Linking Two Holey [60]Fullerenes
Published in Chemistry
![Linking Two Holey [60]Fullerenes](https://images.zapnito.com/cdn-cgi/image/metadata=copyright,fit=scale-down,format=auto,quality=95/https://images.zapnito.com/users/427349/posters/1d3f9017-664c-47cb-bcf9-0402e45eb44d_large.png)
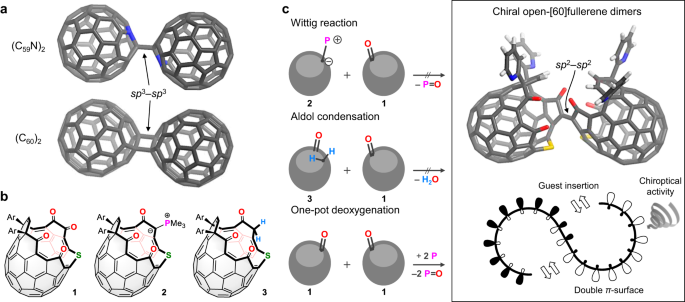
Natural carbon allotropes adopt a variety of topologies depending upon bond connection modes of carbon atoms, thus generating different shapes and dimensionalities such as sphere (fullerene, 0D), tubular (nanotube, 1D), and sheet (graphene, 2D). Coalescence of fullerenes is a fundamental thinking model when one aims to construct carbon allotropes that are otherwise unavailable in nature. A minimum unit of coalesced fullerenes is a fullerene dimer. Hence, organic chemists have continuously paid considerable attention for synthesizing and structurally characterizing fullerene dimers such as (C60)2 and (C59N)2, in which two fullerene cages are linked by one or two single bonds, consequently disconnecting π-conjugation between the two cages. In contrast, coalesced fullerene dimers with full π-conjugation have been found only as transiently-producible species in a physical process under high electron voltage through TEM (transmission electron microscopy). Since serial coalescence of fullerenes has been believed to reach long-envisioned hypothetical fullerene-based nanomaterials, such as fullerene sponge or fullerites, it is of particular importance to set a rational strategy toward synthetic interfullerene allotropes.
In the paper "Synthesis of inter-[60]fullerene conjugates with inherent chirality" recently published in Nature Communications, we focused on holey [60]fullerenes with functional groups on their hole. This type of fullerenes is usually called as open-[60]fullerenes which are π-conjugated hosts utilized in supramolecular single-molecule science as well as electron acceptors in solar cell application. Importantly, open-[60]fullerenes possess inherent chirality (fullerene chirality) which was very recently witnessed as the third function of them and potentially shows better chiroptical activities than most chiral organic materials. Thus, our goal had been set to get an inherently-chiral synthetic interfullerene conjugate. Owing to high designability on molecular and electronic structures of open-[60]fullerenes, we finally achieved the synthesis of non-conjugated and conjugated chiral inter-[60]fullerene hybrids via a phosphine-mediated one-pot deoxygenative C=C bond formation (please see details in manuscript) where the key intermediate is 1-phosphonium-3-carbabetaine (see Behind The Paper and Commun. Chem. 2020, 3, 90.). Reflecting the inherent chirality of the open-[60]fullerene, the non-conjugated inter[60]fullerene hybrid exhibited characteristic heterochiral recognition whose structure was crystallographically and spectroscopically determined. In addition, the inter-[60]fullerene conjugate showed strong intercage interactions during 6-electrons uptake as well as excellent chiroptical activity with a gigantic dissymmetry factor of 0.21 which breaks the record of known chiral organic molecules.
![Synthetic inter-[60]fullerene hybrids](https://images.zapnito.com/cdn-cgi/image/metadata=copyright,fit=scale-down,format=auto,quality=95/https://images.zapnito.com/uploads/8UqsVPOaT7qcQIwTChh7_fig-behind-the-paper.png)
Yoshifumi Hashikawa,* Shu Okamoto & Yasujiro Murata*
Synthesis of inter-[60]fullerene conjugates with inherent chirality.
Nat. Commun. 2024, 15, 514.
DOI: 10.1038/s42004-020-00340-x
More details in Nature Communications via https://www.nature.com/articles/s41467-024-44834-x
Our research group: https://www.scl.kyoto-u.ac.jp/~kouzou/en/index.html
Follow the Topic
-
Nature Communications

An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026
Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in