Main element chemistry enables gas-cylinder-free hydroformylations
Published in Chemistry
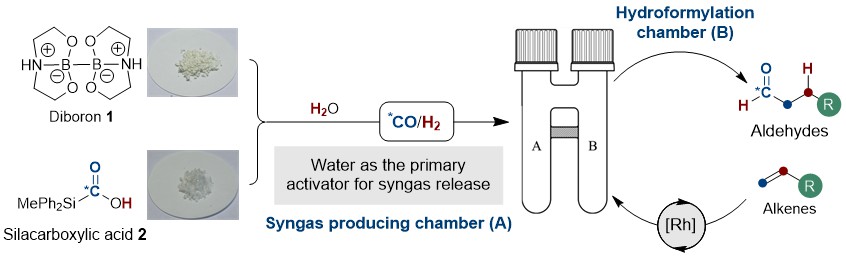
After an incident in 2010 involving a pressurized carbon monoxide (CO) cylinder, the Skrydstrup group has strived towards the development of methods for the safe handling of toxic and harmful low-molecular-weight gasses (e.g. carbon monoxide, dihydrogen, hydrogen cyanide, and ethylene)1. As a result, the two-chamber technology, COware®, emerged along with the development of solid gas precursors and their applications2. The advantage is clear; a laboratory chemist is able to safely measure a stoichiometric amount of a highly reactive gas as a crystalline, bench-stable, and easy-to-handle precursor in one chamber (A) where, in presence of an activator, the toxic gas is liberated and consumed in the other chamber (B), thus completely avoiding the need for costly infrastructure for the handling of these gasses.
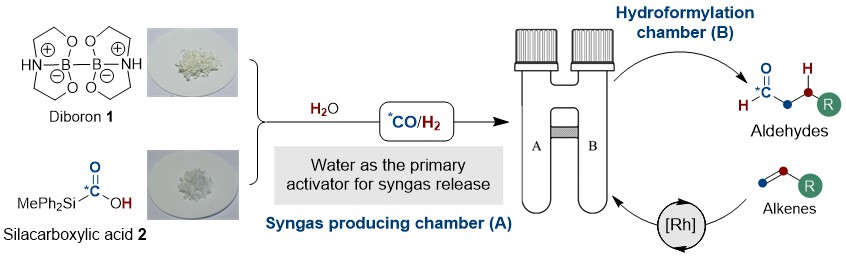
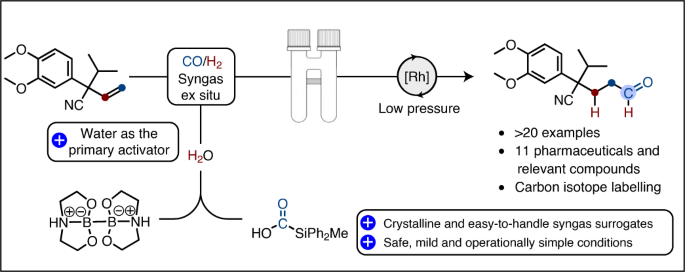
The hydroformylation reaction involves the conversion of olefins to aldehydes via the addition of syngas (CO/H2 mixture) and is mediated by a transition metal (Co or Rh). Industrially, this process is of the outmost importance, producing approximately 12 million tons of the highly valued oxo-product annually and has, since its discovery by Otto Roelen in 1938, become one of the most applied homogeneous transition metal catalysed reaction in the world! Regardless of its importance, this transformation has found little use in the hands of laboratory chemists, like ourselves, primarily due to the toxic and harmful nature of the gases involved. With this limitation in mind, we questioned whether the combination of two solid gas precursors, diboron 13 that efficiently reduces water for H2 gas formation, and silacarboxylic acid 24, which releases CO gas upon activation, could provide a convenient source for stoichiometric syngas generation. To cut a long story short; yes, yes it can. In fact, we realised that the borohydride intermediate resulting from the reduction of water with diboron 1 is able to provide the necessary activation of the silacarboxylic 2 for CO release, thus rendering water as the primary activator for syngas generation! Furthermore, combining this strategy with that of our two-chamber technology and already established catalysis5, we demonstrate the synthetic utility over a range of terminal olefins with varying degree of complexity (24 examples), including a total synthesis. Perhaps the most important application of this method is its adaptability to carbon isotope labelling of pharmaceutically important molecules (8 examples) and relevant compounds thereof (3 examples) via the use of 13CO generated from the readily available carbon-13 version of silacarboxylic acid 26. We believe that this efficient and operationally simple protocol for the hydroformylation of terminal alkenes under mild conditions, without the need for gas cylinders and autoclaves, will provide an important and complimentary tool for drug discovery and development programs in both industry and academia and we look very much forward to follow its future applications.
Link to article: Main element chemistry enables gas cylinder-free hydroformylations
- Ravn, A. K., Johansen, M. B. & Skrydstrup, T. Controlled Release of Reactive Gases: A Tale of Taming Carbon Monoxide. Chempluschem 85, 1529–1533 (2020).
- Friis, S. D., Lindhardt, A. T. & Skrydstrup, T. The Development and Application of Two-Chamber Reactors and Carbon Monoxide Precursors for Safe Carbonylation Reactions. Acc. Chem. Res. 49, 594–605 (2016).
- Flinker, M. et al. Efficient Water Reduction with sp3-sp3 Diboron(4) Compounds: Application to Hydrogenations, H-D Exchange Reactions, and Carbonyl Reductions. Angew. Chemie Int. Ed. 56, 15910–15915 (2017).
- Friis, S. D., Taaning, R. H., Lindhardt, A. T. & Skrydstrup, T. Silacarboxylic acids as efficient carbon monoxide releasing molecules: Synthesis and application in palladium-catalyzed carbonylation reactions. J. Am. Chem. Soc. 133, 18114–18117 (2011).
- Seiche, W., Schuschkowski, A. & Breit, B. Bidentate Ligands by Self-Assembly through Hydrogen Bonding: A General Room Temperature/Ambient Pressure Regioselective Hydroformylation of Terminal Alkenes. Adv. Synth. Catal. 347, 1488–1494 (2005).
- Nielsen, D. U., Neumann, K. T., Lindhardt, A. T. & Skrydstrup, T. Recent developments in carbonylation chemistry using [13C]CO, [11C]CO, and [14C]CO. J. Lab. Comp. Radiopharm. 61, 949–987 (2018).
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in