Insects secrete salivary proteins to manipulate plant defenses
Published in Ecology & Evolution
In nature, plants are continuously challenged by bacteria, fungi, nematodes, and insects. To survive or fend off attacks, plants have evolved multi-layered immune systems from recognizing invaders to activating defense responses. Pattern recognition receptors (PRRs) can perceive the “non-self” molecules known as pathogen-, herbivore- and damage-associated molecular patterns (PAMPs/HAMPs/DAMPs), and activate the pattern-triggered immunity (PTI). To counteract plant immunity, the invaders deliver secretory effectors to target the immune signaling components of PTI and interfere with their activities. However, some effectors are sensed by the plants with time, which further initiates the effector-triggered immunity (ETI). After over millions of years of co-evolution, the invaders have developed dynamic and complex interactions with host plants.
Piercing-sucking insects, such as planthoppers, aphids, and whiteflies, are important invaders that damage host plants by feeding or transmitting viruses. During the feeding process, insects secrete abundant saliva into host plants. These oral secretions, on the one hand, hinder insect performance by activating plant defenses. For example, salivary protein Cathepsin B3 from Myzus persicae can be recognized by tobacco plants, which thus suppresses aphid feeding by triggering ROS accumulation. On the other hand, saliva exerts multiple functions to improve insect performance, such as calcium binding proteins for calcium regulation, DNase II for extracellular DNA degradation, and Helicoverpa armigera R-like protein 1 (HARP1) for plant hormonal manipulation. We presumed that there may have some salivary elicitors triggering plant defenses, while the elicitor-induced plant defenses are then inhibited by other salivary components. However, little is known about the complex interaction in saliva.
Given the importance of saliva, our study first screens the critical salivary proteins in insect feeding. The Laodelphax striatellus salivary protein 1 (hereafter: LsSP1) is among the top 5 most abundant, salivary gland-specific, and planthopper-specific genes, which is thereby selected for further analysis.
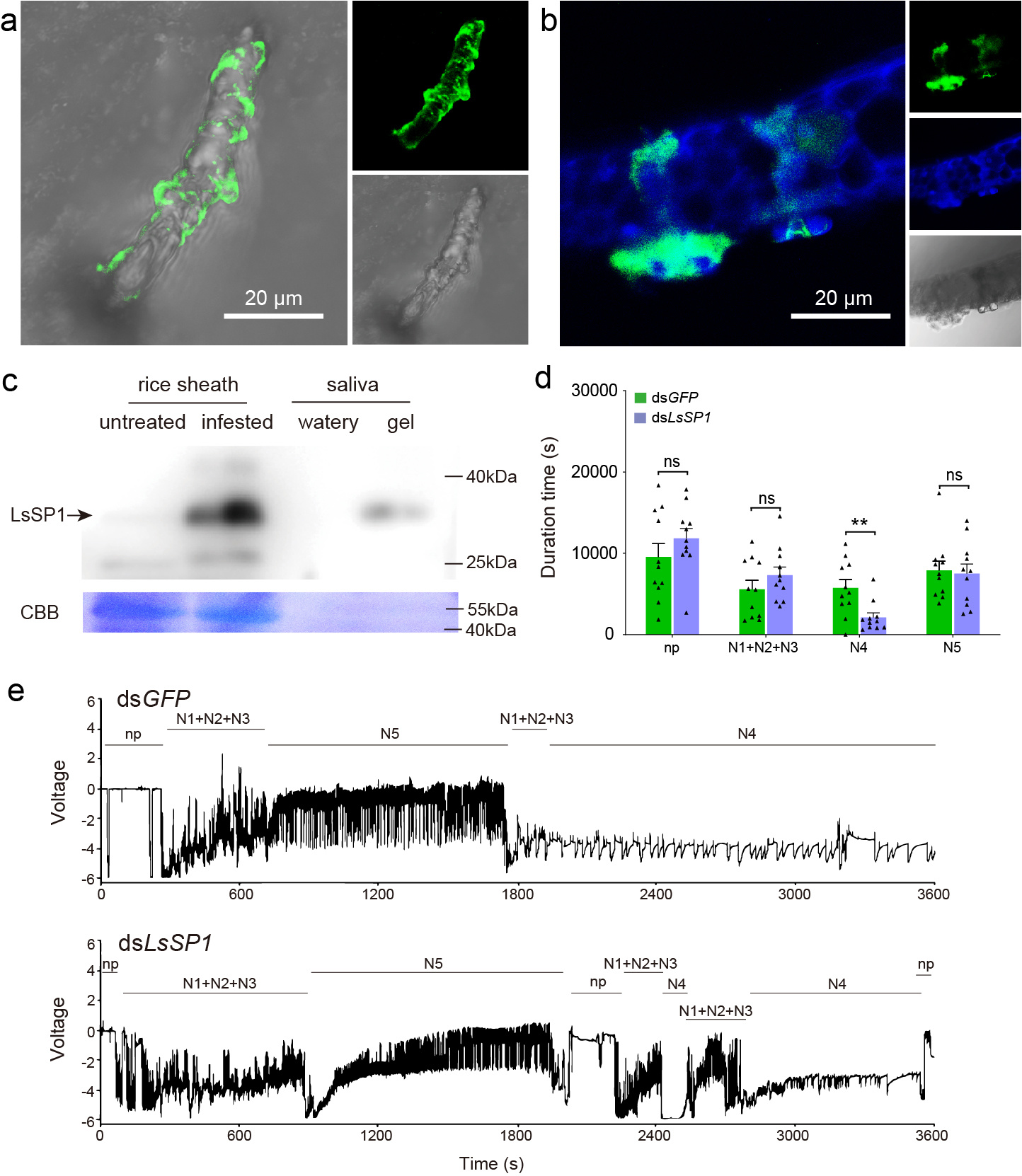 Figure 1. LsSP1 is a salivary sheath protein critical for insect feeding. a,b LsSP1 staining of salivary sheath on parafilm (a) and in rice tissues (b). c Detection of LsSP1 in untreated plants (lane 1) and plants infested by planthoppers (lane 2), watery (lane 3) and gel saliva (lane 4). d Comparison of electrical penetration graph (EPG) parameters between dsGFP-treated and dsLsSP1-treated planthoppers. e Overall typical EPG waveforms over 1 h for dsGFP-treated (upper) and dsLsSP1-treated L. striatellus (lower). The insect feeding behavior was classified into nonpenetration (np), pathway duration (N1+N2+N3), phloem sap ingestion (N4), and xylem sap ingestion (N5) phases.
Figure 1. LsSP1 is a salivary sheath protein critical for insect feeding. a,b LsSP1 staining of salivary sheath on parafilm (a) and in rice tissues (b). c Detection of LsSP1 in untreated plants (lane 1) and plants infested by planthoppers (lane 2), watery (lane 3) and gel saliva (lane 4). d Comparison of electrical penetration graph (EPG) parameters between dsGFP-treated and dsLsSP1-treated planthoppers. e Overall typical EPG waveforms over 1 h for dsGFP-treated (upper) and dsLsSP1-treated L. striatellus (lower). The insect feeding behavior was classified into nonpenetration (np), pathway duration (N1+N2+N3), phloem sap ingestion (N4), and xylem sap ingestion (N5) phases.
First, we validate that LsSP1 is a salivary sheath protein, as it is detected only in the salivary sheath sample but not in the watery saliva sample (Figure 1). Salivary sheath, formed by gel saliva, is secreted during the stylet probing, which provides mechanical stability and lubrication for stylet movement. Using RNAi, Y2H, BiFC, and LUC assays, our study further demonstrated that LsSP1 binds to salivary sheath through another salivary sheath protein, mucin-like protein (LsMLP). LsMLP is the main component of salivary sheath in the planthopper. Also, LsMLP is one of the HAMPs that are capable of inducing plant defenses. Interaction between LsSP1 and LsMLP may prevent the activation of plant defenses by masking the LsMLP, which deserved further investigation.
Then, Y2H screening is performed to understand the potential roles of LsSP1 in insect-plant interaction. The rice immune protein OsOryzain is found to interact with LsSP1. OsOryzain is highly homologous with Arabidopsis RD21, tomato C14, and maize Mir3 cysteine proteases. It contained a predicted N-terminal secretion signal and a self-inhibitory prodomain followed by peptidase, and granulin domains. OsOryzain is a member of papain-like cysteine proteases (PLCPs), which act as a central hub in plant immunity and are required for the full resistance of plants to various pathogens. Using Y2H, GST-pull down, BiFC, and LUC, our study validates that LsSP1 interacted with multiple PLCPs belonging to different subfamilies. Rice plants treated with salicylic acid (SA) and infested by planthoppers induced a similar amount of mOsOryzain in plant cells. However, rice plants infested by planthoppers secreted a lower amount of mOsOryzain into apoplast than that under SA treatment, suggesting that planthoppers potentially reduced the PLCP level in apoplast.
How planthoppers reduce PLCPs in rice apoplast? Do LsSP1-PLCPs interactions participate in this process? To answer these questions, we first investigate the binding capacity of OsOryzain to salivary sheath. Immunohistochemical staining reveals that LsSP1 and OsOryzain overlap in rice plants, with the strongest OsOryzain signal being detected around the salivary sheath. However, OsOryzain fails to bind to salivary sheath that secreted by dsLsSP1-treated planthoppers. Then, we incubate the salivary sheath with mOsOryzain-GFP and GFP proteins. As a result, strong green fluorescence is detected in salivary sheath incubated with mOsOryzain-GFP proteins, but not with GFP proteins. Subsequently, planthoppers are pre-treated with dsGFP and dsLsSP1, and the binding capacity of the secreted salivary sheaths is tested with mOsOryzain-GFP. The results show that the green fluorescence intensity significantly decreases in salivary sheaths secreted from dsLsSP1-treated planthoppers than that secreted from the dsGFP-treated ones, suggesting that LsSP1 is critical for salivary sheaths binding to OsOryzain
Fungal, bacterial, and oomycete pathogens have been reported to secrete effector proteins to suppress the proteolytic activity of PLCP. However, whether insects evolve a similar strategy remains unknown. Our study demonstrates that LsSP1 is capable of inhibiting the activity of apoplastic proteases in rice plants. Interestingly, LsSP1 and other inhibitors do not show sequence similarity or are even restricted to specific species. This suggests that apoplastic proteases play crucial roles in plant defenses against different invaders, and that these invaders employ the independently evolve effectors to target defensive components, thus facilitating colonization.
In this study, we also construct two transgenic rice lines constitutively overexpressing LsSP1 (oeSP1) and knockout OsOryzain (koOry). Overexpressing LsSP1 can well rescue the planthopper feeding defects caused by a deficiency in LsSP1 secretion. However, we find that rice plants knockout of OsOryzain cannot very well rescue the feeding defects caused by a deficiency in LsSP1 secretion. This may be explained by complex interactions between effectors and different plant defense actors. For salivary LsSP1, it targets multiple PLCPs belonging to different subfamilies. The knockout of OsOryzain alone cannot inhibit plant defenses initiated by other PLCPs. In addition, LsSP1 is capable of interacting with other plant and insect proteins. The salivary LsSP1 potentially exerts multiple roles during insect feeding, and affects plant defense in other ways independent of PLCPs, which deserves further investigation.
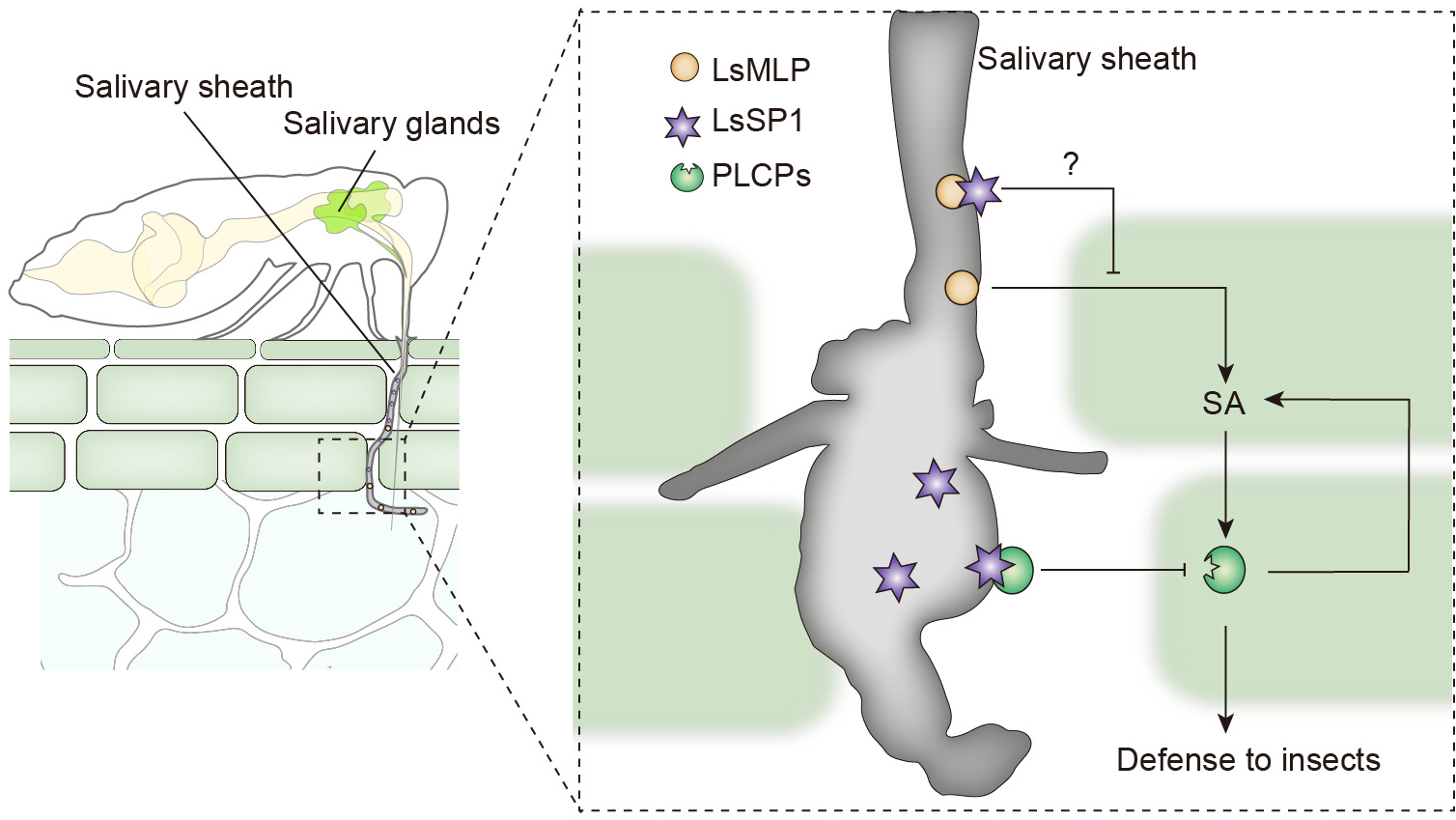
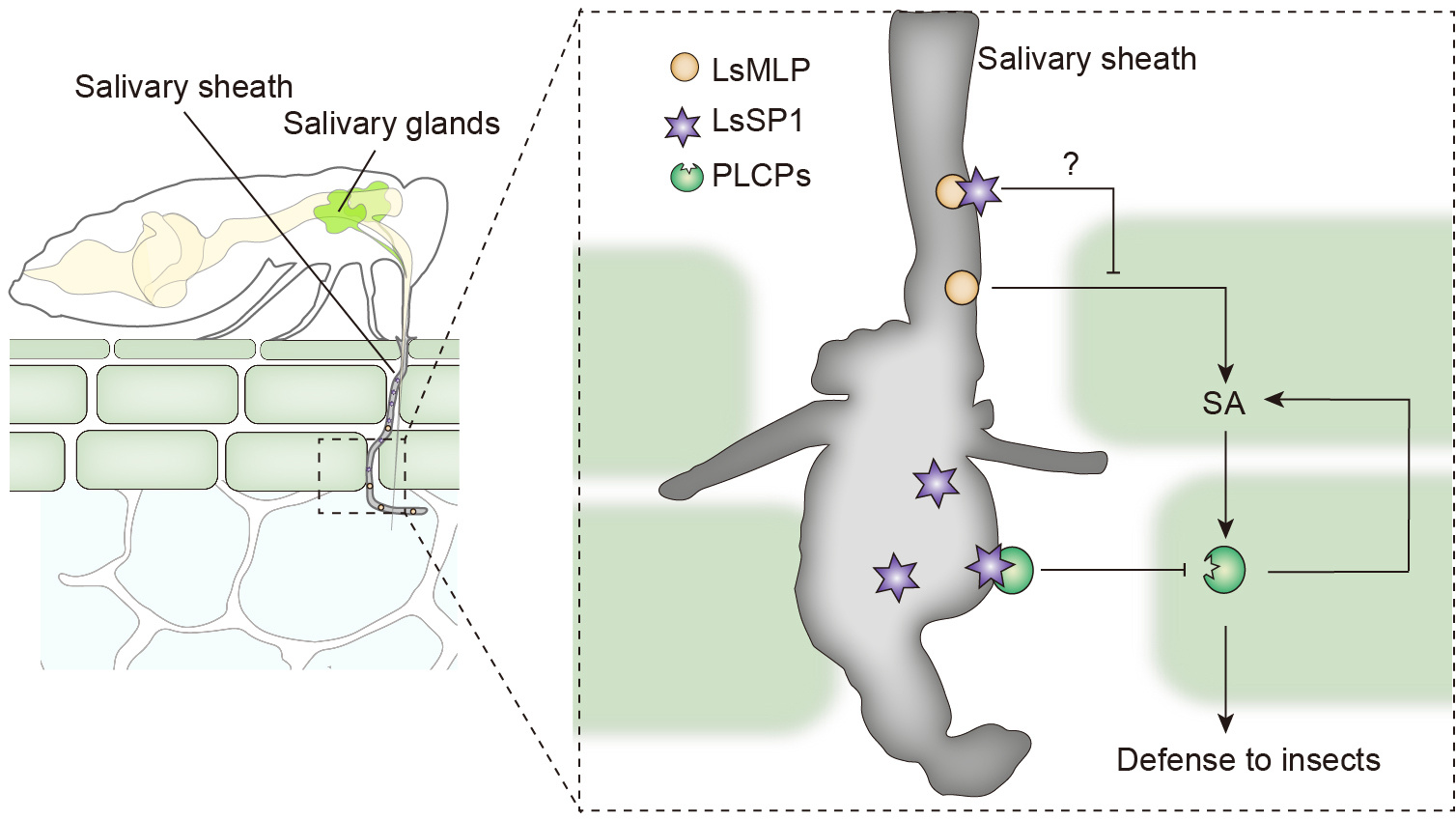
Figure 2. The preliminary model for LsSP1 suppresses plant defenses. This model is mainly drawn by our hypothesis, and needs additional experiments to validate.
Finally, we propose a preliminary model of complex interactions between rice plants and planthoppers (Figure 2). LsMLP is the main component of salivary sheath, which is indispensable for insect feeding. However, LsMLP and other salivary components can be perceived by host plants, thus triggering the SA-induced defense responses, including the up-regulation and secretion of PLCPs. The increased PLCP levels in turn activate the SA signaling, thus establishing a positive feedback loop to inhibit insect feeding. Meanwhile, the salivary sheath LsSP1 is secreted during planthopper feeding and located in the plant apoplast. It potentially prevents the activation of plant defenses by masking the LsMLP. Also, LsSP1 interacts with multiple PLCPs and acts as a sponge to restrict these immune proteases to salivary sheath and inhibit their proteolytic activities. Noteworthy, this model is mainly drawn by our hypothesis, and needs additional experiments to validate.
In further study, we will test whether this model is right? Whether LsSP1 is capable of masking the LsMLP? How PLCP active rice defense? What other PLCPs functions in plant-insect interaction, in addition to OsOryzain? What is the other role of LsSP1 in insect feeding?
References
- Huang, H.J., Wang, Y.Z., Li, L.L., Lu, H.B., Lu, J.B., Wang, X., Ye, Z.X., Zhang, Z.L., He, Y.J., Lu, G., Zhuo, J.C., Mao, Q.Z., Sun, Z.T., Chen, J.P., Li, J.M., Zhang, C.X., 2023. Planthopper salivary sheath protein LsSP1 contributes to manipulation of rice plant defenses. Nature Communications.
- Jiang Y-J, Zhang C-X, Chen R-Z, He S-Y. Challenging battles of plants with phloem-feeding insects and prokaryotic pathogens. Proc Natl Acad Sci U S A 116, 23390-23397 (2019).
- Guo H, et al. An Aphid-Secreted Salivary Protease Activates Plant Defense in Phloem. Curr Biol, (2020).
- Ye W, et al. A salivary EF-hand calcium-binding protein of the brown planthopper Nilaparvata lugens functions as an effector for defense responses in rice. Scientific reports 7, 40498 (2017).
- Huang HJ, et al. Salivary DNase II from Laodelphax striatellus acts as an effector that suppresses plant defence. New Phytol 224, 860-874 (2019).
- Chen CY, et al. An effector from cotton bollworm oral secretion impairs host plant defense signaling. Proc Natl Acad Sci USA 116, 14331-14338 (2019).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in