Innovations in Acetylene Semi-Hydrogenation Catalysts with Barrier-Less Activation Energy
Published in Chemistry
In today's fast-paced world, where efficiency and sustainability are paramount, the quest for innovative catalytic solutions has never been more imperative. Our focus lies in exploring the realm of acetylene semi-hydrogenation catalysts, a field ripe with potential for transformative breakthroughs.
Behind our latest paper A MOF–supported Pd1–Au1 dimer catalyses the semi–hydrogenation reaction of acetylene in ethylene with a nearly barrier–less activation energy (https://www.nature.com/articles/s41929-024-01130-7), we delved into the fascinating world of barrier-less catalysis, where traditional limitations are being challenged and new possibilities are emerging. We uncovered the intricacies of acetylene semi-hydrogenation, exploring recent developments, challenges faced, and most importantly, the catalytic innovations driving progress in this area.
Our story began with a thriving collaboration between our laboratory (https://catsusorg-itq.webs.upv.es/), which joined forces with leading experts from diverse fields of chemical analysis, recognizing the need for research acceleration. This endeavour brought together specialists in the design of porous materials and MOFs, microscopy and computational modelling. Together, we are pushing the boundaries of catalysis and paving the way for more efficient, sustainable, and economically viable processes.
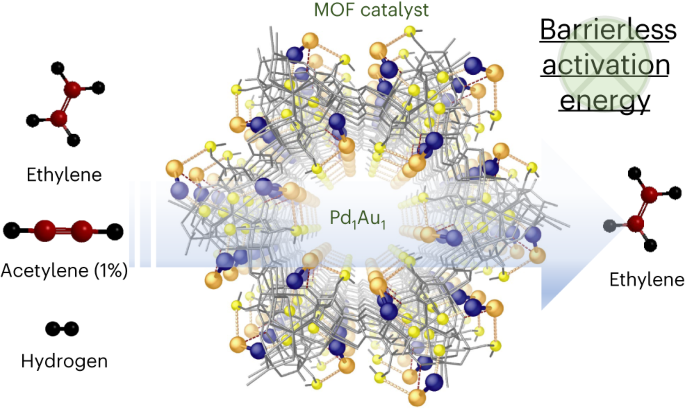
As part of this journey, we attempted to create a unique chemical combination to see if it could help turn one substance into another. We made a powerful mixture called a Pd1-Au1 dimer, that operates like a magical ingredient on a special material derived from a natural amino acid. This material can transform a harmful gas called acetylene into one of the most sought-after chemicals globally, ethylene, with almost no waste and at different temperatures, under conditions similar to those in the factories.
What makes this catalyst so special? Let's take a closer look at its key features:
- Unparalleled efficiency: With a conversion rate of over 99.99% for acetylene and a selectivity of more than 90% towards ethylene, this catalyst excels in transforming raw materials into valuable products. Its efficiency is unmatched, promising significant advancements in industrial processes.
- Adaptability: Operating seamlessly within a broad temperature range of 35 to 150 ºC, the catalyst showcases its adaptability to various industrial settings. Whether it's a high-temperature process or a lower-temperature operation, this catalyst delivers consistent results.
- Minimal Energy Barriers: With an activation energy of just around 1 kcal·mol⁻¹, the catalyst displays impressive efficiency, suggesting a streamlined reaction pathway with minimal energy barriers. This ensures smooth and efficient catalytic reactions, enhancing overall productivity.
- Mechanistic Insights: A combined experimental and computational study provides valuable insights into the catalyst's mechanism. It reveals that the Pd atom acts as the main catalytic site, while the Au atom and thioether moieties of the MOF assist during the dihydrogen dissociation step. This deep understanding of the catalyst's mechanism opens up new avenues for further research and development.
Like any other ambitious scientific project, we have faced a myriad of obstacles, each demanding innovative solutions. The complexity of catalyst design posed a challenge. Balancing the competing demands of efficient catalytic activity and stability required intricate strategies and meticulous attention to detail. Optimizing reaction conditions proved to be another hurdle. Fine-tuning parameters like flow rates and temperature while maintaining catalyst stability demanded careful experimentation and analysis. The theoretical understanding was essential yet elusive. Developing a comprehensive model of the catalytic mechanism at the molecular level required sophisticated computational tools and rigorous validation against experimental data. Characterization and validation have presented their own hurdles, requiring precision and accuracy at every turn. Yet, perhaps our greatest challenge lies ahead: bridging the gap between laboratory research and real-world industrial application. As we strive to demonstrate scalability, stability, and efficiency on a grand scale, our journey continues with determination and perseverance.
In summary, the Pd-Au dimer supported on a MOF is a catalyst with such a performance, adaptability, and mechanistic insights that make it a potential game-changer in the world of semi–hydrogenation reaction of acetylene in ethylene streams. As we continue to unlock its full potential, the possibilities for innovation and advancement are wide. We reflect here that the future of catalysis research for ethylene production is full of endless possibilities. Our adventure has been about teamwork, determination, and making things more efficient by discovering new ways to optimize a process in real-time. Whether you are an experienced industry professional, a curious researcher, or simply someone fascinated by science, we invite you to join us in exploring the exciting frontiers of catalysis. Let's unlock the secrets of innovation and turn our dreams into reality!
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in