Identification of four novel loci associated with psychotropic drug-induced weight gain in a genome-wide association study
Published in Healthcare & Nursing

Patients suffering from mental disorders are at high risk of developing cardiovascular diseases, leading to reduced life expectancy. Both genetic and environmental components (e.g. mental disorder-related factors, such as an unhealthy lifestyle, poor access to somatic care and/or adverse effects of treatment) may explain the observed excessive propensity of patients suffering from mental disorder for developing metabolic diseases. For instance, the use of psychotropic medications, such as antipsychotics (most atypical but also some typical), mood stabilizers (e.g. lithium), and some antidepressants (e.g. mirtazapine) can increase the risk of metabolic disorders including obesity through various mechanisms.
A substantial inter-individual variation in the tolerability of psychotropic drugs is observed in psychiatry, regardless of the type of medication, which represents a significant challenge for physicians and their patients. Adverse metabolic effects induced by psychotropic drugs are a major source of patients’ nonadherence (and/or treatment discontinuation), which leads to higher rates of relapse. Genetic variability can contribute to inherited differences in drug-induced side effects.
Although genome-wide association studies (GWAS) have been a rich source to provide novel candidate genes and pathways of cardiometabolic phenotypes, the explained variance by individual genetics remains very low. Genetic variants can display stronger effects on cardiometabolic features in psychiatric cohorts compared to the general population. This difference is potentially due to an intricate interaction between the mental disorder or the medications used to treat it and metabolic regulations. Previous GWAS on antipsychotic-induced weight gain included a low number of participants and/or were restricted to patients taking one specific antipsychotic.
To our knowledge, we have conducted the largest GWAS investigating the evolution of BMI during psychotropic treatment to date. The present GWAS would improve our understanding of biological processes underlying psychotropic drug-induced weight gain. We conducted a GWAS of the evolution of body mass index (BMI) during early (i.e. ≤ 6) months of treatment with psychotropic medications (i.e. antipsychotics, mood stabilizers and some antidepressants) inducing metabolic disturbances in 1135 patients from the PsyMetab cohort. Clinical data were collected during hospitalization or in outpatient centers during a medical examination based on the department guideline for the metabolic follow-up performed on a routine basis. Monitoring for physical health risk factors included prospective assessments of metabolic factors such as body mass index (BMI) during treatment. Evolution of BMI throughout psychotropic treatment was chosen as the primary measure as it is a well-known adverse side effect of psychiatric medications. Evolution of BMI evaluated both as a change over time (slope) and raw change (difference between BMI before and after treatment), considering various periods of follow-up (1-, 3- and 6- month) were all considered as primary endpoints.
In the PsyMetab cohort, four loci were associated with BMI phenotypes at genome-wide significant level (p < 5 x 10-8); Figure 1. In particular, rs7736552 (near MAN2A1), rs11074029 (in SLCO3A1), rs117496040 (near DEFB1) and rs7647863 (in IQSEC1) were associated with BMI slope, BMI slope at 6 months, BMI change and BMI change at 3 months, respectively. Associations between the four loci and alternative BMI-change phenotypes showed consistent effects.
The identified GW-significant signals were tested for replication in the UK Biobank (UKB), a large population-based cohort from the UK with rich genotype and phenotype information. A subset of UKB participants who were diagnosed with a mental disorder or treated with a psychotropic drug was used as an independent psychiatric replication sample. We also used the UKB as a non-psychiatric population-based sample in order to determine whether the observed associations were unique to the population of subjects who suffer from a mental disorder or were also associated with the evolution of BMI in the population-based cohort. Replication analyses conducted in UK Biobank participants diagnosed with a mental disorder and/or taking a psychotropic drug revealed a sign-concordant association between rs7736552 and BMI slope (p = 0.017), whereas no association was observed between the three remaining loci and BMI slope. In 29,376 population-based (non-psychiatric) participants of the UKB, the four loci were not associated with BMI slope (p ≥ 0.16).
Although not replicated in UKB samples, associations between rs11074029 (in SLCO3A1), rs117496040 (near DEFB1) and rs7647863 (in IQSEC1) and BMI outcomes were consistent with previous studies that reported associations between these genes and/or SNPs and metabolic phenotypes. Interestingly, rs11074029 and rs117496040 displayed significant associations with BMI and psychiatric phenotypes, in agreement with a recent GWAS reporting extensive polygenic overlap between BMI and major mental disorders which emphasized the complex interplay of metabolism-related gene pathways in the pathophysiology of mental disorders.
Although the PsyMetab cohort was large, the number of participants on drugs with a high propensity of weight gain (e.g. olanzapine and clozapine) was relatively small. Larger studies (n > 1000) focused on single psychotropic drugs separately may allow for the identification of specific loci that could provide insights on drug-specific mechanisms underlying metabolic disturbances. On the other hand, PsyMetab is a rich longitudinal data set, allowing proper phenotype correction of potentially confounding variables throughout psychotropic treatment. This cohort is very well positioned to analyze side-effects of psychotropic drugs as all participants have available baseline (i.e., pre-treatment) data. Even if the present results are preliminary, they already reveal that no individual common genetic variant plays a major role in psychotropic drug induced weight gain. Notably, replication in independent studies should be conducted before these results can be considered in clinical settings. In summary, these findings provide new insights into metabolic side effects induced by psychotropic drugs and underline the need for future studies to replicate these associations in larger cohorts.
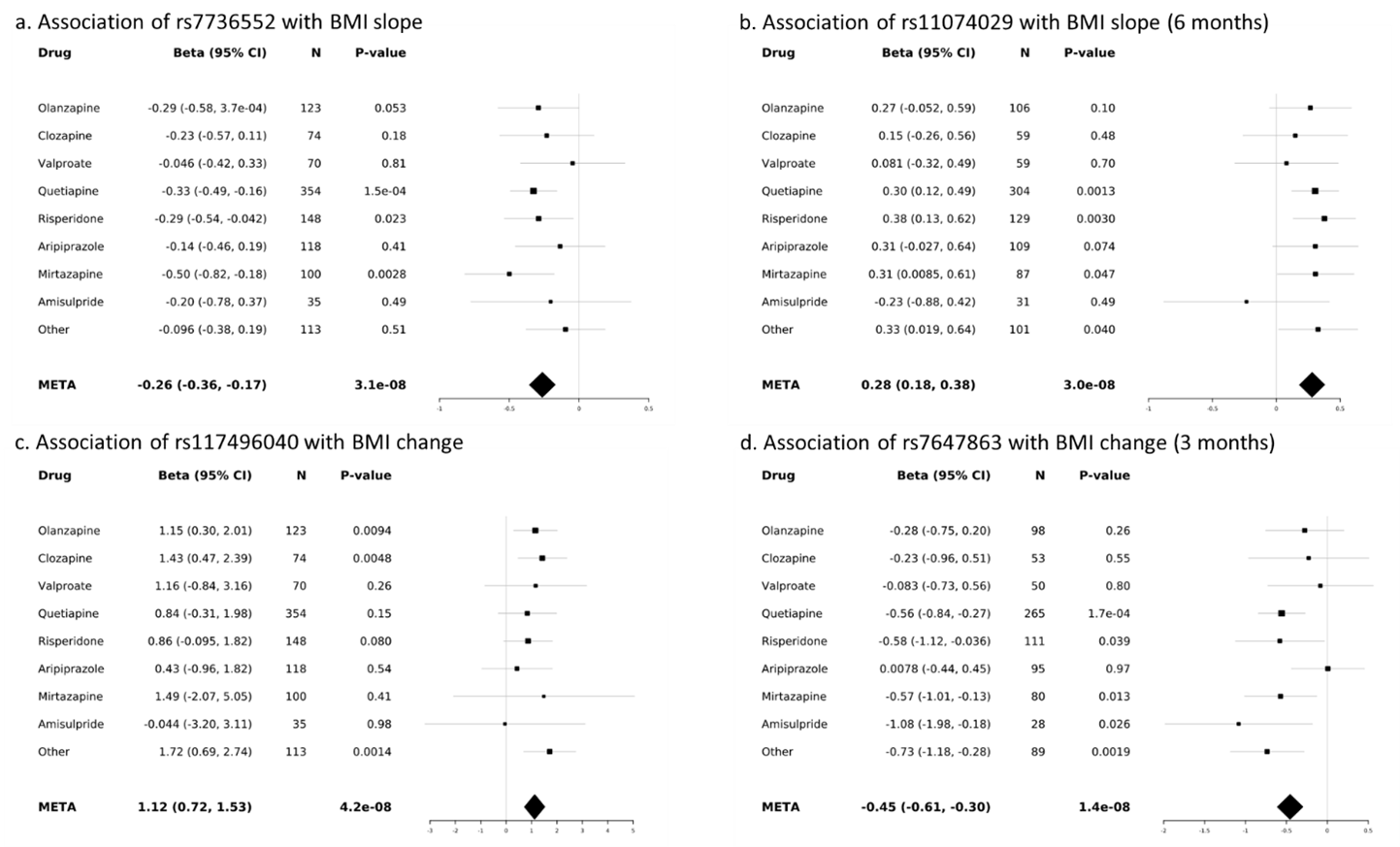
Figure 1. Forest plots of SNP association with metabolic phenotypes in PSYMETAB
Follow the Topic
-
Molecular Psychiatry
This journal publishes work aimed at elucidating biological mechanisms underlying psychiatric disorders and their treatment, with emphasis on studies at the interface of pre-clinical and clinical research.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in