Uemura and co-workers from Kyoto University recently published a paper with Nature Communications entitled “Sequence-regulated copolymerisation based on periodic covalent positioning of monomers along one dimensional nanochannels.”
https://www.nature.com/articles/s41467-017-02736-1
The work describes an artificial synthetic route to producing sequence controlled polymers. These defined polymers are desirable due to their properties and use in various applications, such as, data storage. The inspiration behind the paper is the elegant manner in which peptide chains are produced from the translation of RNA in a ribosome. The authors realised that using informative templates in confined spaces may be key to forming precisely controlled sequences of synthetic polymers. A vast amount of research has already be dedicated to forming defined polymers, but controlling vinyl based polymers that are generated by radical chain growth polymerisation has always been elusive.
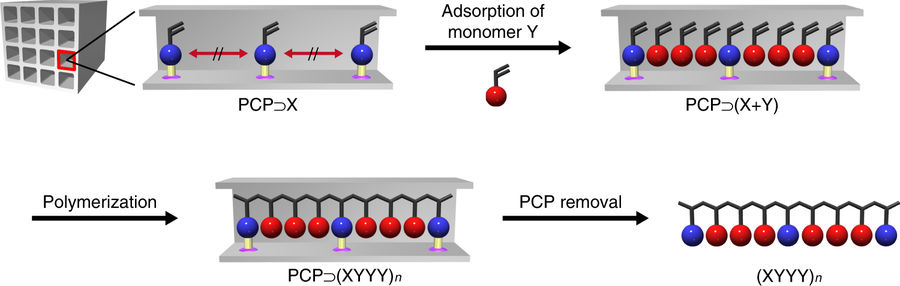
Here the authors assembled a kagomé-type porous coordination polymer (PCP) from copper ions and isophthalate ligands [Cu(styrene-3,5-dicarboxylate)]n. The PCP consists of styryl groups periodically placed in the framework. Both triangular and hexagonal channels are present in the PCP. Using single crystal X-ray diffraction techniques it was found the triangular channels are 4 Å in diameter and the hexagonal channels are 6 Å in diameter. The hexagonal channels are just large enough to incorporate acetonitrile as free guest monomers and thus radical polymerisation between the sytryl groups and acetonitrile occurred when AIBN was used as the initiator. During polymerisation the PCP retained its structure, which was confirmed by NMR and XRPD.
In order to free the polymer formed within the PCP framework, HCl and heat were used to decompose the nanochannels. However, the PCP can be recycled by recrystallization of unreacted styrene-3,5-dicarboxylate with copper ions.
The compositions of the polymers generated varied depending on the ratio of acetonitrile to styryl groups, but the polymers produced have high molecular weights and fairly narrow polydispersity indices. The predominant polymer sequence observed was XYYY. Computational analysis was carried out to investigate the mechanism behind these copolymerisation reactions.
The route of exploiting crystalline templates, constructed by self-assembly of metal ions and ligands, to form sequence controlled vinyl copolymers is scalable, recyclable and generalizable (successful when acetonitrile was substituted by methyl vinyl ketone). Additionally, the use of these PCP templates to imprint their periodic structure within the resulting polymer sequence is easier than using polymeric templates that require complex organic synthesis or small molecular templates that can potentially lead to cross-linking between the polymer chains. Therefore, this methodology allows a material platform to produce many periodic sequences for copolymers, which have desirable applications.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in