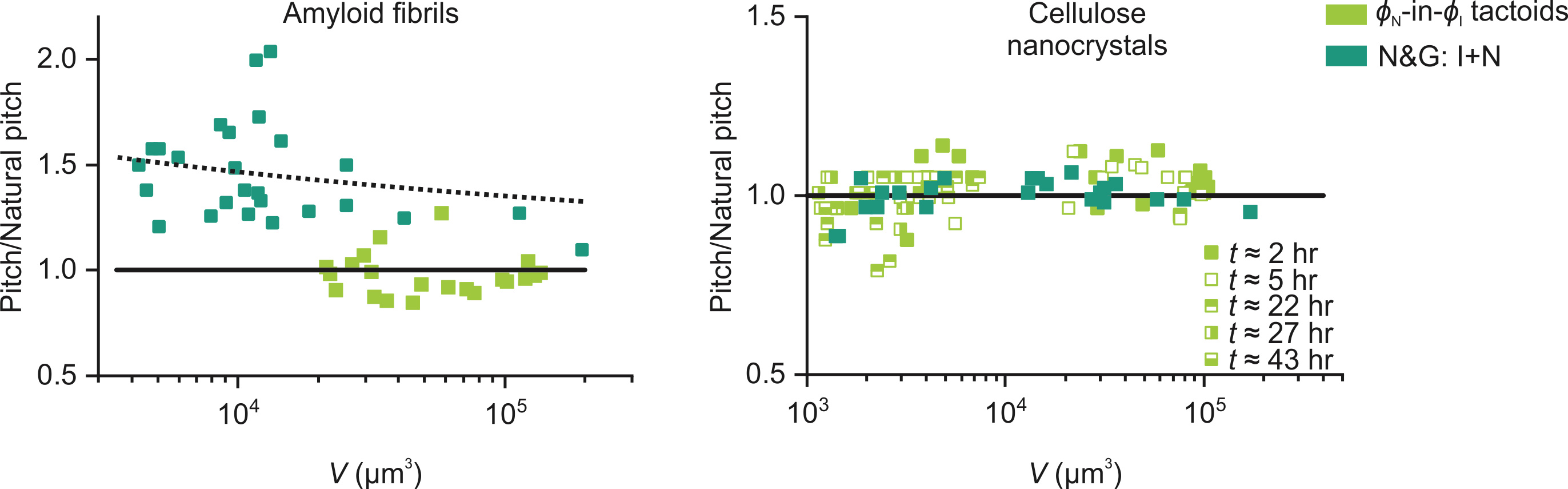
Figure 2. Evolution of the characteristic pitch of cholesteric tactoids with their volume in the case of amyloid (left) and sulphated cellulose nanocrystals (right).
Put it in other words: let amyloids, nanocellulose, collagen or any other filamentous colloid phase separate by LLCPS (5); then take two droplets of identical size, one formed via the common N&G path, the other made by our new concept (see Figure 1): the droplet formed by N&G will carry information from the kinetics phase separation process, while our droplet will enjoy pure, splendid thermodynamic equilibrium! We also see a number of other very interesting effects in droplets formed at thermodynamic equilibrium by Rayleigh-Plateau instability, such as smaller droplets or a wider phase diagram that are not at reach via conventional N&G, and we can even combine these effects to design multicomponent colloidal systems with precise structural control, well beyond what possible via N&G.
References
- Flory, P. J. (1953).Principles of Polymer Chemistry. Ithaca, New York: Cornell University Press.
- Onsager, L. (1949). The Effects of Shape on the Interaction of Colloidal Particles. NY Acad. Sci. 51, 627–659.
- Nyström, G., Arcari, M., and Mezzenga, R. (2018). Confinement-induced liquid crystalline transitions in amyloid fibril cholesteric tactoids, Nature Nanotechnology, 13, 330-336.
- Khadem, S.A., Bagnani, M., Mezzenga, R., Rey, A.D. (2020). Relaxation dynamics in bio-colloidal cholesteric liquid crystals confined to cylindrical geometry. Nature communications 11, 1-10.
- Azzari, P., Bagnani, M., and Mezzenga, R. (2021). Liquid-Liquid Crystalline Phase Separation in Biological Filamentous Colloids: Nucleation, Growth and Order-Order Transitions of Cholesteric Tactoids. Soft Matter, 17, 6627–6636.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Healthy Aging
Publishing Model: Open Access
Deadline: Dec 31, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in