Mechanochromism Induced through the Interplay between Excimer Reaction and Excited State Intramolecular Proton Transfer
Published in Chemistry
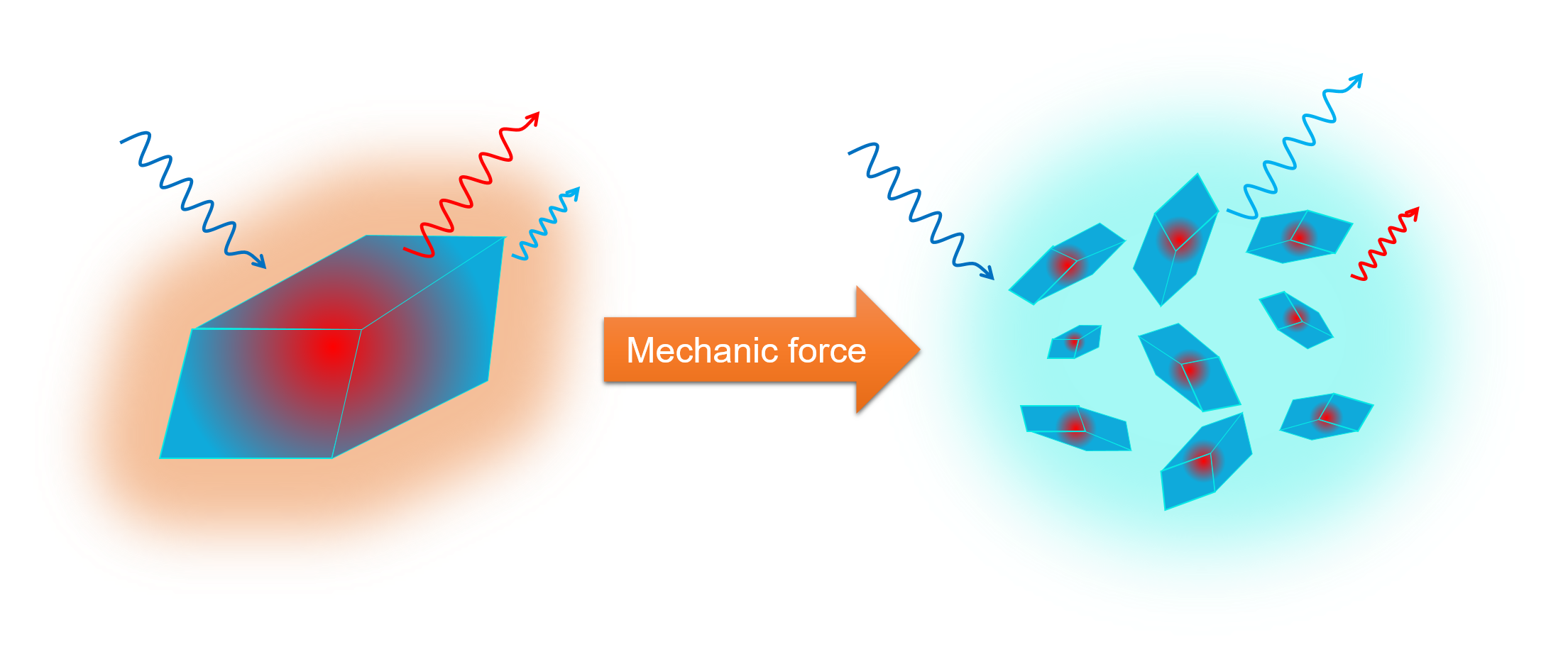
Mechanochromism, the change of absorption or emission intensity induced by mechanical force, has drawn much interest for its versatile applications such as pressure sensors, fluorescent switch and data recording devices. However, the capital origin of mechanochromism for organic compounds is still in vague owing to the complicated interaction in solid state such as π-π interaction, excited-state intermolecular electron/proton transfer and excimer formation, etc., leading to the hardness to systematically predict the emission properties and hence the fabricated devices. Herein we propose a rational perspective to elucidate the origin of the mechanochromism from our studied compounds, which is the ratio between the crystal surface and kernel regions since the air/solvent-solid interface prohibits the molecular packing on the crystal surface. Therefore, applying mechanical force on crystal rises the surface to volume ratio, increasing the random packing molecules and resulting in the change of the emission color.
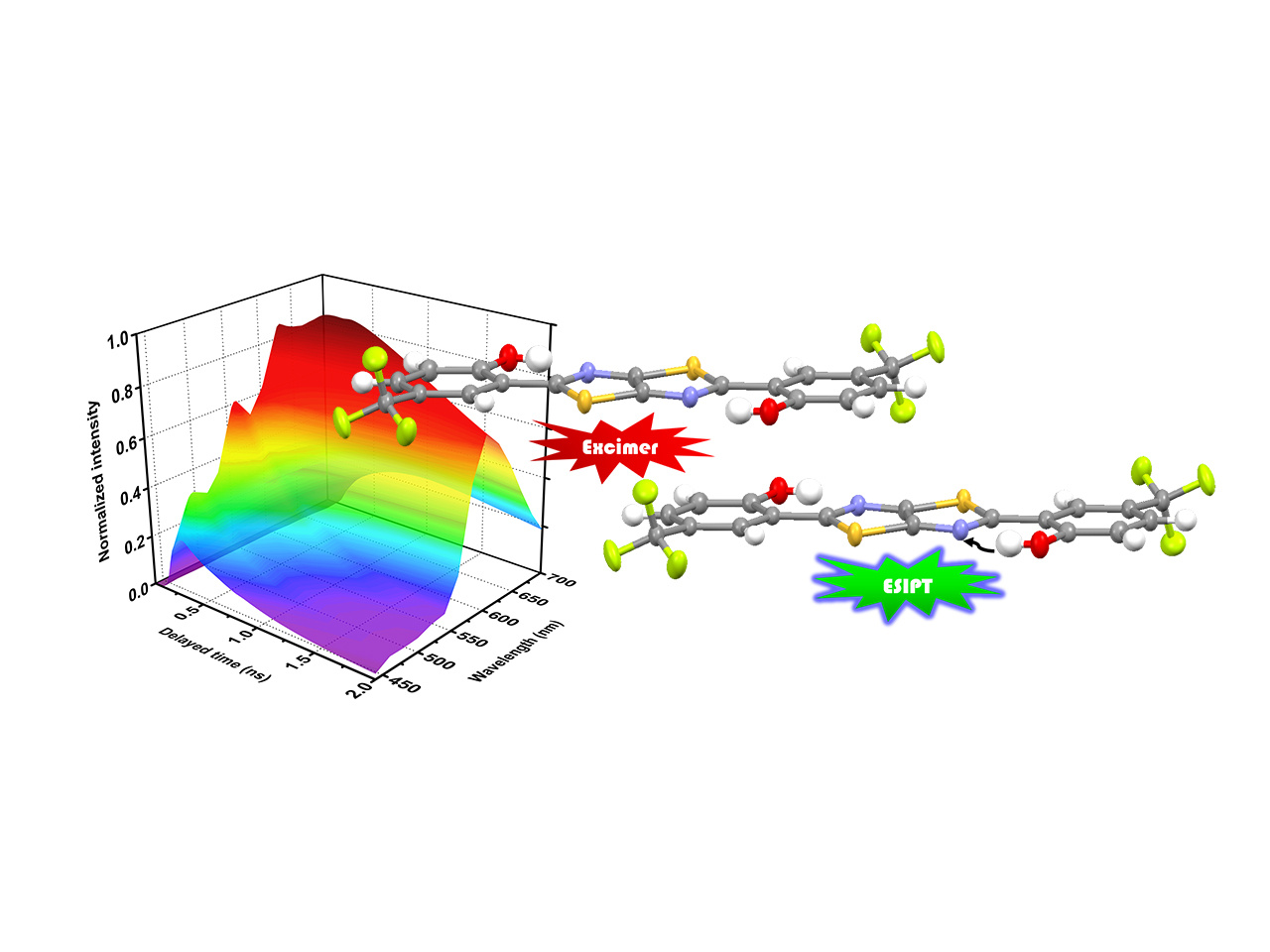
Actually finding this phenomenon is in serendipity. At first we synthesized the series of dithiazole molecules for studying the excited-state proton transfer (ESIPT) properties. Intriguingly, one of the molecules CF3-HTTH (2,2'-(thiazolo[5,4-d]thiazole-2,5-diyl)bis(4-(trifluoromethyl)phenol) in pristine powder and solid crystal exhibited panchromatic emission from 420 nm to 750 nm, resulted from the coupling between the ESIPT and excimer formation reactions, the phenomenon of which has never been observed before. The associated formation dynamics is verified by the time-resolved emission spectroscopy and kinetic simulation, showing the parallel kinetic model of the ESIPT coupled excimer reaction.
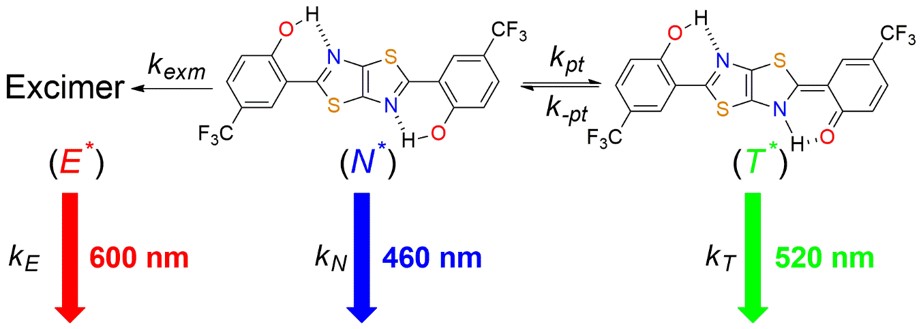
Before realizing the surface-kernel effect, there’s an additional decay channel (τ = 1.7 ns) which cannot be explained by the kinetic model. Furthermore, the portion of this decay pathway increases from solid crystal to pristine powder, correlating well with the ratiometric change of intensity in steady-state emission spectra in different morphology of solid state. Based on these results, we conclude that the surface-to-volume ratio should play an important role in the corresponding mechanism. With the aid of two-photon excitation microscopy, we can separate the surface and kernel emission of the crystal and the results coincide our presumption. The 1.7 ns decay pathway arises from the crystal surface with only ESIPT taking place since the excimer formation relies on the well-ordered packing structure interrupted on the crystal surface. In comparison, the crystal kernel exhibit dominant excimer formation coupling with ESIPT reaction. Incorporating this surface-kernel effect into ESIPT coupled excimer reaction, all the novel behaviors of CF3-HTTH in solid state can be unriddled and the hypothesis of surface-kernel effect is solidified. We believe that this effect should be adaptable for most mechanochromic and aggregation-induced emission (AIE) materials, clarifying the rationale of multi-emission in solid state.
More information about the stories of mechanochromism, ESIPT and excimer formation is in this article: Wei, Y.-C., Zhang, Z., Chen, Y.-A., Wu, C.-H., Liu, Z.-Y., Ho, S.-Y., Liu, J.-C., Lin, J.-A., Chou, P.-T. Mechanochromism induced through the interplay between excimer reaction and excited state intramolecular proton transfer. Communications Chemistry 2, 10 (2019).
DOI: https://doi.org/10.1038/s42004-019-0113-8
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Sustainable waste management through polymer upcycling
Publishing Model: Open Access
Deadline: May 31, 2026


![After the Paper | Photophysics in Thiazolo[5,4-d]thiazole Phenol (TzTz-ph) Crystalline Aggregates](/cdn-cgi/image/metadata=copyright,fit=scale-down,format=auto,quality=95,width=256,height=256/https://public-storage.zapnito.com/hWXdEkJ28XW7jCwVyTMaGaWOTNc20-XY58VLjMRohmc)
Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in